Behind the Paper: Chronic pancreatitis pain is not one disease; and that matters
Published in Healthcare & Nursing and Biomedical Research
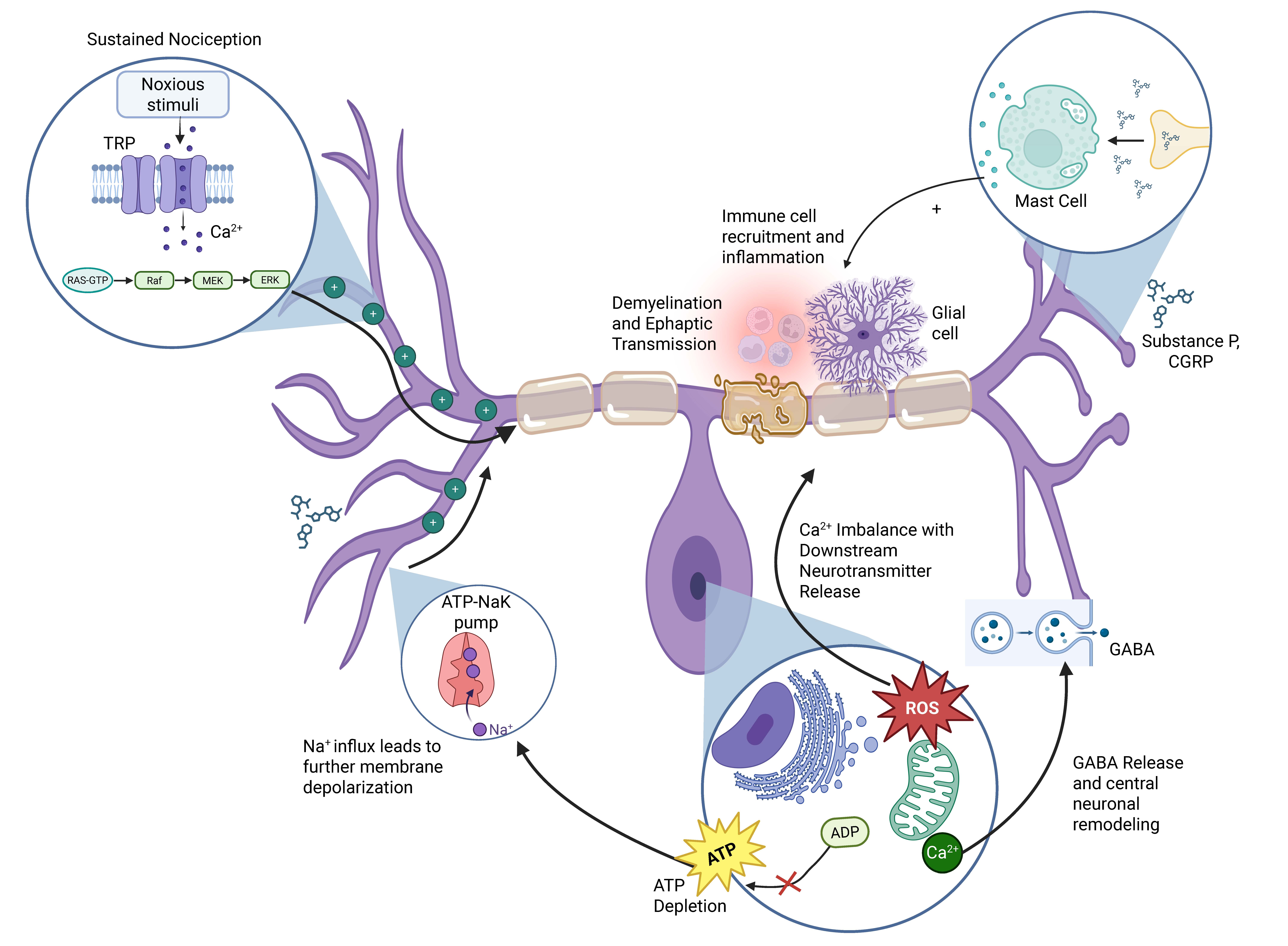
When we began working on this review, we were not starting from the sense that the field lacked important work. In many ways, the opposite was true. There was already thoughtful clinical research on pain patterns, treatment burden, and patient outcomes. There were also emerging mechanistic insights from genetics, neurobiology, and molecular studies. But as we read across these areas together, we kept returning to the same impression: the pieces were there, yet they did not fully come together.
That was what made this paper feel worth writing.
