Prenatal Testing Through Molecular Pathology
Published in General & Internal Medicine
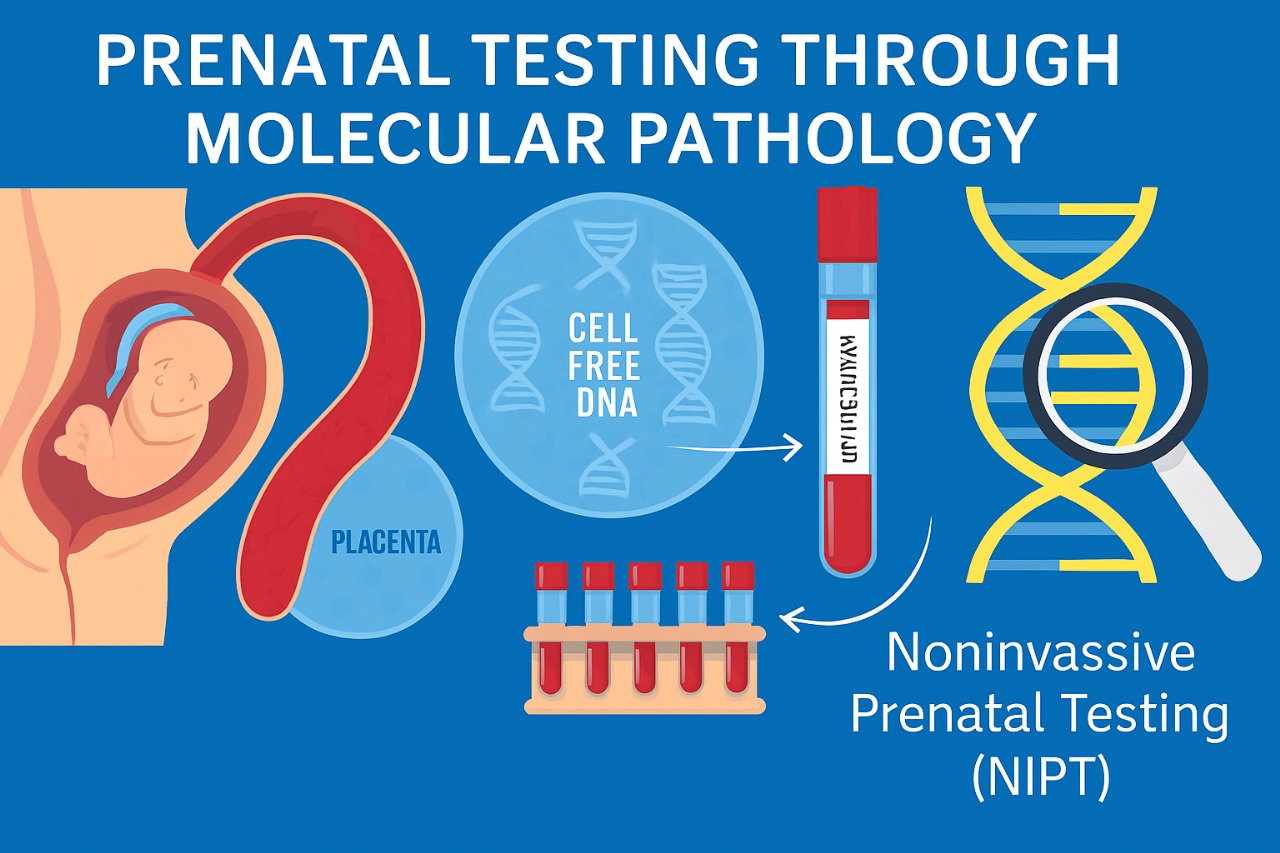
Techniques in Prenatal Molecular Testing
Prenatal molecular testing employs both non-invasive and invasive methods to assess fetal genetic health. These approaches vary in application, accuracy, and risk, and together they form the cornerstone of modern prenatal diagnostics.
