Phosphine-Mediated Hydrogen Bond and Phosphorescence Energy Transfer for Tunable Chiroptical Afterglow in Stacked Polymers
Published in Chemistry
Chiral organic afterglow materials have fascinated scientists for more than a decade, promising applications ranging from anti-counterfeiting and information encryption to advanced photonic displays. Yet, achieving afterglow emission together with chiroptical activity remains a formidable challenge. The reason is deceptively simple: the excited triplet states of organic molecules are easily quenched by molecular motion, heat, or environmental perturbations. Introducing chirality further complicates the picture, often at the cost of emission efficiency or spectral tunability.
In our recent work, we asked a fundamental question: can non-covalent interactions be engineered precisely enough to simultaneously stabilize long-lived afterglow and achieved chiroptical signals in soft polymer systems? Our answer lies in an underexplored but powerful interaction—phosphorus-mediated hydrogen bonding.
Most polymer-based afterglow systems rely on carboxyl, hydroxyl, or boronic acid groups to immobilize luminophores through hydrogen bonds. While effective, these interactions are often too weak or geometrically disordered to support ultralong lifetimes, especially under elevated temperatures or ambient conditions. Phosphonic acid groups, however, offer a unique alternative. Their tetrahedral geometry, dual proton-donor capability, and strong P=O polarization favor the formation of near-linear, three-dimensional hydrogen-bond networks with high binding energies.
By embedding a phosphonic acid-functionalized carbazole (2PACz) into poly(vinyl alcohol) (PVA), we constructed a robust supramolecular network that effectively suppresses non-radiative decay. The result is a transparent, flexible polymer film exhibiting intense blue afterglow with a lifetime exceeding three seconds and a photoluminescence quantum yield above 33.3%. Compared with carboxyl-based analogues, the phosphonic acid system shows markedly improved thermal stability, highlighting the decisive role of hydrogen bond strength and directionality.
Chirality was introduced not by modifying the luminophore itself, but through a stacked polymer design. By coating the afterglow-active PVA film with a thin layer of chiral polylactic acid (PLA), we achieved efficient chiral amplification via interfacial polarization. This modular strategy avoids complex chiral synthesis and yields circularly polarized afterglow with dissymmetry factors up to 3×10-2, which are among the highest reported for polymer-based systems.
Beyond blue emission, spectral tunability is essential for real-world applications. Leveraging the long-lived triplet excitons of the phosphonic-acid-stabilized matrix, we introduced phosphorescence energy transfer to fluorescent dyes, enabling green, yellow, and red afterglow without sacrificing afterglow properties. When combined with chiral PLA layers, these stacked films deliver multicolor circularly polarized afterglow spanning the visible spectrum.
The implications extend well beyond fundamental photophysics. The water solubility, processability, and flexibility of these polymers allow their use as afterglow inks, coatings, and printable security features. Information can be encoded not only by color and time, but also by chirality, adding an additional, invisible dimension to encryption and anti-counterfeiting technologies.
More broadly, this work highlights how rational supramolecular engineering, guided by hydrogen-bond geometry and electronic structure, can unlock new functionalities in organic optoelectronic materials. We hope it inspires further exploration of unconventional non-covalent interactions as design tools for next-generation luminescent systems.
