How gut bacteria team up to heal the gut – a story of two papers
Published in Biomedical Research
Two years ago, we set out to answer a seemingly simple question: Can the bacteria living in our gut directly talk to intestinal stem cells and help repair a damaged gut lining?
The answer, as often happens in science, turned into a much longer but a more exciting journey than we ever expected. It eventually led to two back‑to‑back papers in Nature Communications, a split manuscript, a lost reviewer, and a renewed appreciation for microbial teamwork.
The problem: when the gut barrier fails
Inflammatory bowel disease (IBD) affects millions of people worldwide. Current treatments mainly focus on suppressing inflammation – but they don’t fix the underlying problem: a leaky, damaged gut lining. We realised that simply calming the immune system is not enough. We needed to understand how to rebuild the barrier.
Our previous work had shown that certain bacterial metabolites, especially indole‑3‑propionic acid (IPA), can protect against colitis. But we didn’t know how IPA works at the stem cell level, nor which bacteria produce it and how they cooperate.
The breakthrough: two papers, one story
After years of experiments from germ‑free mice to intestinal organoids, from RNA‑sequencing to lineage tracing..., we uncovered a multi‑step, cross‑kingdom conversation:
-
Paper 1 (IPA‑PPARα‑HMGCS2‑BHB axis): We discovered that IPA directly binds to a receptor (PPARα) on intestinal epithelial cells, turning on a ketone body (BHB) production line. This BHB then wakes up LGR5⁺ active stem cells, driving rapid repair. The effect is so strong that giving BHB alone mimics IPA’s healing power.
