Flexible electronics armed to the teeth: Wearable theranostic dental patch for intraoral sensing and drug delivery
Published in Electrical & Electronic Engineering

Dental caries is the most prevalent non-communicable disease worldwide caused by the elevation of cariogenic microbiome in the oral cavity. Despite dental caries is acknowledged as a global public health challenge, it is still neglected in the personal oral health management. Surgical interventions after the cavity formation remain as the major clinical strategy for caries management, since the early changes of the tooth enamel are undetectable with the traditional clinical visual inspection and dental radiograph. Therefore, developing a point-of-care sensing system with high sensitivity integrated with therapy is highly desired for early prevention and timely treatment of dental caries.
Recently, a team led by Prof. Qingjun Liu at Biosensor National Special Laboratory, Zhejiang University have reported a fully integrated wearable and battery-free dental patch that can wirelessly monitor oral microenvironment in situ and deliver drug on demand (Fig. 1). The dental patch consisted of the control circuit and the functional electrode array is miniaturized and flexible, which can conformally interface on the tooth. An electrochemical potentiometric sensor is developed for the detection of the topical acidic environment fluctuation caused by oral microbial metabolism on the tooth surface, thus warn about the potential caries lesions. Through electrical stimulation, fluorides are timely delivered to give a feedback therapy.
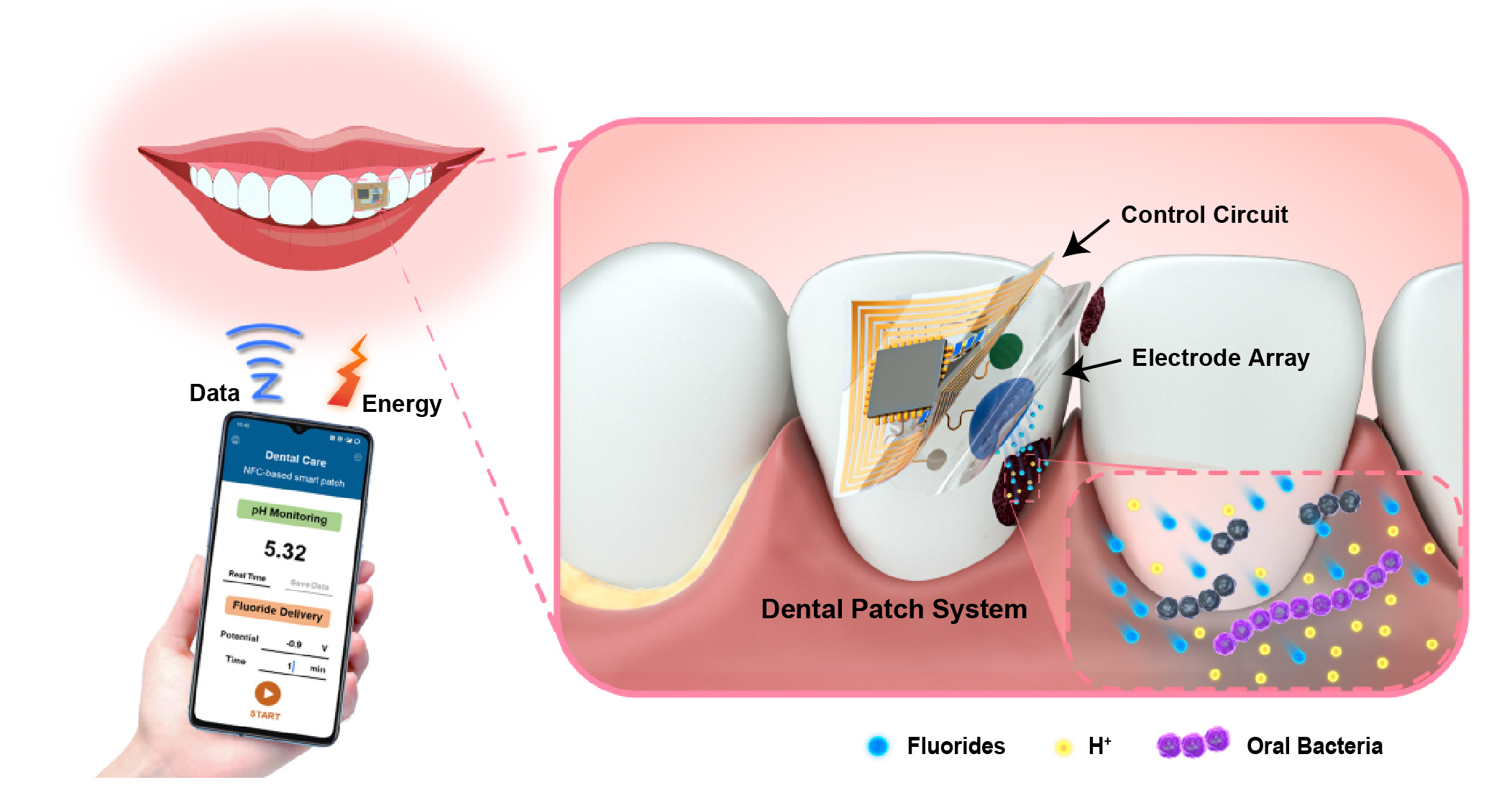
