From Resistance to Remission: Overcoming MEN1 Mutations by Switching Menin Inhibitors in Acute Myeloid Leukemia
Published in Cancer
Acute myeloid leukemia (AML) is a high-risk, biologically heterogeneous hematologic malignancy driven by the clonal proliferation of immature myeloblasts. The development of differentiation therapies that block leukemogenesis to restore normal hematopoiesis has introduced a treatment paradigm that differs from traditional cytotoxic chemotherapy.1 One prominent example is that of menin inhibition, which disrupts the interaction between the scaffolding protein menin and MLL1 (KMT2A). For acute leukemias where the menin-MLL1 interaction is essential, such as those driven by NPM1 mutations (NPM1m) or KMT2A rearrangements (KMT2Ar), menin inhibitors have shown tremendous promise. Indeed, the menin inhibitors revumenib and ziftomenib have both been approved for relapsed or refractory (R/R) NPM1m AML and revumenib is additionally approved for R/R KMT2Ar acute leukemias.2–4
Despite these advances, one clear weakness of first-generation agents of this class appears to be the relatively limited duration of response (DOR). Across available datasets, the median duration of CR/CRh for revumenib, ziftomenib, and other menin inhibitors with available preliminary data appear to be relatively similar and limited, generally on the order of 6 months or less.5 When resistance emerges, outcomes are poor and subsequent therapies rarely yield durable responses.6
One important mechanism driving the limited DOR is the rapid development of de novo MEN1 resistance mutations. MEN1 encodes the menin protein, which contains an N-terminal, thumb, palm, and C-terminal domains. The menin-MLL1 binding interface is formed by the thumb-palm pocket and as detailed by Perner et al., treatment with revumenib can rapidly select for MEN1 mutations (e.g. M327I, G331R, T349M, M327V) that disrupt revumenib binding while preserving the menin-MLL1 interaction.7 These mutations, which were noted in nearly 40% of patients treated with revumenib on AUGMENT-101 using highly sensitive droplet digital PCR (ddPCR), can arise as early as after 2 cycles of therapy, leading to clinical resistance. MEN1 mutations have also been reported with ziftomenib, although due to inter-trial variability in testing methods and resistance assessment make it difficult to fully define the contribution of MEN1 alteraions to resistance across agents, and this remains an active area of investigation.
As multiple menin inhibitors are developed in parallel and succession, preclinical studies have begun to delineate differential binding properties across different MEN1 resistance mutations and menin inhibitors. As elegantly shown by Kwon et al. and more recently Miao et al., bleximenib (JNJ-75276617) and ziftomenib can retain binding to menin in the presence of certain MEN1 mutations.8,9 Specifically, bleximenib retained effective binding in the setting of MENIM327I but not MEN1T349M, whereas ziftomenib retained effective binding with MEN1T349M and MEN1G331R but not MEN1327I or MEN1G331D.
In this Letter, we demonstrate the clinical feasibility of applying these preclinical insights by treating a patient with adverse-risk KMT2Ar AML (t(6;11), complex karyotype) who achieved an initial response to revumenib monotherapy but developed resistance after 17 cycles of therapy. At relapse, she was found to harbor a MEN1M327I mutation and was subsequently transitioned to bleximenib, achieving MRD-negative complete remission that was consolidated with allogeneic hematopoietic stem cell transplantation. She remains in remission more than 15 months after bleximenib initiation.
We further demonstrated rapid and complete eradication of the MEN1M327I clone by ddPCR and did not detect additional emergent MEN1 or non-MEN1 mutations by next generation sequencing (Figure 1). Of note, the patient’s initial response to revumenib also illustrated an important feature of differentiation therapy, in which clearance of leukemic blasts by immunophenotyping preceded full resolution of cytogenetic abnormalities. This pattern was consistent with the differentiation of leukemic cells harboring t(6;11) KMT2A::MLLT4 into phenotypically normal myeloid cells (Figure 2).
In this case report, we provide proof of concept of menin inhibitor switching guided by MEN1 mutational profiling as a strategy to overcome a well-characterized mechanism of resistance. While this approach has not been previously described in AML, it is well established in other hematologic malignancies, such as chronic myeloid leukemia, where testing for BCR::ABL1 kinase domain mutations is standard of care at defined points of treatment failure.
As present, the generalizability of this approach remains uncertain. Additional cases and systematic study will be required to better define its utility, but this is currently limited by lack of guideline-supported MEN1 mutational testing and variability in assay availability across institutions, though commercial panels are starting to address this need. Moreover, a subset of patients develop resistance through MEN1-independent mechanisms, in which sequential menin inhibitor switching may have unclear utility. Strategies to prevent or overcome resistance, including sequential menin inhibitor therapy and combination approaches with chemotherapy, venetoclax-based regimens, or other investigational agents, will be important areas of future study.10–12 We believe that systematic and consistent resistance profiling in menin inhibitor development, early identification of emerging mutations, and development of best practices for long-term menin inhibitor use will be critical to optimizing outcomes in patient population.
