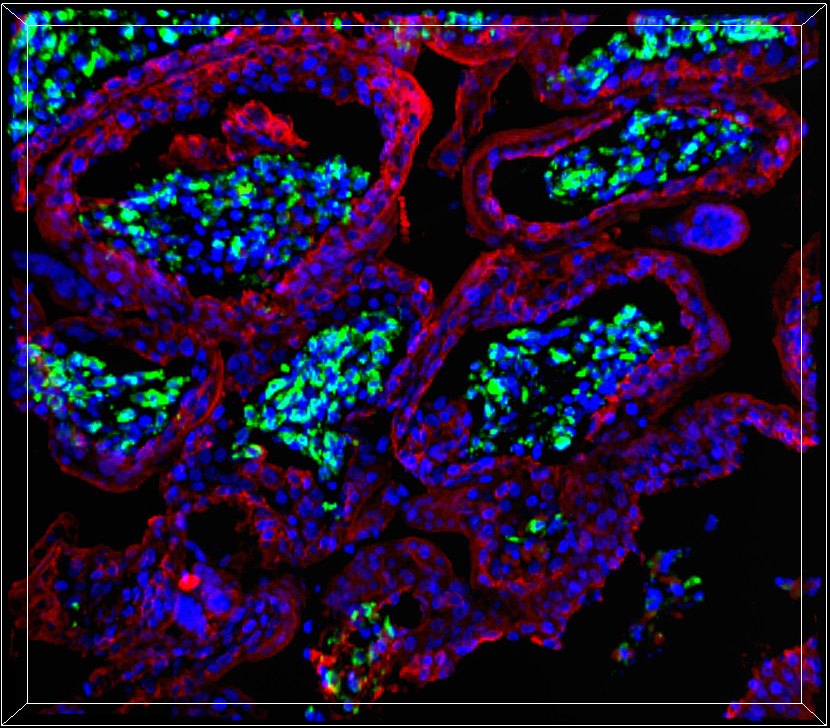
Hepatitis E virus (HEV) is a small RNA virus that usually causes asymptomatic infection or acute self-limiting illness. However, in emerging countries with poor sanitation, infection with HEV genotype 1 (HEV-1) during pregnancy often results in fulminant hepatic failure (15 to 30% of cases) associated with severe placental diseases raging from eclampsia, hemorrhage to spontaneous abortion or stillbirth. On the other hand, in Western countries, where genotype 3 (HEV-3) prevails, HEV infection of pregnant women is rather harmless. Even if significant effort has been made towards understanding the severity of HEV-1 during pregnancy, the causal relationship between infection and placental dysfunction remain elusive. Our understanding of this genotype-specific pathogenesis was hampered by the lack of appropriate experimental models and the difficulty to propagate the virus in vitro.
Viruses that infect the mother can be amplified at the maternal fetal-interface before spreading to the fetal compartment. Our working hypothesis is that both the viral tropism and the consequences of the infection are responsible for the severity of HEV during pregnancy. To test this hypothesis and provide insights into the genotype-specific pathogenicity of HEV during pregnancy, we developed a new ex vivo model of HEV infection at the maternal-fetal interface using organ cultures from the first-trimester maternal decidua and fetal placenta. Using this physiologically relevant model and clinical viral strains, we deciphered the pathogenicity mechanisms of HEV-1 compared to HEV-3 (Figure 1).
We discovered that HEV-1 replicates more efficiently than HEV-3 in decidua and placenta explants as well as in primary stromal cells derived from these tissues, produces higher amounts of infectious progeny virions and causes severe morphological alterations. Furthermore, the excessive viral replication correlates with major alterations in the cytokine, chemokine and growth factor networks at the maternal-fetal interface, resulting in exacerbated tissue injuries. Finally, our study also revealed that HEV-1 partially inhibit interferon lambda (IFN-l) in both decidual and placental tissues. The fact that addition of exogenous IFN-l was able to restrict HEV-1 replication further strengthens the potential role of this pathway in the genotype-specific exacerbation of HEV pathogenesis.
In conclusion, our study provides an innovative model to study HEV pathogenesis, based on ex vivo organ cultures and primary stromal cells derived from the human maternal-fetal interface. With this model, we provided mechanistic insights into the severity of HEV-1 during pregnancy. Therefore, our data advocate that the viral tropism and efficient replication combined with abnormal pro-inflammatory microenvironment dictate the extent of tissue damage at the maternal-fetal interface and might be responsible for HEV-1-associated pregnancy disorders. Furthermore, our clinically relevant model paves the way for the testing and the development of novel therapeutic strategies directed towards supporting human pregnancy.