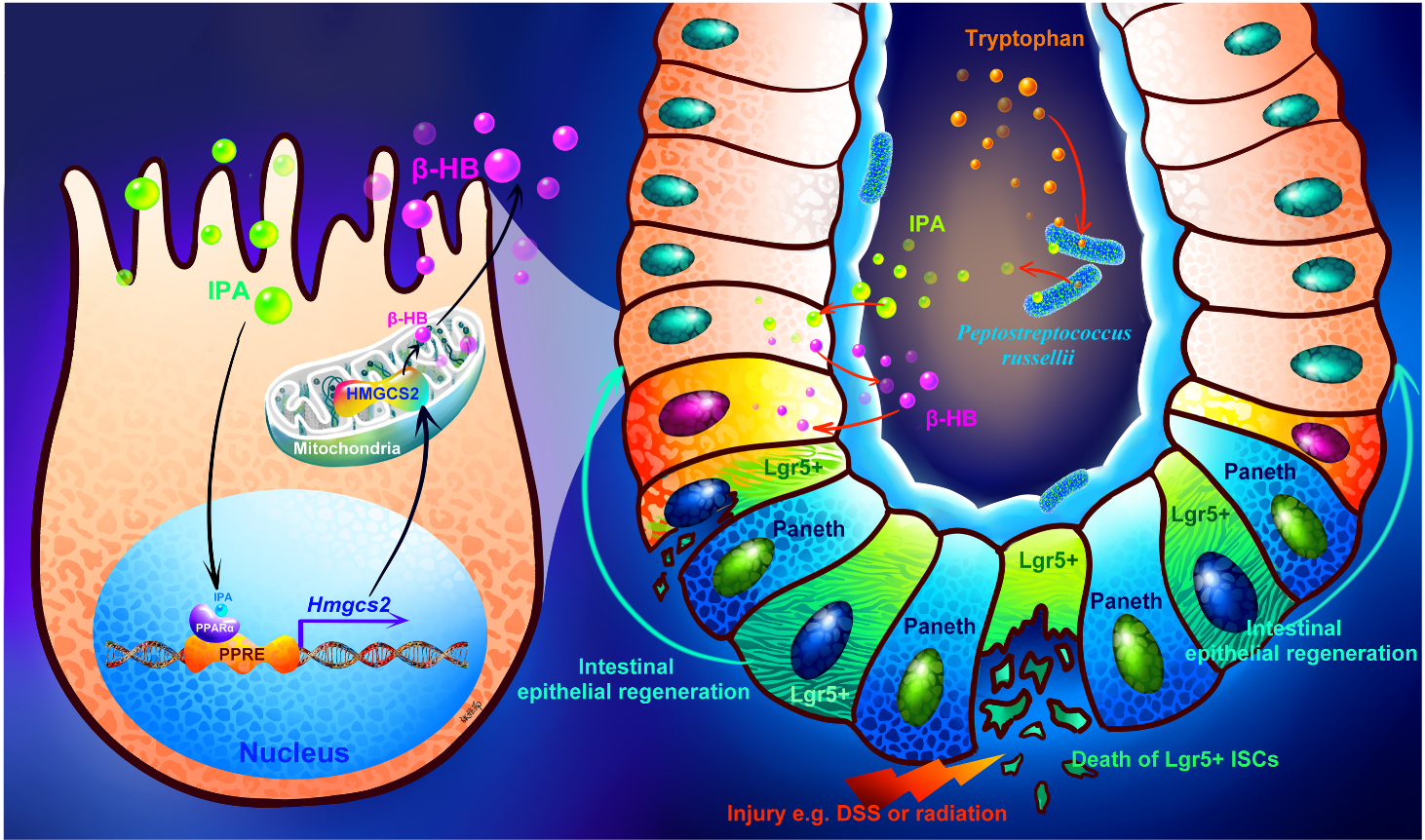
-
Paper 2 (BC‑ILA‑IPA‑HOPX⁺ reserve stem cell axis): We identified a specific bacterium – Blautia coccoides (BC) – that is depleted in IBD patients. BC cannot make IPA directly; it produces another metabolite, indole‑3‑lactic acid (ILA). ILA itself is inactive. But when BC lives together with IPA‑producing bacteria like Peptostreptococcus russellii or Clostridium sporogenes, ILA is converted into IPA. This activates a hidden reserve stem cell population (HOPX⁺ cells) that rebuilds the entire stem cell pool.
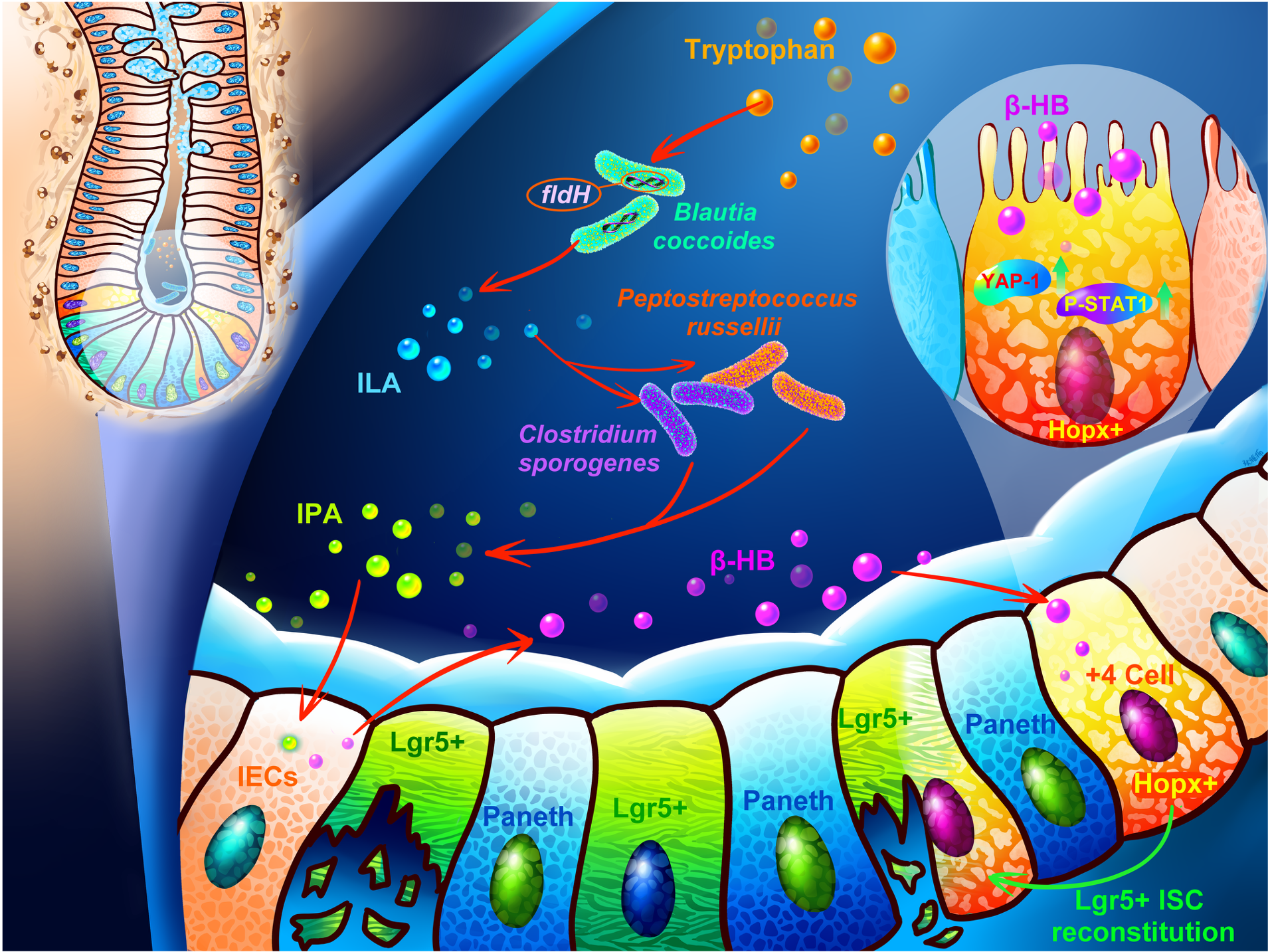
In other words: BC sets the table, and its partners cook the meal.
The struggles along the way
We would be lying if we said it was smooth sailing.
-
The split decision: Our original manuscript was too dense – one reviewer rightly said it tried to tell two stories at once. After much debate, we split it into two papers. That meant double the revisions, double the figures, and double the editor meetings. But it made each story cleaner.
-
The lost reviewer: Just as we thought we were done, our primary reviewer dropped out due to personal issues. The editor had to find a new arbitrator, adding months of delay. We watched new papers on tryptophan metabolites pop up almost weekly, feeling the pressure of being scooped.
-
Technical walls: We wanted to knock out the fldH gene in BC to prove it’s essential for ILA production. But BC is genetically intractable – no reliable tools exist. We had to settle for heterologous expression in E. coli, which worked but left a limitation we had to openly discuss.
Where do we go from here?
Our two papers open several exciting doors:
-
Therapeutic avenues: Could we use IPA or BHB as a pill to treat IBD? Or engineer a probiotic that produces the full ILA‑IPA cascade? We are already testing these ideas in preclinical models.
-
A new view of the microbiome: Microbial cross‑feeding is not just a footnote – it’s the rule. We are now mapping other metabolite handoffs in the gut, using the same “consortia” approach we developed for BC and its partners.
A personal note
Science is often portrayed as a series of “eureka” moments. In reality, it’s more like a long, slow climb with occasional rockfalls. We are deeply grateful to the reviewers who pushed us to split the paper, to the editor who found a new reviewer mid‑stream, and to our lab members who repeated experiments when the first three attempts failed.
If you take away one thing from our story, let it be this: the gut is a community, and healing is a conversation. Bacteria talk to each other, then talk to our stem cells, and together they repair the damage. Understanding that conversation might just change how we treat inflammatory diseases.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
-
A Collection of original research articles on all aspects of gut microbiota; spanning molecular and structural basis, in vitro models, symbiotic bacterial engineering, intervention approaches, and innovative translational or clinical therapeutics targeting gut microbiota.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in