1. Non-Invasive Prenatal Testing (NIPT)
NIPT primarily screens for chromosomal aneuploidies such as Trisomy 21 (Down syndrome), Trisomy 18 (Edwards syndrome), Trisomy 13 (Patau syndrome), and sex chromosome abnormalities. NIPT offers high sensitivity and specificity without any risk to the fetus, making it a preferred screening method. However, it remains a screening tool rather than a diagnostic method, and positive results require confirmation through invasive testing.
-
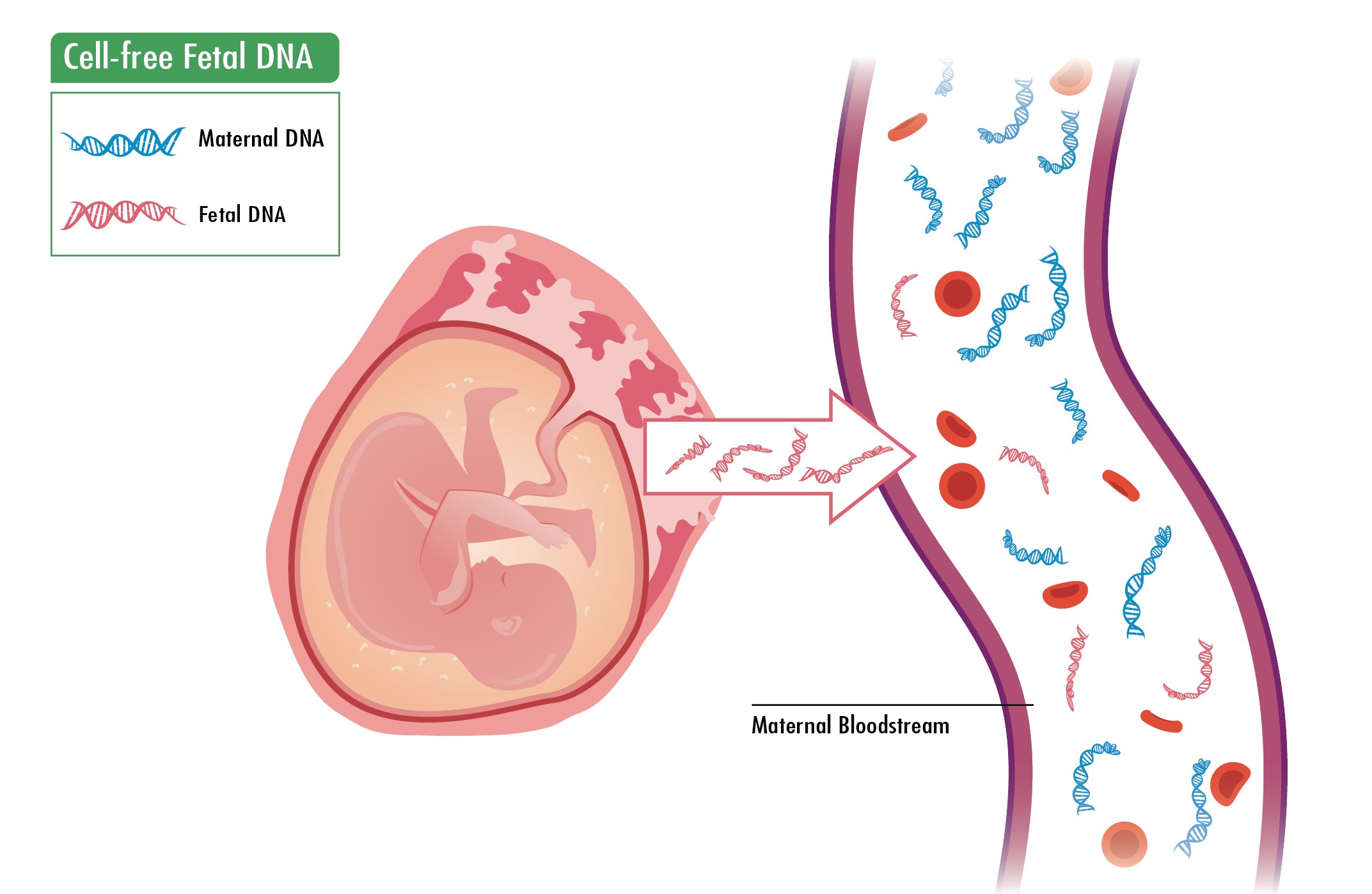
Principle: Detects cell-free fetal DNA (cffDNA) present in maternal blood.
