Designing Better Hydrogen Catalysts—One Atom at a Time
Hydrogen is often hailed as the fuel of the future—but unlocking its full potential requires catalysts that are not only efficient but also affordable and scalable. Platinum has long been the gold standard for the hydrogen evolution reaction (HER), but its scarcity and cost pose major barriers to widespread adoption.
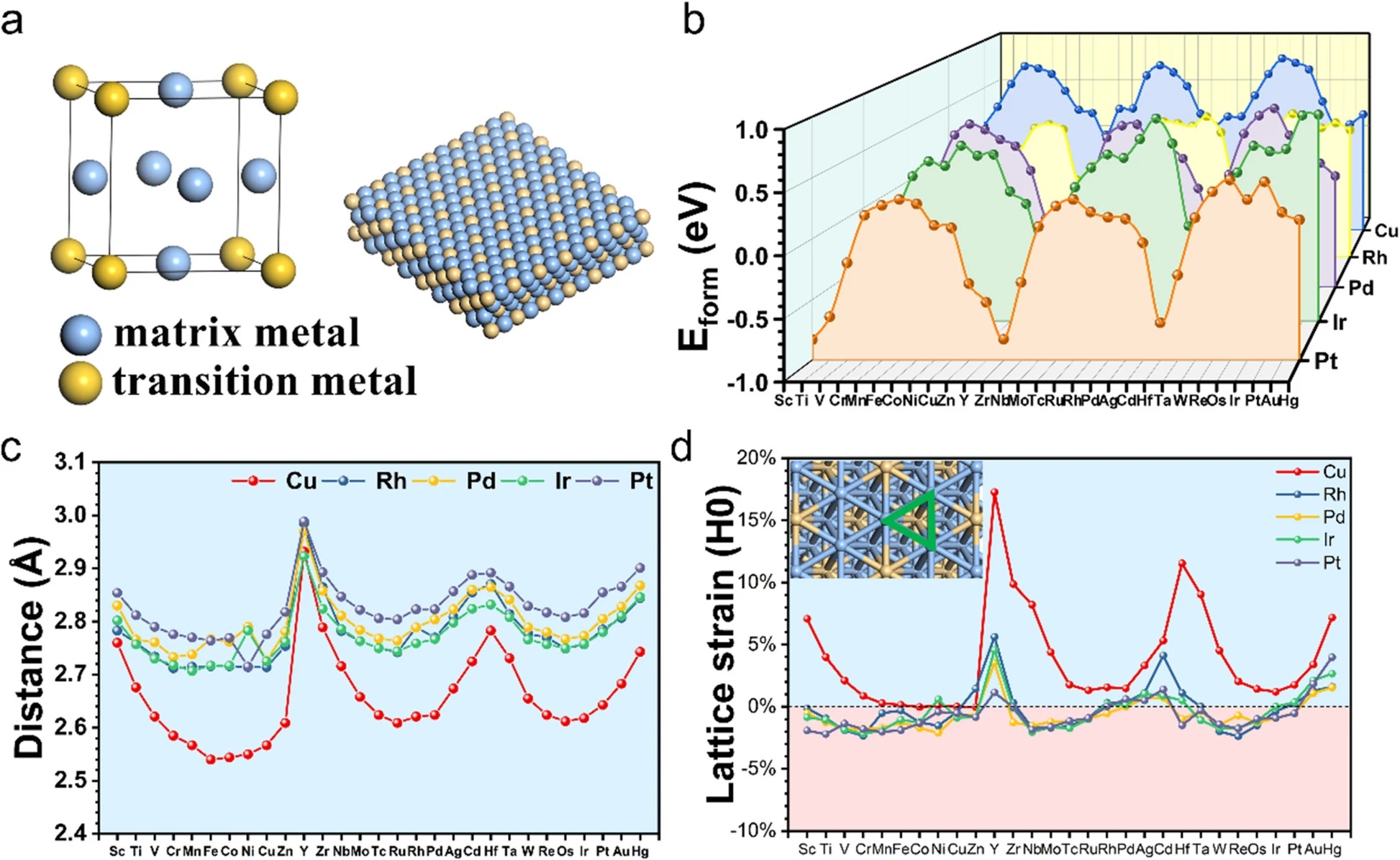
In our latest study, we set out to explore whether intermetallic compounds (IMCs)—specifically those doped with transition metals—could offer a viable alternative. Using density functional theory (DFT) calculations, we systematically screened 145 L1₂-type IMCs, combining noble metals like Rh, Pd, Ir, and Pt with a wide range of 3d, 4d, and 5d transition metals.
What we found was both surprising and exciting.
Key Insights
-
Noble metals still matter—but not in the way we thought.
Over 70% of the optimal active sites were dominated by noble metal atoms, while the doped transition metals primarily acted as electronic modulators, tuning the catalytic behavior without directly participating in the reaction. -
The volcano-shaped trend is predictive.
We observed a volcano-shaped relationship between hydrogen adsorption free energy and catalytic activity, confirming the Sabatier principle and offering a roadmap for identifying high-performance materials. -
14 IMC catalysts outperformed platinum.
Among the 145 candidates, 14 IMCs—including Pt₃H, Pd₃Zr, and Rh₃Cr—showed superior HER activity and thermal stability compared to conventional Pt catalysts. -
The d-band center theory holds up—even in complex systems.
Our results extend the applicability of d-band center theory to multi-metal environments, revealing how orbital hybridization and lattice strain influence HER performance at the atomic level.
This work is more than a computational exercise—it’s a step toward rational catalyst design, where atomic-scale insights guide the development of materials that are not only effective, but also economically viable.
We hope our findings will inspire further experimental validation and accelerate the transition to a hydrogen-powered future.
Follow the Topic
-
Catal
Catal is an open access journal covering full spectrum of catalysis critical advances. From biocatalysts to heterogeneous catalysts, it integrates fundamental and applied sciences. Catal offers a primary platform for researchers and practitioners in the field.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in