As displayed in Fig. 2, the dental patch is miniaturized, thin and lightweight. The near field communication (NFC) module is developed to achieve the wireless transmission of energy and data with mobile terminals, realizing the intraoral sensing and actively controlled drug delivery without on-board batteries. The reduction of circuit components makes the system more integrated, improving the comfort and safety of wearing greatly for the intraoral application.

Fig.2 Optical images of the smart dental patch. (a) The smart dental patch on a dandelion. (b) The flexible electrode array.
Cooperating with the research group at Stomatology Hospital in Zhejiang University, the team validates the performances of the dental patch system through in vitro cariogenic bacteria dental plaque growth experiments and on-body human trials. The oral cariogenic bacteria in the microenvironment metabolize carbohydrates from food debris and produce acids over time, which cause the demineralization of tooth, thereby resulting in caries lesion. The developed smart dental patch can evaluate the acidification condition of the oral microenvironment through the real-time detection of pH fluctuation on the tooth surface, realizing the monitoring and early warning of caries lesion in progress. When the acidic oral microenvironment is frequently observed, alert would be sent by the smartphone that an oral cleaning as well as the fluorides treatment is required (Fig. 3).
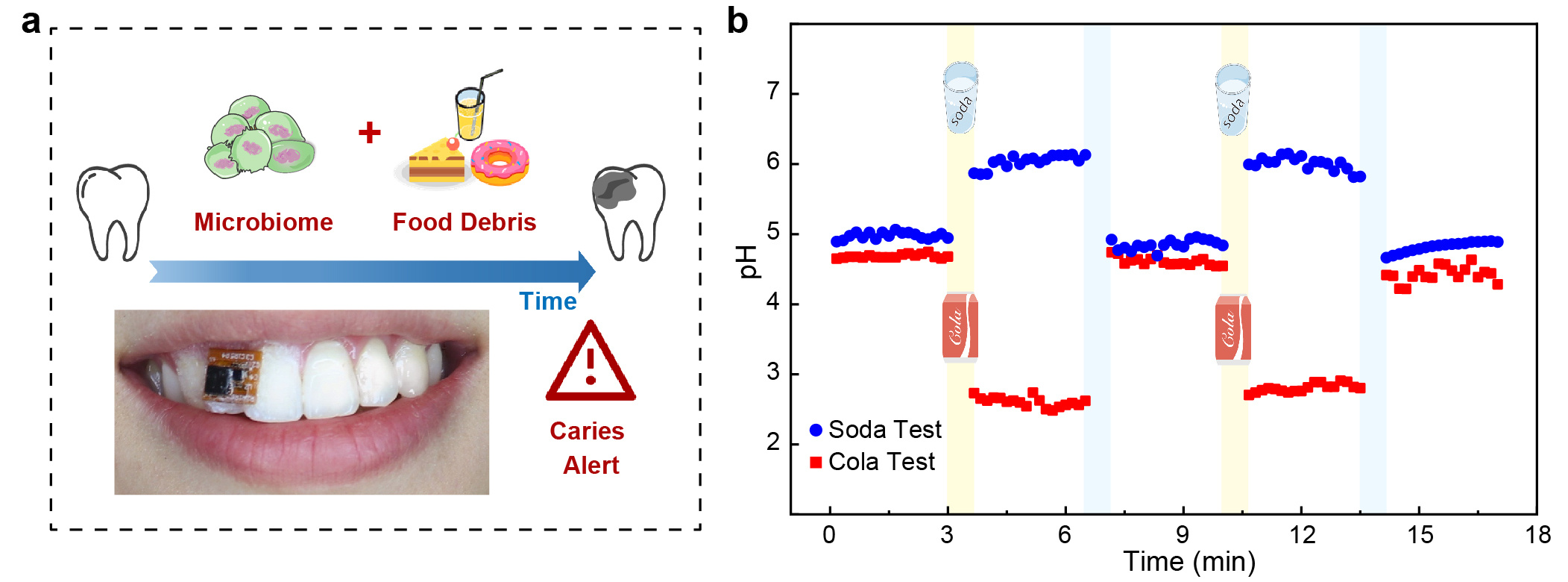
Fig. 3 In situ oral microenvironment monitoring with the dental patch. (a) Schematic of caries lesion detection by monitoring the topical oral microenvironment pH fluctuation. (b) Real-time monitoring of oral pH after volunteer drank pure water (pH=6.8), acidic cola (pH=2.5) and alkaline soda water (pH=8.0).
