Lithography-free, Pd-based bimorph cantilever switches for zero-standby-power chemo-mechanical H2 detection
Published in Bioengineering & Biotechnology and Materials
Hydrogen (H2) is attracting growing attention as a clean energy source. It can be used in fuel cells, industrial processes, and future energy systems that aim to reduce carbon emissions. However, because it is colorless, odorless, and highly flammable, even small leaks of hydrogen must be detected quickly and reliably to ensure safe use.
For that reason, hydrogen sensing has long been an important topic in microsystems research. Conventional hydrogen sensors have mainly relied on changes in electrical resistance. Although this approach is widely used, these sensors operate in a continuously powered mode, consuming energy at all times regardless of whether hydrogen leakage is actually present. They require persistent standby power, which reduces energy efficiency and increases maintenance demands such as battery replacement. This limitation becomes even more important in settings where electrical infrastructure and maintenance resources are limited. In such environments, event-driven hydrogen sensing, in which the device remains inactive with near-zero standby power until triggered by hydrogen exposure, offers a more practical route for long term operation.
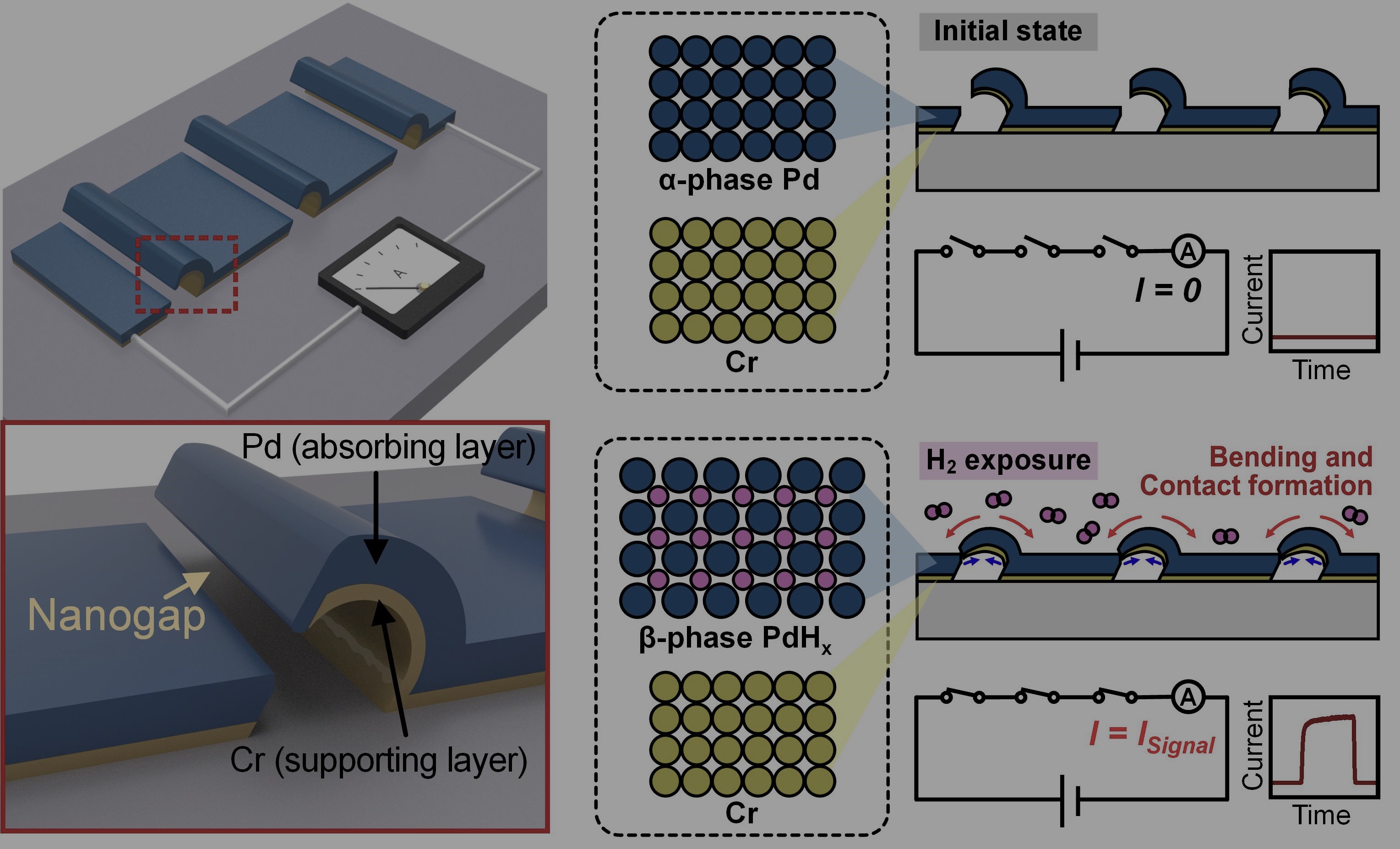
In our recent study, published in Microsystems & Nanoengineering, we demonstrated a chemo-mechanical hydrogen switch fabricated through a simple lithography-free process, which converts hydrogen-induced material expansion directly into an electrical switching event. The schematic of the proposed sensor and its working principle are illustrated in Figure 1. The device is based on a suspended palladium (Pd) and chromium (Cr) bilayer structure positioned above an electrode with a nanoscale gap.
