Behind the Paper: Engineering deutenzalutamide to reduce CNS-related toxicity—especially fatigue, falls and seizures
Published in Cancer and Biomedical Research
One of the most difficult moments in treating metastatic castration-resistant prostate cancer (mCRPC) is when the disease progresses after widely used therapies such as abiraterone and (for many patients) docetaxel. At that point, the question is no longer only “what works?”, but also “what can a patient realistically stay on?”
For long-lasting oral treatments, the adverse effects that most often limit adherence are not always the ones that look most dramatic in a table. In everyday practice, CNS-related toxicities—particularly persistent fatigue, as well as dizziness, cognitive slowing, falls, and the rare but feared risk of seizures—can be decisive. These effects matter because mCRPC is frequently managed like a chronic disease: patients may take therapy for months or years, while also managing pain, sleep, comorbidities, and polypharmacy.
This real-world concern shaped the development of deutenzalutamide (HC-1119; deuterated enzalutamide) and the design of our phase III HC-1119-04 trial. While the study demonstrated meaningful disease control (improved rPFS), what I want to share here is the “behind the scenes” safety story—how a metabolism question turned into a CNS-safety hypothesis, and how the data surprised us in a good way.
Why deuteration? A small chemical change with a safety goal
Deutenzalutamide is based on a straightforward medicinal chemistry idea: replace selected hydrogen atoms with deuterium (a stable, naturally occurring “heavy hydrogen”). This can slow certain metabolic steps (the “kinetic isotope effect”), leading to a different balance of parent drug and metabolites over time.
But deuteration was not pursued as an academic exercise. It was intended to address a clinical problem: CNS-related toxicity seen with some AR inhibitors, including fatigue, falls, and in rare cases seizures.
A commonly discussed mechanism for seizure risk with enzalutamide is inhibition of the GABA-A–gated chloride ion channel in the brain (GABA is one of the brain’s main inhibitory pathways). If a compound (or one of its metabolites) reaches sufficient concentrations in the CNS and inhibits GABA-A, excitability can increase—raising seizure risk and potentially contributing to other CNS symptoms.
So our core development question became:
Can we keep robust AR inhibition in the body while reducing “problematic” CNS exposure?
The development journey: M0 vs M2—and an early worry we had to test
During development, we focused on two key circulating forms:
- M0: the parent compound
- M2: a major active metabolite
From in vitro work, M0 and M2 showed similar anti-tumor activity at the androgen receptor. That initially sounds reassuring—until you consider the CNS.
Deuteration changed the metabolic balance:
- M2 decreased
- M0 increased
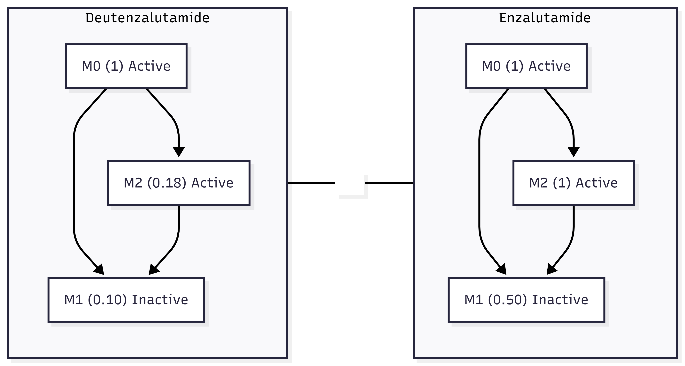
Fig. 1. Deuteration shifts the steady state plasma concentration ratios (M0 normalized to 1). M1 is inactive at both AR and GABA-A.
At first, this raised a serious concern for us. A reasonable worry was: if we reduce M2 formation but increase M0, could the overall active exposure (M0 + M2) remain similar in plasma yet unintentionally increase brain exposure, especially if we assumed one form might cross the blood–brain barrier more easily?
This is the kind of question that cannot be answered by theory alone. We needed actual measurements.
The key “aha”: M2 is more brain-penetrant than M0—and more potent at GABA-A inhibition
When we examined distribution and pharmacology in the nonclinical development package, two findings clarified the picture:
- M2 showed ~3-fold greater brain penetration than M0 across the nonclinical species datasets used during development (mouse, rat, and monkey).
- In electrophysiology studies, M2 was ~3-fold more potent than M0 at inhibiting GABA-A channel function.
Together, these results changed how we interpreted the metabolic shift:
- Even if M0 increased in plasma, reducing M2 could still meaningfully reduce CNS risk, because M2 is both better at getting into the brain and stronger at inhibiting GABA-A.
In other words, the metabolite we were reducing (M2) appeared to be the one with a more concerning CNS profile.
To communicate this simply for non-specialists: we learned that not all “active” drug forms are equal. Two molecules can both block the androgen receptor (good for cancer control), while one of them is much more likely to enter the brain and interfere with brain signaling (bad for CNS tolerability).
