Crafting 'Smarter' Drug Testing to Facilitate Harm Reduction
Published in Chemistry, Biomedical Research, and General & Internal Medicine

Every investigation has a story!
Herein, we detail the unanticipated yet fortuitous events leading to our recent Harm Reduction Journal publication, titled “Harm Reduction in the Fourth Wave of the Opioid Epidemic: A Case Series Investigation of the Effectiveness of Definitive vs Presumptive Urine Drug Testing for Opioid Use Disorder with Co-Occurring Polysubstance Use.” 1
INTRODUCTION
Our inquiry presents the first peer-reviewed investigation to evaluate evidence-based effectiveness, medical necessity, and expense of 'smarter' drug testing2—using near-real-time mass spectrometry rather than less costly immunoassay testing to guide both clinical decision-making and harm reduction in substance use disorder patients with co-occurring polysubstance use disorder in a real-world, outpatient, buprenorphine MOUD (medication for opioid use disorder) setting.
Acknowledging that an illustration can provide an attractive alternative to a 2,500-word manuscript, we offer the following:
.png)
BACKGROUND
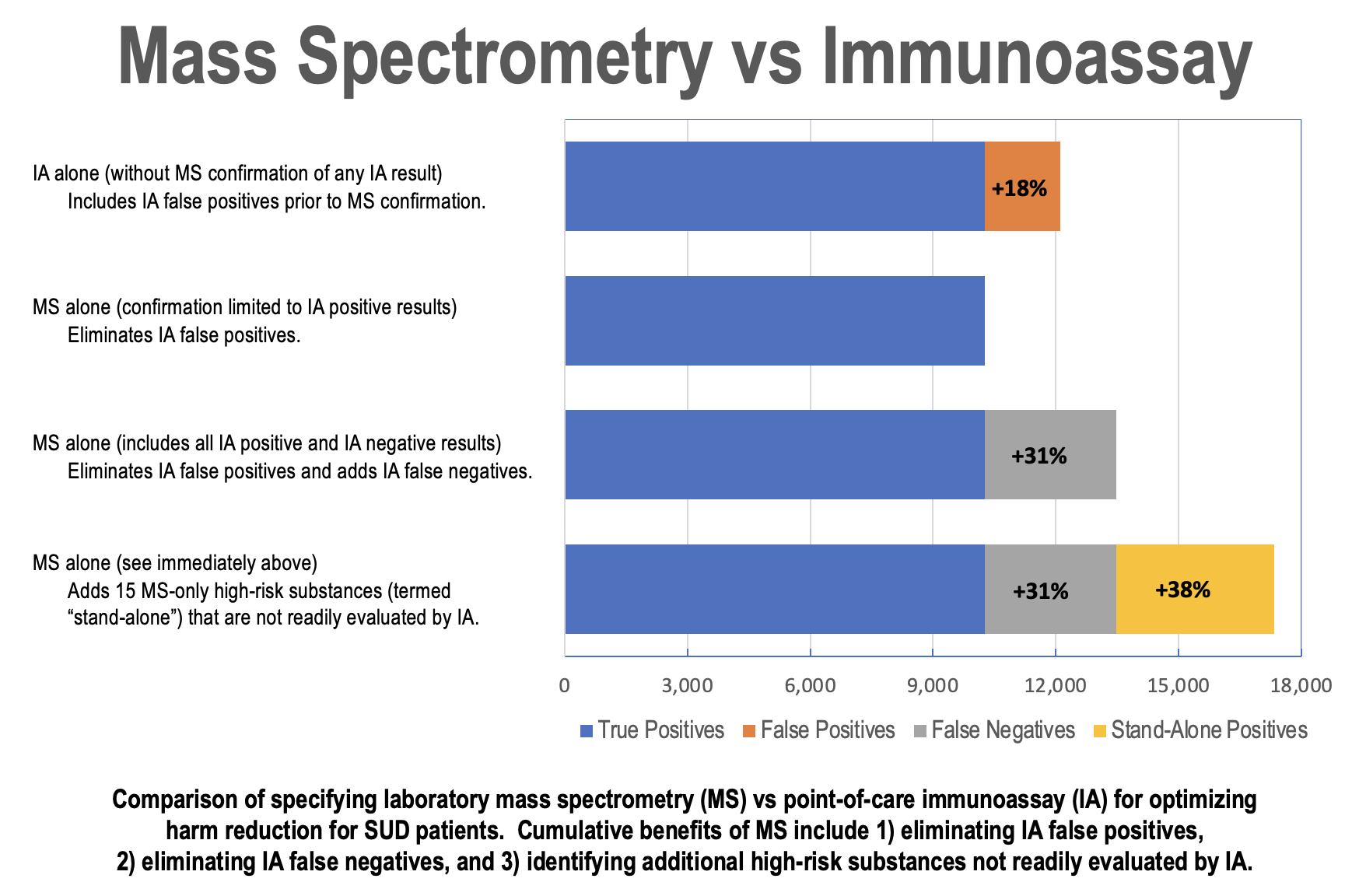
When we established our clinic network a decade ago, we initially adopted point-of-care immunoassay as our testing modality. Very quickly, however, we encountered two concerning trends. First, nearly two-thirds of point-of-care immunoassay samples revealed one or more substances requiring laboratory confirmation using mass spectrometry. Second, as part of the confirmation process, the laboratory using mass spectrometry to process our specimens reported finding a disturbing number of additional, unsuspected substances—which they detected as part of their routine, in-house quality assurance processes—in our test samples that a) we had not ordered, and b) represented drugs most often found in autopsies of fatal overdoses (ie, fentanyl, cocaine, amphetamine, methamphetamine, and benzodiazepines).