In the initial state, the suspended bilayer remains separated from the counter electrode by a nanogap, so the circuit stays open and essentially no current flows, meaning near-zero standby power. Notably, Pd is a well-known hydrogen absorbing material. When exposed to hydrogen, Pd incorporates hydrogen atoms into its lattice and undergoes volume expansion, while Cr is mechanically stable. When hydrogen is introduced, the Pd layer transforms from α-phase Pd to β-phase PdHx and expands. This expansion induces bending in the bilayer, and once the deformation is large enough, the suspended structure makes physical contact with the opposite side electrode. The result is a clear electrical transition from the off state to the on-state with a higher current on/off ratio over 105. Rather than relying on small, continuously varying resistance changes, the device produces a distinct switching response with a high current on/off ratio through physical contact formation.
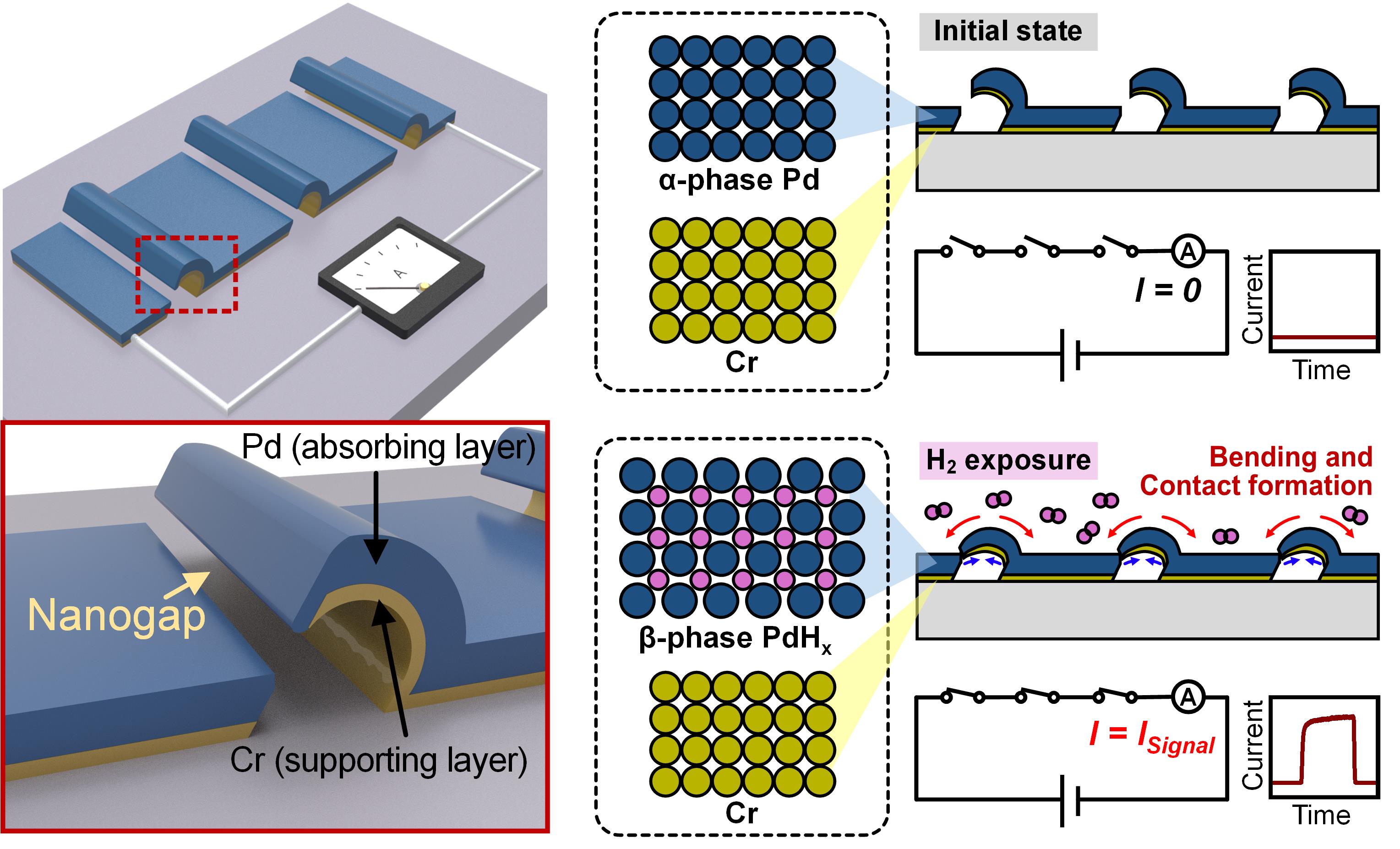
Figure 2a summarizes the lithography-free fabrication process. We first formed aligned polyethylene oxide (PEO) nanofibers, by directional electrospinning. PEO is a water soluble polymer, and here it serves as a sacrificial template that temporarily defines the space where the suspended bimorph structure and nanogap will later form. After placing these aligned nanofibers on the substrate, Cr and Pd were deposited at a tilted angle. This tilted deposition is important because it allows the metal layers to form a suspended bilayer geometry around the nanofiber template. Then, an additional Pd layer was deposited to further reduce the gap distance. Finally, the sample was immersed in deionized water, which dissolved and removed the sacrificial PEO nanofibers, leaving behind the suspended Pd and Cr bilayer structure with a nanoscale gap, as shown in the scanning electron microscope (SEM) image in Figure 2b. Instead of relying on conventional lithographic patterning and complex release steps, the switch structure can be created through directional electrospinning, angled deposition, and a simple water based template removal process. This makes the overall process comparatively straightforward and potentially lower cost, while still producing the nanoscale geometry required for chemo-mechanical switching.
Figure 2c shows an example of how the device responds as the hydrogen concentration is increased under the deposition parameters of a 15° tilted angle and a 40 nm additional Pd layer. The sensor exhibited a clear switching response once the hydrogen concentration exceeded the detection threshold of 0.3%, and the response increased monotonically up to 4% hydrogen. As the hydrogen concentration becomes higher, Pd absorbs more hydrogen, the bilayer bends more strongly, and the physical contact area increases, leading to a larger current signal. We further verified stable and repeatable switching over 50 cycles under both low hydrogen exposure at 0.3% and high hydrogen exposure at 2%, demonstrating the robustness of the device across a wide concentration range.