Translating to humans: why we expected lower CNS burden
Using development data, we estimated and compared brain exposure to (M0 + M2):
At similar plasma exposure (M0+M2), the calculated brain exposure (M0+M2) was ~40% higher with enzalutamide than with deutenzalutamide, consistent with the substantially lower M2 levels on deutenzalutamide.
The underlying logic is important: CNS risk is not just “how much drug is in plasma.” It is more like:
(How much reaches the brain) × (how strongly it affects GABA-A)
When M2 is both more brain-penetrant and more potent at GABA-A inhibition, reducing M2 becomes a direct strategy to lower the combined seizure/fatigue/fall risk potential.
What we observed clinically: signals consistent with a favorable CNS-related profile
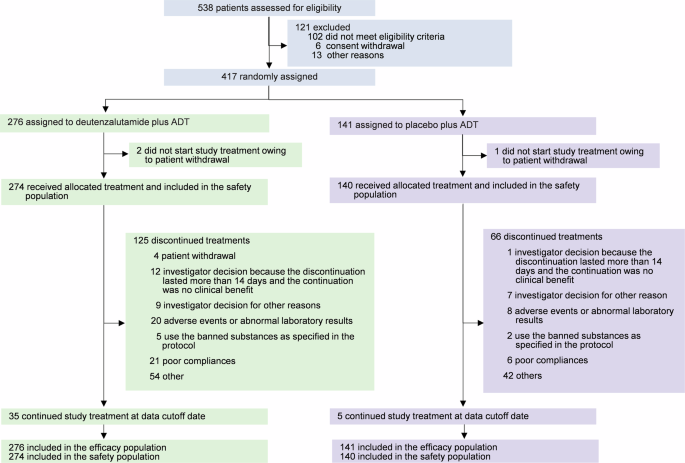
In the randomized, double-blind phase III HC-1119-04 trial (417 patients; 276 received deutenzalutamide), deutenzalutamide significantly improved rPFS (HR 0.58). From a safety standpoint, several observations stood out, particularly in areas relevant to CNS tolerability.
Overall treatment-related safety
- Treatment-related grade ≥3 adverse events occurred in 22.3% with deutenzalutamide vs 15.0% with placebo.
- The most common treatment-related adverse event was anemia (any grade 21.2% vs 17.9%; grade 3/4 6.6% vs 2.9%).
CNS-related observations: no seizures or falls reported
Notably, no seizures or falls were reported. Absence of events in a single trial must always be interpreted cautiously—rare events can require very large populations or longer follow-up to fully characterize. However, given longstanding concerns about CNS-related tolerability within this therapy class, this observation is a clinically meaningful signal.
Fatigue (“asthenic conditions”): a potential differentiator worth watching
Fatigue deserves special emphasis. Across AR-targeted therapies, fatigue is among the most common reasons patients say, “I can’t keep taking this.” In mCRPC—where many patients are older and already managing baseline fatigue, reduced reserve, and multiple medications—tolerability is not a secondary issue. It directly affects the ability to continue therapy.
In HC-1119-04, fatigue was captured under “asthenic conditions.” When viewed against historical enzalutamide reports (e.g., AFFIRM and prescribing information), enzalutamide reported asthenic conditions around 51% (with 9% grade 3/4). In contrast, deutenzalutamide showed 22% for all grades, with 3.3% grade 3/4.
It is important to be careful here: cross-trial comparisons have real limitations. Differences in patient populations (HC-1119-04 trial enrolled a later-line, generally more advanced cohort), prior treatments, trial design, and adverse-event reporting can materially affect observed rates. Still, the magnitude of the reduction in reported fatigue/asthenic conditions relative to historical enzalutamide experience is encouraging—and consistent with the original deuteration rationale.
What this means (and what it doesn’t)
These results do not mean CNS-related symptoms can be ignored—fatigue in advanced cancer is multifactorial, and careful monitoring remains essential. But the development story of deutenzalutamide offers a useful example of how a drug can be engineered with tolerability in mind:
- Start from a known effective mechanism (AR inhibition)
- Identify a limiting toxicity domain (CNS-related fatigue/falls/seizures)
- Understand which circulating forms drive that risk (M2 vs M0)
- Measure brain penetration and off-target CNS pharmacology (GABA-A)
- Then test the hypothesis in controlled clinical trials
As additional real-world data and longer-term follow-up become available, we will learn more about how these signals translate into everyday patient experience. For now, it is gratifying to see a development hypothesis—rooted in metabolism and CNS pharmacology—align with the clinical safety observations.
Article link: [https://www.nature.com/articles/s41392-026-02618-3]
Clinical trial registration: NCT03851640
Follow the Topic
-
Signal Transduction and Targeted Therapy
This is an international, peer-reviewed, open-access journal publishing articles related to signal transduction in physiological and pathological processes, alongside signal transduction-targeted therapeutics in the form of biological agents and small molecular drugs used to treat human diseases.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in