-
Timing: Performed as early as 10 weeks of gestation.
-
Techniques Used:
-
Next-Generation Sequencing (NGS) – Enables genome-wide analysis for detecting chromosomal aneuploidies.
-
Digital PCR – Provides accurate quantification of specific genetic sequences.
-
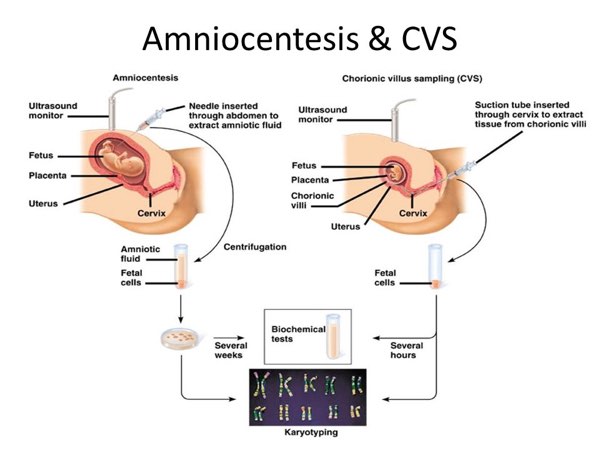
2. Invasive Diagnostic Methods
When definitive diagnosis is needed, invasive procedures such as Chorionic Villus Sampling (CVS) and amniocentesis are employed. CVS, performed between 10–13 weeks, involves sampling placental tissue, enabling early detection of chromosomal and genetic disorders. Amniocentesis, usually conducted after 15 weeks, involves collecting amniotic fluid containing fetal cells. Both methods allow direct genetic analysis but carry a small risk of complications, including miscarriage.
3. Molecular Techniques for Fetal Cell Analysis
Fetal cells obtained from CVS or amniocentesis undergo advanced molecular testing using several techniques.
-
Polymerase Chain Reaction (PCR)
-
Detects known mutations or small deletions/insertions in single-gene disorders (e.g., cystic fibrosis, sickle cell disease).
-
-
Multiplex Ligation-Dependent Probe Amplification (MLPA)
-
Identifies copy number variations (CNVs) such as microdeletions or duplications.
-
-
Fluorescence In Situ Hybridization (FISH)
-
Rapid detection of major aneuploidies (e.g., chromosomes 13, 18, 21) and some structural rearrangements.
-
-
Next-Generation Sequencing (NGS)
-
Comprehensive detection of point mutations, microdeletions, and rare disorders through whole-exome or targeted panels.
-
Ethical and Clinical Considerations
The implementation of prenatal molecular testing raises important ethical and legal considerations. Informed consent, genetic counseling, and respect for patient autonomy are essential components of responsible testing. The disclosure of results must maintain confidentiality while supporting parents in making informed decisions regarding pregnancy continuation or termination. As technology advances, concerns regarding incidental findings and equitable access to testing remain central to ethical practice.
Future Perspectives
The future of prenatal molecular pathology lies in whole-genome and whole-exome sequencing, which will enable comprehensive detection of genetic variants from a non-invasive maternal blood sample. Integration with artificial intelligence and machine learning algorithms will further enhance predictive accuracy and interpretation.
Additionally, emerging CRISPR-based diagnostic platforms hold promise for ultra-sensitive, rapid detection of pathogenic mutations, paving the way for earlier interventions and personalized prenatal care.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in