Although the available medical literature initially provided limited insight, we found several resources that pointed to the need for exploratory research on using point-of-care immunoassay vs laboratory/mass spectrometry technology for clinical management of substance use disorder patients.
- ASAM: A 2013 white paper introducing the concept of 'smarter' drug testing, followed in 2017 by a more detailed consensus statement on the topic.2
- Barthwell et al: Two publications (cited in manuscript), including a literature review, underscoring the importance of specifying laboratory testing as the primary methodology to minimize both false positives and false negatives.
- Five additional publications (cited in manuscript) spanning the last decade detailing the experiences of Millenium Health’s reference laboratory, which mirror our findings of significant, concerning discovery of unordered and unexpected substances detected in urine samples.
- Nora Volkow’s NIDA editorial perspective in 2023 in the New England Journal of Medicine calling for evaluation of the feasibility of expanding drug testing to include laboratory testing to facilitate harm reduction in real-world clinical settings.3
To the best of our knowledge, our investigation is the first peer-reviewed effort to address the potential for leveraging near-real-time laboratory testing to expand the realm of harm reduction for substance use disorders in a real-world, outpatient clinical setting.
INVESTIGATION
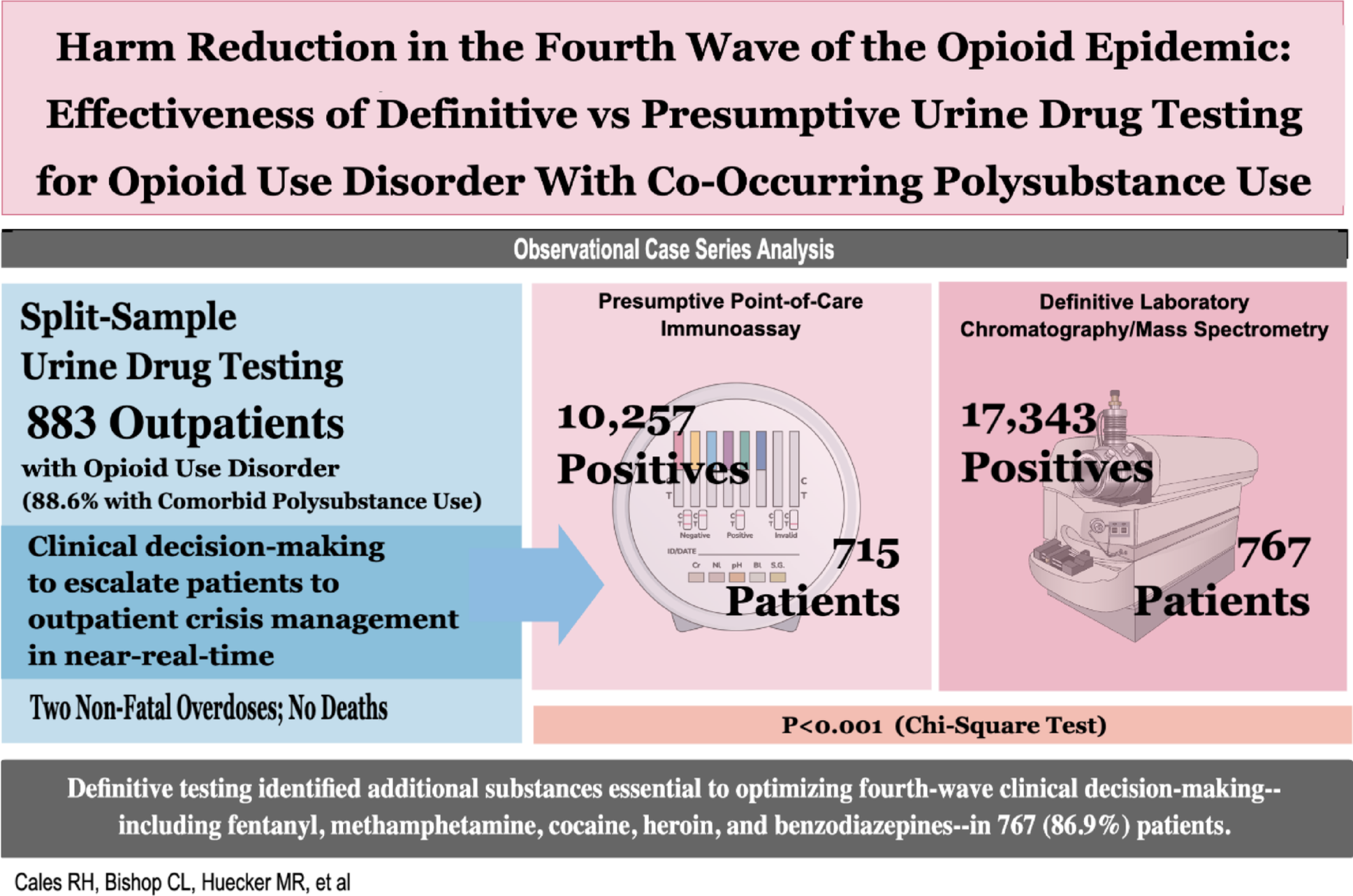
Our case series included all opioid use disorder patients, most of whom exhibited co-occurring polysubstance use, treated with buprenorphine MOUD for one year in a single outpatient clinic. Interventions entailed head-to-head, split-sample urine drug testing that compared definitive laboratory chromatography/mass spectrometry testing to less-costly presumptive point-of-care immunoassay testing, which is more susceptible to false-positive and false-negative results. Outcomes included return to use during treatment, morbidity, and mortality.
Urine Drug Testing
With regard to the superiority of laboratory mass spectrometry vs point-of-care immunoassay , our investigation is one of the first to report effectiveness of both the incidence and clinical relevance of using MS in a real-world, community clinic setting, not merely by eliminating false positives and false negatives but also by further leveraging mass spectrometry technology to identify other high-risk substances critical to optimizing harm reduction.
Harm Reduction
