gamma-delta T cells in cancer: manning the front lines
Published in Cancer
Do gd T cells matter in cancer?
After decades of controversy, immune evasion has finally been recognised as a hallmark of cancer1 and T cells, in particular, have been implicated2. T cells come in two broad flavours, alpha-beta (ab) and gamma-delta (gd) T cells. ab T cells are by far more numerous and are capable of specific recognition of cancer neoantigens. gd T cells on the other hand comprise less than 10% of peripheral blood T cells and do not commonly recognise cancer neoantigens3. Nonetheless, these cells are conserved throughout vertebrate evolution, hinting at important and non-redundant functions.
Immunotherapies targeting ab T cells have revolutionised cancer therapy, but most patients do not benefit, and current strategies are approaching an asymptote. Thus, there is now an increasing focus on utilising other immune cells. gd T cells would seem like ideal candidates. Despite their numerical disadvantage compared to ab T cells, gd T cells are enriched at body surfaces in which cancers develop and can recognise cancer cells independent of neoantigens or MHC. Therefore, they are both well placed to detect malignant transformation and offer alternative surveillance mechanisms compared to ab T cells.
Our current understanding of gd T cells and key outstanding questions.
Girardi and colleagues were the first to demonstrate a role for these cells in cancer immunosurveillance. They found that mice genetically deficient in gd T cells were more susceptible to cutaneous carcinogenesis and that their contribution to immunosurveillance was both independent of and synergistic with ab T cells4,5. Subsequently, others have also independently demonstrated that gd T cells restrain cancers in mice6,7 and are associated with a good prognosis in clinical disease8–12. Set against this, there is compelling evidence in murine models that some gd T cells, particularly those that produce interleukin-17 (IL-17), can promote cancers13,14. Moreover, clinical trials of gd T cell therapy have so far shown very little efficacy15.
Several aspects of gd T cell biology may underpin these seemingly conflicting observations. It is clear in mice that IFNg-producing gd T cells are cancer-restraining whilst IL-17-producing gd T cells are cancer-promoting. Nonetheless, IL-17-producing gd T cells have been difficult to find in humans, and in the rare cases that have been described, only after extensive ex vivo manipulation10,11,16–18. Furthermore, gd T cells, like ab T cells, are composed of distinct subsets occupying different functional niches. Despite this, many clinical studies have hitherto considered gd T cells as one entity, in part owing to the difficulty of detecting these rare cells in clinical samples. Likewise, almost all cancer immunotherapy trials to date have utilised the Vd2 subset of gd T cells as these cells are more easily obtained. Against this backdrop, we set out to answer several key questions regarding human gd T cells in cancer. Firstly, which subsets of gd T cells are found within human epithelial tissues and the carcinomas that arise from them? Secondly, what are the unmanipulated, in situ phenotypes and functional potentials of these cells? In particular, do they show any evidence of the tumour-promoting T-helper-17 (Th17) skew observed in murine models? Finally, does the presence of gd T cells, or subsets thereof, predict clinical outcome?
Results from the CRUK TRACERx Study.