- Issa, G. C., Stein, E. M. & DiNardo, C. D. How I treat acute myeloid leukemia with differentiation therapy. Blood 145, 1251–1259 (2025).
- Issa, G. C. et al. Menin Inhibition With Revumenib for KMT2A-Rearranged Relapsed or Refractory Acute Leukemia (AUGMENT-101). JCO JCO.24.00826 (2024) doi:10.1200/JCO.24.00826.
- Arellano, M. L. et al. Menin inhibition with revumenib for NPM1-mutated (NPM1m) relapsed or refractory acute myeloid leukemia: AUGMENT-101. Blood https://doi.org/10.1182/blood.2025028357 (2025) doi:10.1182/blood.2025028357.
- Wang, E. S. et al. Ziftomenib in Relapsed or Refractory NPM1-Mutated AML. J Clin Oncol 43, 3381–3390 (2025).
- Boussi, L., Cai, S. F. & Stein, E. M. Advances in menin inhibition in acute myeloid leukemia. Trends in Cancer 11, 889–900 (2025).
- Chin, K.-K. et al. Outcomes of relapsed or refractory acute myeloid leukemia after menin inhibition failure. Blood Adv bloodadvances.2025018178 (2025) doi:10.1182/bloodadvances.2025018178.
- Perner, F. et al. MEN1 mutations mediate clinical resistance to menin inhibition. Nature 1–7 (2023) doi:10.1038/s41586-023-05755-9.
- Kwon, M. C. et al. Preclinical Efficacy of the Menin-KMT2A Inhibitor JNJ-75276617 in Combination with Venetoclax and Azacitidine in AML. Blood 142, 4167 (2023).
- Miao, H. et al. Discovery and preclinical activity of the menin-KMT2A inhibitor ziftomenib in acute leukemia models. Blood blood.2025031201 (2026) doi:10.1182/blood.2025031201.
- Zeidner, J. F. et al. Azacitidine, Venetoclax, and Revumenib for Newly Diagnosed NPM1-Mutated or KMT2A-Rearranged AML. JCO 0, JCO-25-00914 (2025).
- Bourgeois, W. et al. Mezigdomide is effective alone and in combination with Menin inhibition in pre-clinical models of KMT2A-r and NPM1c AML. Blood blood.2023021105 (2023) doi:10.1182/blood.2023021105.
- Thakur, R. K. & Wang, E. S. The promise of menin inhibitors: from approval to triplet regimens. Hematology Am Soc Hematol Educ Program 2025, 599–606 (2025).
Follow the Topic
-
Leukemia
This journal publishes high quality, peer reviewed research that covers all aspects of the research and treatment of leukemia and allied diseases. Topics of interest include oncogenes, growth factors, stem cells, leukemia genomics, cell cycle, signal transduction and molecular targets for therapy.
Your space to connect: The Myeloid cell function and dysfunction Hub
A new Communities’ space to connect, collaborate, and explore research on Clinical Medicine and Cell Biology!
Continue reading announcement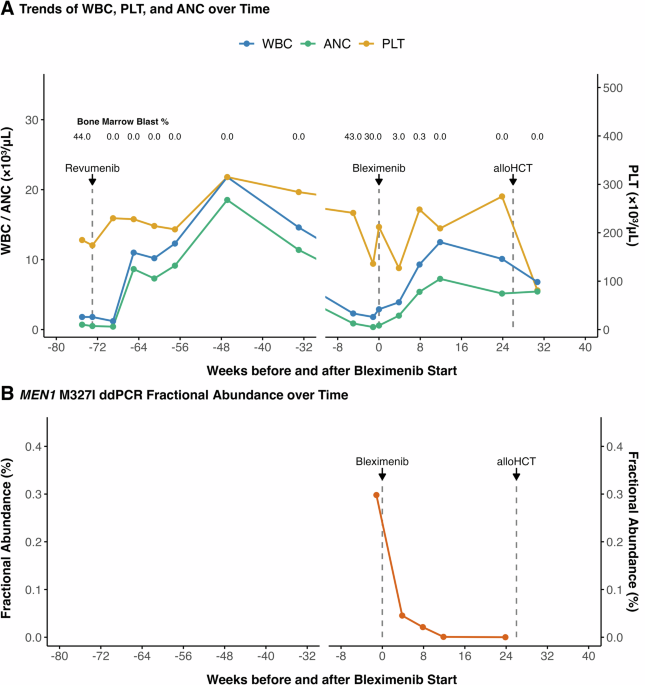






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in