Although the term “harm reduction” most often appears in the context of substance use disorders and intravenous drug use, the public health community has long used the term to characterize a broad spectrum of preventive considerations, as illustrated by the following:

In this regard, our investigation is one of the first to respond to NIDA’s exhortation3 for clinicians to contribute to the ever-expanding scope of drug testing to include both real-time immunoassay test strips and near-real-time laboratory mass spectrometry testing to facilitate harm reduction.
Although some of our findings regarding the sensitivity and specificity of presumptive testing are neither new nor unexpected, we believe that our overall findings and resulting conclusions are unique in that they are the first to be reported a) in a single cohort of OUD patients with co-occurring polysubstance use employing split-sample presumptive vs. definitive testing and b) in the context of medical decision-making based on definitive UDT to facilitate harm reduction by guiding and escalating care in near real-time.
TAKEAWAYS
Our investigation offers three takeaways of particular relevance to real-world clinical practice:
- Although point-of-care immunoassay may suffice for population-based screening (initial detection and referral), medical decision-making (diagnosis and management) for MOUD is better served by near-real-time definitive laboratory testing.
- Our 'smarter' drug testing experience demonstrates that using non-quantitative, near-real-time testing for MOUD patients virtually eliminates the need for point-of-care testing—which requires confirmatory laboratory testing for more than half (57.6%) of all point-of-care tests due to one or more positive presumptive findings—yet costs only $10 more per patient encounter.
- These findings have facilitated the opportunity to integrate MOUD with harm reduction by refining 'smarter' definitive laboratory testing protocols to detect emerging and novel substances, including but not limited to kratom, xylazine, carfentanil, dexmedetomidine, tianeptine, levamisole, bromazolam, and nitazenes.
Hopefully, our exploratory findings will be serve to inform more robust additional research.
REFERENCES
- Cales RH, Bishop CL, Huecker MR, et al. Harm reduction in the fourth wave of the opioid epidemic: A case series investigation of the effectiveness of definitive vs presumptive urine drug testing for opioid use disorder with co-occurring polysubstance use. Harm Reduct J. 22, 187 (2025). https://doi.org/10.1186/s12954-025-01335-4
- Jarvis M, Williams J, Hurford M, et al. Appropriate use of drug testing in clinical addiction medicine. J Addict Med. 2017;11(3):163–173.