As the fluorides exposure is one of the most effective strategies for treating the early dental caries, an electrically controlled drug delivery module is integrated on the dental patch for the on-demand caries lesion treatment. Once the treatment command is given through the smartphone, the fluorides can be released locally on the tooth surface by the negative potential applied to the electrodes (Fig. 4a). The release rate under the electrical stimulation is obviously higher than the spontaneous release, which indicated the good regulation capability of the drug delivery module on the fluorides releasing (Fig. 4b). The fluorides can interfere with the metabolism of caries-related bacteria, thus inhibiting acid production and maintaining or even restoring the pH of the microenvironment (Fig. 4c). Acting as the catalyst, fluorides also assist with the redeposition of calcium and phosphate into tooth.
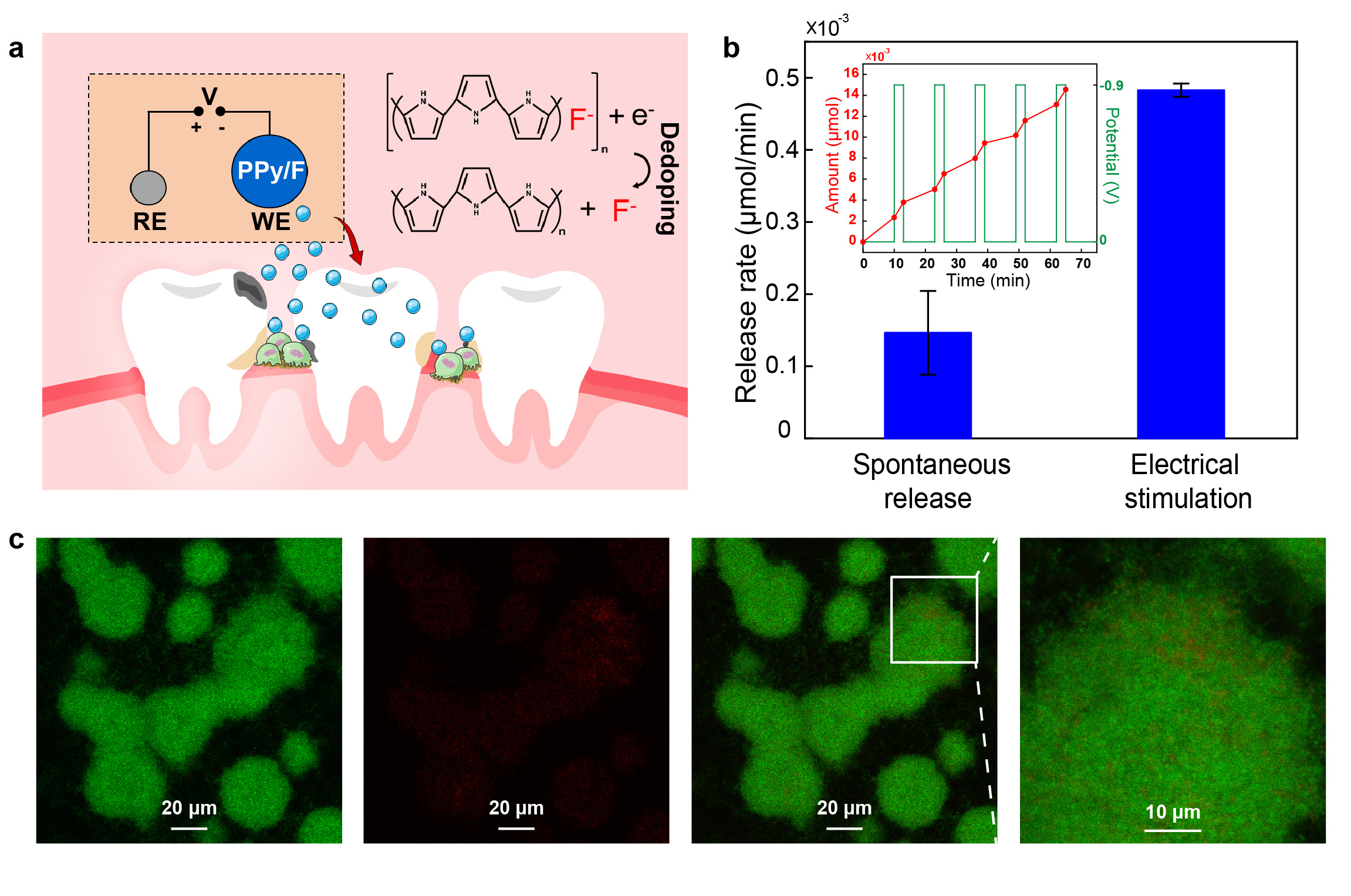
Fig. 4 Evaluation of the electrically controlled fluorides delivery. (a) Schematic of electrically controlled release of fluorides from the drug delivery module. (b) Release rate comparison between the spontaneous release and the electrical stimulation. (c) Confocal laser scanning microscopy (CLSM) images of S. mutans samples stained by the bacterial viability kit for evaluation of antibacterial activity of the drug delivery module.
With the increasingly concerns on oral health, dental caries management is shifting from reactively intervention to proactively prevention. Through real-time microenvironment monitoring and on-demand fluorides delivery, the dental patch can detect and treat the invisible lesion in time rather than wait until the formation of a cavity, which provides a novel strategy for clinical and family caries prevention. Apart from the acidity fluctuation caused by acidogenic bacteria associated with dental caries, there are abundant microbiome-related biomarkers in oral microenvironments like metabolites, antibodies and enzymes, which can provide