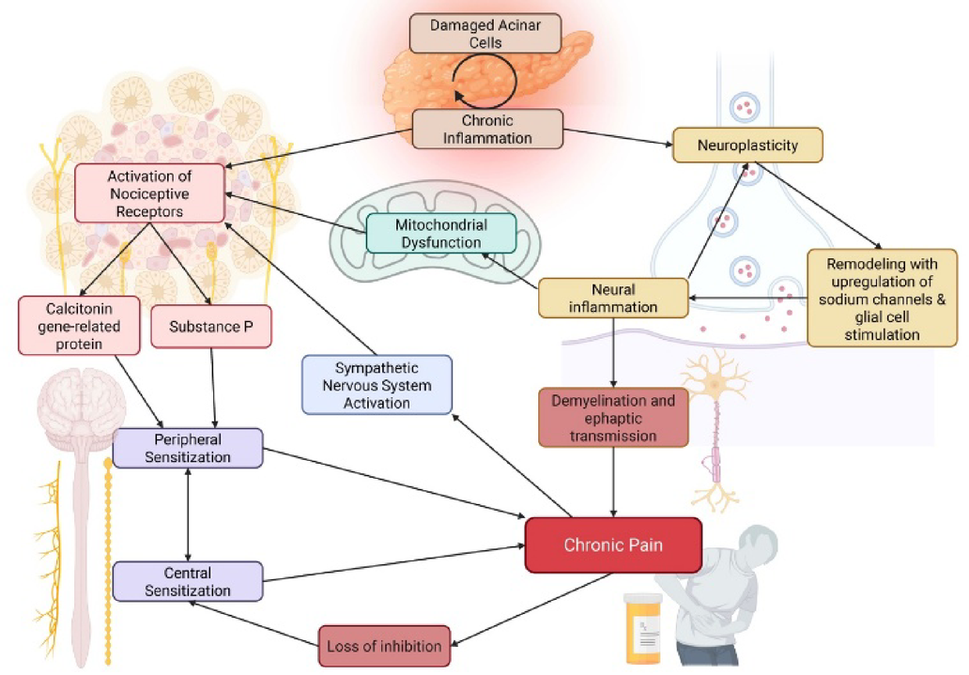
Chronic pancreatitis pain is one of those problems that resists simple framing. It does not fit neatly within anatomy alone, and it is not adequately captured by symptom severity alone either. It unfolds over time, is shaped by treatment histories and lived burden, and likely reflects overlapping processes rather than a single dominant mechanism. The more we discussed the literature, the more we felt that the real question was not only how to describe pain more carefully, but whether the categories we use are truly aligned with the biology we hope to understand.
That question became the center of the review.
What made the writing process especially meaningful was the opportunity to think across different forms of expertise. The clinical perspective kept bringing us back to what pain actually means in practice: disrupted daily life, repeated care decisions, opioid exposure, uncertainty around interventions, and the cumulative burden that patients carry over time. The omics perspective opened a different window. It encouraged us to ask whether some of the heterogeneity clinicians see every day is not simply complexity to be managed, but biology that remains insufficiently resolved by our current frameworks.
Some of the most important insights in the paper came from sitting with that tension rather than trying to smooth it away.
One of the clearest lessons for us was that integration is not valuable for its own sake. It is easy to be drawn to the promise of more molecular layers, more modalities, and more sophisticated analysis. But more data does not automatically create more clarity. If the phenotype itself is broad, unstable, or strongly shaped by treatment and care pathways, then even a technically impressive multimodal approach may remain more descriptive than truly explanatory. That realization shaped the tone of the review just as much as the content. We wanted to write with optimism about what integrated omics could offer, but also with honesty about what the field still needs in order to make those approaches meaningful.
In that sense, this paper became more than a review of published work. It became a way of articulating a problem more clearly. We found ourselves returning again and again to the idea that chronic pancreatitis pain is biologically heterogeneous, clinically consequential, and still only partially captured by the tools and categories we most often rely on. That is not a reason for pessimism. If anything, it is a reason to be more precise about the next questions we ask.
Looking back, what feels most important about this paper is not that it offers a final framework, but that it reflects a moment of clearer focus. It helped bring clinical and molecular thinking into the same conversation. It also reinforced something that often becomes obvious only through collaboration: that some of the best scientific questions emerge not when one perspective dominates, but when different perspectives make the problem sharper.
If there is one idea we hope readers carry forward, it is that chronic pancreatitis pain will not be understood through anatomy alone, and it will not be transformed through omics alone. The real opportunity lies in connecting molecular insight to clinically meaningful pain phenotypes in ways that are interpretable, testable, and useful for patients.
That, for us, was the story behind the paper: not the feeling that the field had arrived at an answer, but the recognition that it was finally ready to ask a better question.
Follow the Topic
-
Inflammation Research
Inflammation Research (IR) publishes peer-reviewed papers on all aspects of inflammation and related fields including histopathology, immunological mechanisms, gene expression, mediators, experimental models, clinical investigations and the effect of drugs.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in