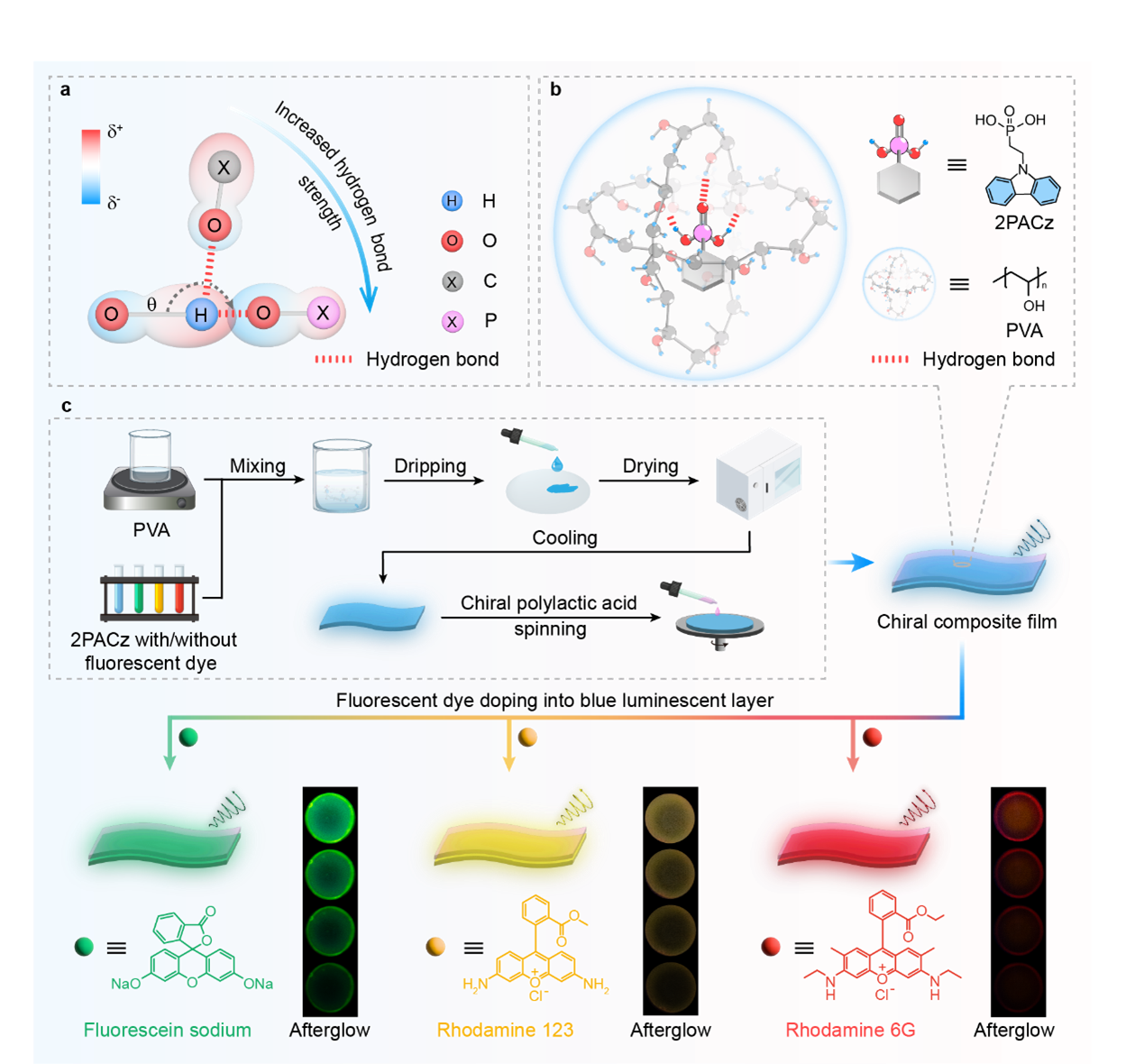
Fig. 1 | Schematic diagram for constructing multi-color chiral organic afterglow (COA) by hydrogen bond mediation and phosphorescence energy transfer. a Principle drawing for hydrogen bond strength mediation through modulating electronegativity of hydrogen bond acceptor and spatial arrangement of hydrogen bond. b Formation of a three-dimensional hydrogen bond network mediated by phosphorus in polyvinyl alcohol (PVA) film and the molecular structure of 2PACz and PVA. c Preparation procedure and corresponding afterglow photographs of multi-color COA films, as well as the molecular structures of the incorporated multi-color fluorescence emitters.
The related paper has been published in Nature Communications. Please see details: Zhisheng Gao, Shuai Huang, Xiaona Lian, Xin Yan, Hengyu Cao, Shuman Zhang, Peng Zhang, Qi Jia, Huanhuan Li*, Hui Li, Runfeng Chen, Gaozhan Xie, Yun Ma, Ting Wang, Wei Huang* & Ye Tao*. Phosphine-Mediated Hydrogen Bond and Phosphorescence Energy Transfer for Tunable Chiroptical Afterglow in Stacked Polymers. Nat. Commun. 2026, https://doi.org/10.1038/s41467-026-69324-0.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in