Hammering ammonia into alkenes to build aromatic N-heterocycles via electricity
Published in Chemistry
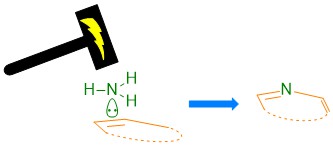
The reaction design. Aromatic N-heterocycles have wide applications in pharmaceutical products and materials. Typically, the synthesis of N-heterocycles is achieved via the prefunctionalization of substrates, for example, with oxidation to carbonyl groups, condensation, and sometimes postoxidation (Figure 1a). To enhance the step economy and atom economy, a single step protocol using the simplest nitrogen source and substrate without preoxidation would be a solution. It has been proposed that ammonia is the ultimate nitrogen source.1 In our previous study conducted by Ph. D student Mr Li in 2019, ammonia worked as an efficient, safe, and readily available hydrogen source in the metal-free electrochemical hydrogenation reactions.2 This prompted us to find a method using ammonia as a nitrogen source in electrochemical reactions. This concept was proved by the Noel group3 and our group4 later on in the aziridination reaction. We envisioned that if ammonia could be inserted into cyclic alkenes, a reaction could provide almost quantitative atom economy. This plan was outlined in a single-step reaction (Fig. 1b), in which the only side product would be hydrogen.
Exploration of the reaction working with ammonia and alkene. Ph. D student Mr. Liu started to find the first molecule that could realize this assumption. After several months, we find that indene A is a good candidate. In an undivided cell with a graphite felt anode and Ag cathode, just plug in a balloon charged with ammonia, indene was converted to isoquinoline B via electrolysis in hours with methanol as the solvent (Fig. 1c). Graphite felt is essential for the anode. If Pt was used, we observed the decomposition of MeOH at the anode, similar to that in the fuel battery. If RVC was used, we observed the degradation of the anode itself.
Then, Liu expanded the substrate scope to a variety of substituted indenes. In addition, penta-substituted pyridine is also synthesized from cyclopentadiene. The reaction could be conducted in commercial MeOH without further purification. In these transformations, the theoretical atom economy is between 98% and 99.2%. Since ammonia only has a very narrow flammable range, the reaction is safe enough to conduct in common hoods.
After this exploration, Mr Liu further showed that electricity is a strong driving force to edit N-heterocycles. This plan is shown in Figure 3. The initial N-heterocycle was a substituted Hantzsch ester C. This compound was first subjected to electrochemical dehydrogenation, forming a pyridine intermediate D. Without isolation, by only changing the anode from graphite felt to zinc and the addition of water and BF3, the electrolysis continued to undergo a cascade reaction (dearomatization, rearrangement, rearomatization) to offer pyrrole intermediate E. At this stage, ethyl acetate was cut off the ring. Additionally, without isolation, the mixture containing intermediate E was subjected to the insertion of ammonia under electrolysis. Then, the final product pyrimidine F was isolated in 12% yield in a tandem and cascade electrochemical transformation sequence from dihydropyridine, pydridine, pyrrole, to pyrimidine (Fig. 1d).
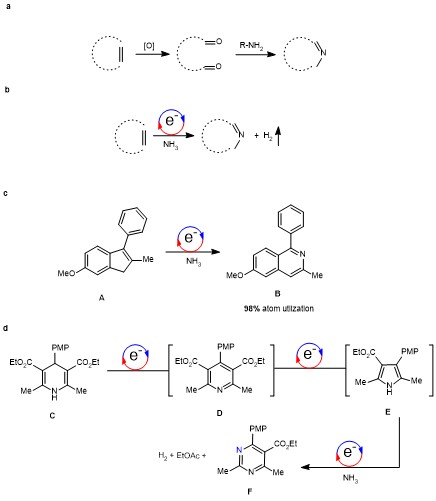
Fig. 1 The insertion of ammonia into alkene to build aromatic N-heterocycles. a. Established stepwise protocols to build aromatic N-heterocycles. b. Single-step insertion of ammonia in this report. c. The first working example. d. Editing the N-heterocycles by electrolysis.
Next, we started to obtain some information on the mechanism. We carried out CV analysis and found that in MeOH, indene could be oxidized prior to ammonia. In a scaled-up reaction, we could isolate an aziridine intermediate. Both lines of evidence support an alkene activation mechanism. We found aziridine was unstable even at -18 °C for a couple of days. However, this tendency only led to a complex mixture at room temperature or -18 °C. Only the electrochemical conditions could convert aziridine to the corresponding product. Therefore, we proposed a four–electron transfer pathway, including the activation of alkenes to cationic radicals, nucleophilic addition of ammonia to aziridine, activation of aziridine to N-cationic radicals, rearrangement, and oxidative dehydrogenation.
Inspirations during the review process. The reviewers gave us plenty of pertinent suggestions. For example, we are aware of the limitations of this protocol. The substituents on alkenes play a pivotal and complicated role in the reaction, which limits the structure of the substrate. The kinetic profile of the intermediate was suggested by the reviewer to support the stepwise insertion of ammonia into alkene. Aqueous ammonia was also suggested as an alternative by the reviewer and was confirmed by the experiment. This change in the N source would facilitate the experiment when access to gaseous ammonia is limited.
The initial work was carried out by Mr Liu when he was still a Ph.D student. Then, he defensed his Ph. D thesis and got the doctor degree. When the revision was conducted, Dr. Liu carried out partial revision works at his new position (Fig. 2). We took this situation as an opportunity to check if the result could be repeated at different laboratories. To our delight, we can obtain even improved yields for several examples.

Fig. 2 Dr. Shuai Liu at his new position.
The link to the publication:
Journal: Nature Communications.
DOI : 10.1038/s41467-022-28099-w
Title : Insertion of ammonia into alkenes to build aromatic N-heterocycles.
Reference:
- Kim H, Chang S. The Use of Ammonia as an Ultimate Amino Source in the Transition Metal-Catalyzed C–H Amination. Acc. Chem. Res. 50, 482-486 (2017).
- Li J, He L, Liu X, Cheng X, Li G. Electrochemical Hydrogenation with Gaseous Ammonia. Angew. Chem. Int. Ed. 58, 1759-1763 (2019).
- Ošeka M, et al. Electrochemical Aziridination of Internal Alkenes with Primary Amines. Chem 7, 255-266 (2021).
- Liu S, et al. Electrochemical Aziridination of Tetrasubstituted Alkenes with Ammonia. CCS Chem. (2021), DOI: doi:10.31635/ccschem.021.202100826.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in