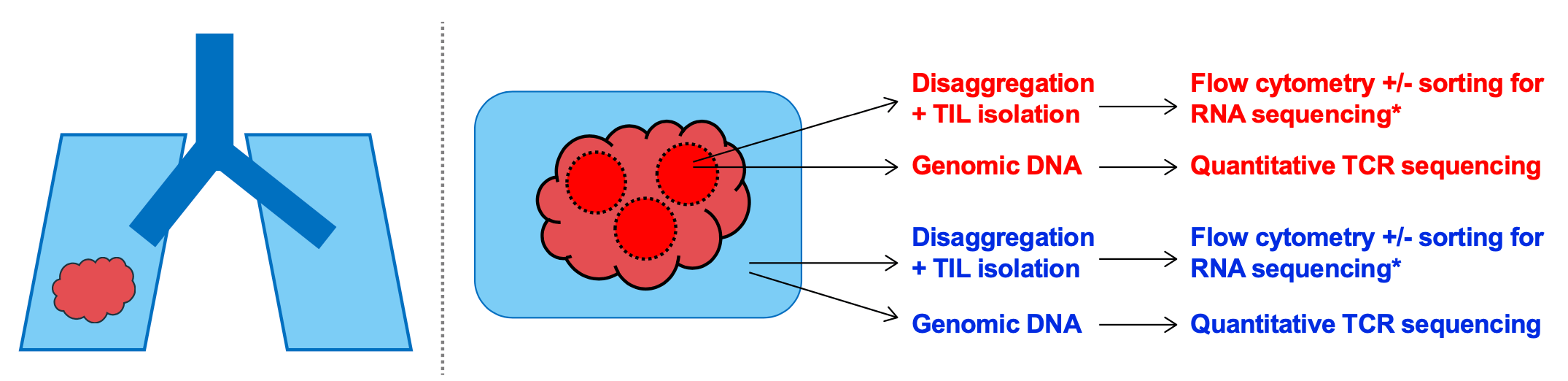
To address these questions, we have leveraged samples and clinical data collected in the TRACERx [TRacking non-small-cell lung Cancer (NSCLC) Evolution through therapy (Rx)] Study19. To overcome the well-recognised difficulty in characterising gd T cells in clinical tissue samples20, we combined flow cytometry and TCR-sequencing (TCRseq) to robustly identify and quantify gd T cell subsets within non-tumour lung tissues, paired NSCLCs, and peripheral blood. This was made possible by the comprehensive TRACERx study protocol which banked tissue/tumour infiltrating lymphocytes (TILs) for flow cytometry as well as region-matched genomic DNA for paired TCRseq.
We found that both non-tumour lung tissues and NSCLCs harboured gd T cells. Of these, Vd1 T cells were the predominant subset in contrast to peripheral blood gd T cells where Vd2 cells predominate. Moreover, these cells appeared to be bona fide tissue-resident T (TRM) cells as they were mostly CD103+ and possessed a core TRM transcriptional profile. Thus, our findings, together with other studies10,11,21, strongly suggest that Vd1 T cells are the signature tissue-resident gd T at human body surfaces. By employing RNAsequencing and in vitro activation assays of directly isolated TILs, we demonstrated that intratumoural Vd1 T cells possessed a cancer-restraining phenotype in situ. In contrast to murine models, we could find no evidence of a cancer-promoting Th17-skew in these cells. Functionally, Vd1 T cells resembled natural killer (NK) and CD8+ T cells, two prototypic cytotoxic immune cells consistently associated with improved outcomes in cancer9. Moreover, intratumoural Vd1 T cells possessed transcriptional features suggestive of a capacity for self-renewal, akin to “stem-like” CD8+ T cells that are critical to effective cancer immunosurveillance22. In short, Vd1 T cells were resident within tumours and, in many key respects, resembled prototypic cancer-killing NK cells and CD8+ T cells. Nonetheless, as the idiom goes, the proof is in the pudding. Thus, we examined the association of intratumoural Vd1 T cells with clinical outcome. Indeed, the presence of Vd1 T cells predicted favourable survival in our cohort whereas ab T cells, found in several magnitude great numbers, did not predict survival.
We extended these findings by demonstrating a significant association of a Vd1 gene signature with survival in over 800 patients with NSCLC in The Cancer Genome Atlas. Conversely, we found no association between intratumoural Vd2 T cells and survival. Strikingly, we also observed a strong and significant association of Vd1 T cells in non-tumour tissue with improved survival. This suggests that resident Vd1 T cells within juxtapositional normal tissues left in situ at ground-zero after surgical resection may provide ongoing cancer immunosurveillance.
The proof is in the pudding.
Less is known about human Vd1 T cells compared to peripheral blood Vd2 T cells and there is a temptation to draw parallels with tissue-associated gd T cells in mice. Our study provides direct insights into the biology of human Vd1 T cells within tissues and tumours and adds to the growing appreciation that these cells restrain cancer. Nonetheless, it remains to be seen if cancer immunotherapies utilising Vd1 T cells will improve upon the modest bar set by Vd2 T cells. A first-in-human trial of Vd1 T cells in acute myeloid leukaemia (NCT05001451) is currently underway and may provide the proof in the pudding.
References
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov 12, 31–46 (2022).
- Waldman, A. D., Fritz, J. M. & Lenardo, M. J. A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat Rev Immunol 20, 651–668 (2020).