An especially important result is that the switching behavior can be tuned by controlling the fabrication parameters. As shown in Figure 2d and 2e, the tilted angle and the additional Pd layer thickness significantly affect both the current on/off ratio and the hydrogen detection limit. In practical terms, these parameters determine how easily the suspended bilayer can make contact with the opposite electrode under hydrogen exposure. A smaller deposition angle and a thicker additional Pd layer reduce the nanogap, enabling easier contact formation, and a lower detection threshold. Under optimized conditions, the device showed a current on off ratio approaching 105 and detected hydrogen down to 0.3%. This tunability is an important strength of the proposed platform. Since the switching threshold can be adjusted through fabrication, the same device can be tailored for different safety scenarios without changing the overall sensing mechanism, while preserving the advantage of near-zero standby power.
Figure 2. Fabrication process and switching performance of the chemo-mechanical hydrogen switches.
a Schematic illustration of the summarized fabrication process. PEO nanofibers were first formed by directional electrospinning as sacrificial templates, followed by tilted angle deposition of Cr and Pd, additional Pd deposition, and PEO removal. b Cross sectional SEM image showing the nanogap and bilayer structure. c An example of switching responses of the device under increasing hydrogen concentrations. d Current on/off ratio as a function of hydrogen concentration for different fabrication parameters. e Hydrogen detection limit for devices fabricated with different tilted deposition angles and additional Pd layer thicknesses.
In summary, this work demnstrates a chemo-mechanical hydrogen sensing switch based on a suspended Pd and Cr bimorph structure with lithography-free process. By combining a simple fabrication process with event-driven switching operation, the device achieves near-zero standby power while providing a clear electrical response only when hydrogen is present. The switching threshold can also be tuned through control of fabrication parameters, making the platform adaptable to different safety requirements. Overall, this approach offers a practical route toward compact, low maintenance hydrogen leak alarms and other long term safety monitoring systems where energy efficiency and reliability are essential.
Follow the Topic
-
Microsystems & Nanoengineering
This journal, with a target for a high-end journal for years to come, seeks to promote research on all aspects of microsystems and nanoengineering from fundamental to applied research.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in