Full paper can be found @ https://www.nature.com/articles/s41467-018-07200-2
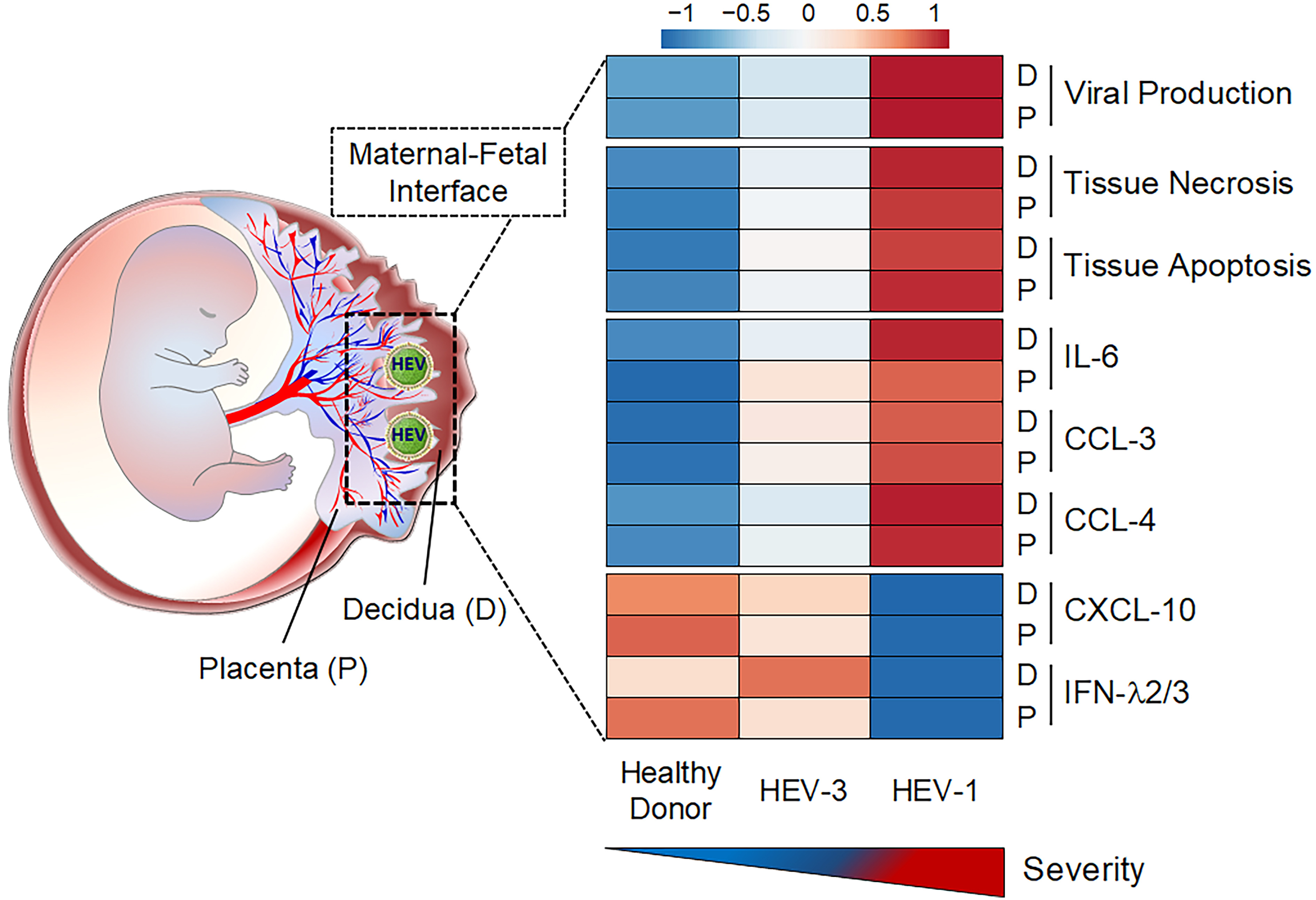
Figure 1. Genotype-specific pathogenicity of HEV at the human maternal-fetal interface. Graphical abstract summarizing the differences observed between HEV-1 and HEV-3 infection in the maternal decidua (D) and fetal placenta (P).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in