- Hayday, A. C. γδ T Cell Update: Adaptate Orchestrators of Immune Surveillance. J Immunol 203, 311–320 (2019).
- Girardi, M. et al. Regulation of Cutaneous Malignancy by γδ T Cells. Science 294, 605–609 (2001).
- Girardi, M. et al. The Distinct Contributions of Murine T Cell Receptor (TCR)γδ+ and TCRαβ+ T Cells to Different Stages of Chemically Induced Skin Cancer. J Exp Medicine 198, 747–755 (2003).
- Gao, Y. et al. γδ T Cells Provide an Early Source of Interferon γ in Tumor Immunity. J Exp Medicine 198, 433–442 (2003).
- Dalessandri, T., Crawford, G., Hayes, M., Seoane, R. C. & Strid, J. IL-13 from intraepithelial lymphocytes regulates tissue homeostasis and protects against carcinogenesis in the skin. Nat Commun 7, 12080 (2016).
- Bialasiewicz, A. A., Ma, J. X. & Richard, G. Alpha/beta- and gamma/delta TCR(+) lymphocyte infiltration in necrotising choroidal melanomas. The British journal of ophthalmology 83, 1069–1073 (1999).
- Gentles, A. J. et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nature medicine 21, 938–945 (2015).
- Wu, Y. et al. An innate-like Vδ1+ γδ T cell compartment in the human breast is associated with remission in triple-negative breast cancer. Sci Transl Med 11, (2019).
- Foord, E., Arruda, L. C. M., Gaballa, A., Klynning, C. & Uhlin, M. Characterization of ascites- and tumor-infiltrating γδ T cells reveals distinct repertoires and a beneficial role in ovarian cancer. Sci Transl Med 13, eabb0192 (2021).
- Craven, K. E., Gökmen-Polar, Y. & Badve, S. S. CIBERSORT analysis of TCGA and METABRIC identifies subgroups with better outcomes in triple negative breast cancer. Sci Rep-uk 11, 4691 (2021).
- Coffelt, S. B. et al. IL-17-producing γδ T cells and neutrophils conspire to promote breast cancer metastasis. Nature 522, 345–348 (2015).
- Jin, C. et al. Commensal Microbiota Promote Lung Cancer Development via γδ T Cells. Cell 176, 998–1013 (2019).
- Kabelitz, D., Serrano, R., Kouakanou, L., Peters, C. & Kalyan, S. Cancer immunotherapy with γδ T cells: many paths ahead of us. Cell Mol Immunol 17, 925–939 (2020).
- Almeida, A. R. et al. Delta One T cells for immunotherapy of chronic lymphocytic leukemia: clinical-grade expansion/ differentiation and preclinical proof-of-concept. Am Assoc Cancer Res 22, clincanres.0597.2016 (2016).
- Tan, L. et al. A fetal wave of human type 3 effector γδ cells with restricted TCR diversity persists into adulthood. Sci Immunol 6, eabf0125 (2021).
- Caccamo, N. et al. Differentiation, phenotype, and function of interleukin-17–producing human Vγ9Vδ2 T cells. Blood 118, 129–138 (2011).
- Jamal-Hanjani, M. et al. Tracking the Evolution of Non–Small-Cell Lung Cancer. New Engl J Medicine 376, 2109–2121 (2017).
- Jungbluth, A. A. et al. Immunohistochemical Detection of γ/δ T Lymphocytes in Formalin-fixed Paraffin-embedded Tissues. Applied immunohistochemistry & molecular morphology : AIMM 00, 1–3 (2018).
- Mikulak, J. et al. NKp46-expressing human gut-resident intraepithelial Vδ1 T cell subpopulation exhibits high antitumor activity against colorectal cancer. JCI Insight 4, 8900–20 (2019).
- Eberhardt, C. S. et al. Functional HPV-specific PD-1+ stem-like CD8 T cells in head and neck cancer. Nature 597, 279–284 (2021).
Follow the Topic
-
Nature Cancer
This journal aims to provide a unique forum through which the cancer community will learn about the latest, most significant cancer-related advances across the life, physical, applied and social sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Cancer Neuroscience: from mechanisms to therapy
Publishing Model: Hybrid
Deadline: Jan 30, 2027



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in