insights into the health information from the molecular level. Given the capability to provide in situ detection and treatment topically, the smart dental patch is expected to enlarge applications on monitoring and regulating diverse microenvironments by interfacing on different parts in the oral cavity.
Owing to the design flexibility of the microcontroller, other typical electrochemical methods including cyclic voltammetry, differential pulse voltammetry and chronoamperometry can be further developed on the dental patch for real-time quantitative analysis. According to the therapeutic demands, specific drugs like antibiotics can be selectively loaded on the patch for the electrically controlled topical drug delivery. As a proof-of-concept, the proposed intraoral wearable dental patch system serves as an inspiring diagnostic and therapeutic mobile health platform to oral diseases, shedding lights on wearable medical electronics based on microbiome metabolism monitoring toward personalized medicine.
For more details, please read the original version of the manuscript:
Zhenghan Shi#, Yanli Lu#, Shuying Shen, Yi Xu, Chang Shu, Yue Wu, Jingjiang Lv, Xin Li, Zupeng Yan, Zijian An, Chaobo Dai, Lingkai Su, Fenni Zhang, Qingjun Liu*, Wearable battery-free theranostic dental patch for wireless intraoral sensing and drug delivery. npj Flexible Electronics 6, 49 (2022).
Link: https://www.nature.com/articles/s41528-022-00185-5
Follow the Topic
-
npj Flexible Electronics

This journal publishes high-quality papers related to flexible electronic systems, including plastic electronics and emerging materials, new device design and fabrication technologies, and applications.
Related Collections
With Collections, you can get published faster and increase your visibility.
Ferroelectrics based Flexible Electronics
Publishing Model: Open Access
Deadline: May 12, 2026
Neuromorphic Circuits and Bio-inspired Systems
Publishing Model: Open Access
Deadline: Mar 31, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in