- Volkow ND, Califf RM, Sokolowska M, Tabak LA, Compton WM. (2023). Testing for fentanyl—Urgent need for practice relevant and public health research. NEJM. 388(224):2214-2217.
Follow the Topic
-
Harm Reduction Journal
This journal publishes research and commentary on approaches diminishing the harm of stigmatization, marginalization and criminalization of public health, human rights and social justice issues, as well as rebuking the de facto criminalization of marginalized and stigmatized communities.
Related Collections
With Collections, you can get published faster and increase your visibility.
Repairable damage: harm reduction and US policy
Recent shifts in US policy and the approach to public health have placed renewed pressure on communities that already face significant health and social challenges. Policy decisions increasingly prioritize criminalization or abstinence-based strategies over interventions that have been consistently shown to reduce harm. These choices reflect a broader trend of sidelining empirical evidence, with real-world consequences for public health.
As a result, morbidity and mortality have increased among people who use drugs, experience homelessness, or who are involved in the US criminal justice system. Rates of preventable illness and infectious disease are also rising. Rhetoric and stigma further undermine public trust and discourage engagement with health services, disproportionately affecting marginalized populations.
Harm Reduction Journal has commissioned a collection of articles to highlight how evidence-based harm reduction strategies – grounded in science, dignity and practical outcomes – can inform more effective, humane public health responses.
This Collection supports and amplifies research related to SDG 3, Good Health and Well-Being, SDG 5, Gender Equality, SDG 10, Reduced Inequalities, and SDG 16, Peace, Justice and Strong Institutions.
All submissions in this collection undergo the journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief. As an open access publication, this journal levies an article processing fee (details here). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief.
Publishing Model: Open Access
Deadline: Aug 18, 2026
Indigenous communities and harm reduction
Indigenous peoples, defined as the original inhabitants of lands with longstanding cultural, political, and spiritual ties that predate colonization, face unique challenges that stem from historical injustices, social marginalization, and systemic discrimination. These factors create complex environments where public health, human rights, and social justice intersect. By deepening our understanding of these challenges, we not only enrich academic discourse, but also pave the way for more effective, culturally relevant harm reduction strategies that resonate with Indigenous communities.
Significant advances have already been made in understanding the specific needs and realities of Indigenous communities concerning harm reduction. For instance, the integration of traditional healing practices with contemporary harm reduction strategies has demonstrated promising outcomes. Indigenous-led research initiatives have brought forth unique insights into the effectiveness of harm reduction programs tailored to the cultural and social contexts of Indigenous peoples. Furthermore, collaborative partnerships between Indigenous organizations and public health entities have begun to reshape policy discussions, ensuring that Indigenous voices are front and center in the formulation of harm reduction strategies.
However, there is still much work to be done. The future of this research area holds immense potential for transformative change. By continuing to explore the intricate relationships between Indigenous identities, cultural practices, and harm reduction, we can anticipate the development of innovative, community-driven programs. These programs could harness the power of Indigenous knowledge systems to inform public health approaches, fostering resilience and healing in the face of adversity. Advancements in technology may also allow for more nuanced data collection and analysis, enabling a deeper understanding of the lived experiences of Indigenous individuals and communities.
Topics of interest within this Special Collection on Indigenous communities and harm reduction include, but are not limited to:
● Traditional healing and harm reduction
● Culturally competent care
● Patterns of substance use
● Policy impacts on Indigenous health
● Collaborative research methodologies
● Historical trauma and recovery.
● Indigenous rights and harm reduction
● Community-led initiatives
● Community-based participatory research/methods
● Public health vending machines
● Stimulant use and associated harm reduction strategies
● Indigenous-led syringe services programs
● Indigenous-led models and “culture-as-healing” in harm reduction
● Sovereignty, criminalization, and decolonizing policy & practice
● Youth, kinship, and intergenerational harm reduction
● Methods/metrics: Indigenous data sovereignty, CBPR, and evaluation
● Access in rural/remote settings: SSPs, MOUD, telehealth, & mobile care.
This Collection supports and amplifies research related to SDG 3, Good Health and Well-Being and SDG 10, Reduced Inequalities.
All submissions in this collection undergo the journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief. As an open access publication, this journal levies an article processing fee (details here). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief.
Publishing Model: Open Access
Deadline: Sep 01, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in