Quieting the Inflammatory Storm After Stroke: Insights from the NUVISTA Trial
Published in General & Internal Medicine
TRANSLATIONAL STROKE EXCHANGE — TSE BLOG
ABOUT THE STUDY
Despite remarkable advances in reperfusion therapy for acute ischemic stroke, up to half of patients with large vessel occlusion (LVO) still face significant disability even after successful thrombectomy. The inflammatory cascade unleashed by ischemia and reperfusion continues to damage vulnerable brain tissue long after the artery is reopened — yet anti-inflammatory drug trials have repeatedly failed to alter this trajectory. Could neuromodulation be a different path forward?
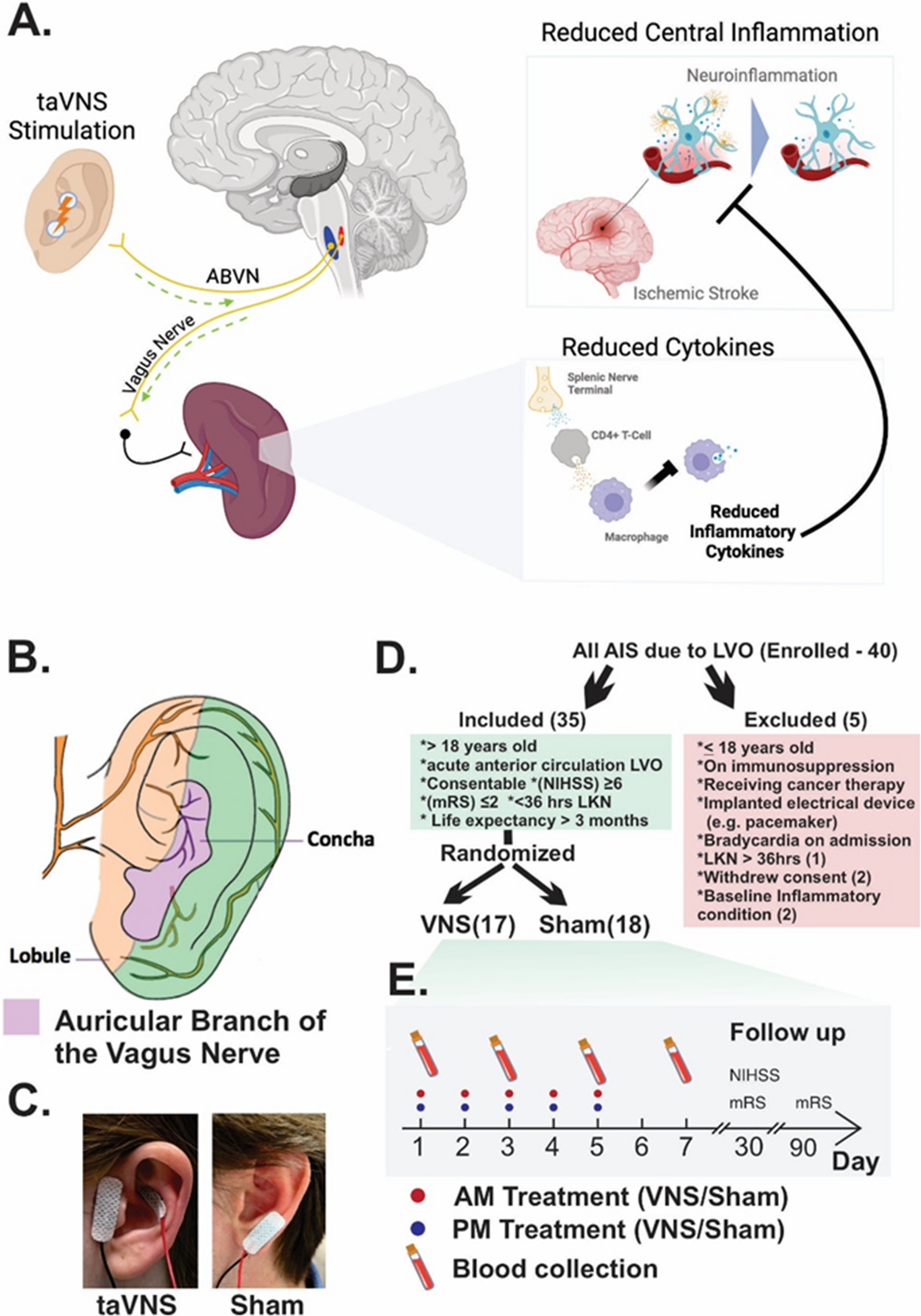
The NUVISTA trial, recently published in Translational Stroke Research (Leuthardt et al., 2025; DOI: 10.1007/s12975-025-01405-6), offers a compelling proof of concept. Using a small, non-invasive device clipped to the ear, the trial tested transcutaneous auricular vagus nerve stimulation (taVNS) as an adjunct to standard LVO stroke care. The primary biological target: the post-stroke inflammatory response, measured through circulating cytokine levels. The headline result was striking — taVNS significantly reduced IL-6 levels compared to sham treatment, with effects most pronounced around day 3, precisely when the inflammatory surge typically peaks.
To unpack the science, the clinical implications, and the road ahead, the TSE Editorial Team spoke with Dr. Eric Leuthardt, the trial’s lead investigator and a neurosurgeon-scientist at Washington University in St. Louis, whose work sits at the intersection of neuromodulation, brain-computer interfaces, and acute brain injury.
THE INTERVIEW
Q1. First, congratulations on this important study. What do you see as the central take-home message of this trial for the stroke field, especially in patients with large vessel occlusion?
The headline finding is both simple and exciting: a small, non-invasive device clipped to the ear can meaningfully quiet the inflammatory storm that follows a major stroke — and it does so safely. We’ve long known that inflammation makes brain injury worse after acute ischemic stroke, and despite many attempts, anti-inflammatory drugs have largely failed in this setting. What the NUVISTA trial shows is that stimulating the vagus nerve through the ear — without surgery, without systemic drugs, without significant side effects — can measurably change the biology of that inflammatory response. Specifically, taVNS significantly changed IL-6 levels compared to sham treatment, with an overall p-value of 0.0001. For a field that has been searching for adjunct therapies to complement thrombectomy, that’s a genuinely meaningful proof of concept.
Q2. This study focused on post-stroke inflammatory biomarkers, with IL-6 emerging as the clearest signal. What led you to prioritize inflammation as the key therapeutic target for taVNS in this patient population?
This is really the heart of the scientific rationale, and it comes down to a convergence of biology and unmet need. Even after successful recanalization with mechanical thrombectomy, up to half of patients remain with significant disability — highlighting the need for adjunct therapies to improve outcomes. Reperfusion saves tissue, but it also triggers a cascade of inflammatory injury that continues to damage the brain in the hours and days that follow. Neuroinflammation has long been recognized as an important contributor to ischemic brain injury, and in the acute phase of stroke, elevated inflammatory biomarkers are associated with exacerbation of that injury.
What makes taVNS so appealing as a tool to address this is the underlying neurobiology. It is postulated that taVNS leads to immunomodulation through the cholinergic parasympathetic nervous system anti-inflammatory pathways that innervate the spleen, culminating in T cell release of acetylcholine, which interacts with acetylcholine receptors on immunocompetent cells to inhibit cytokine release in macrophages. In other words, the vagus nerve is essentially the body’s own built-in anti-inflammatory circuit — and we’re just activating it with a gentle electrical signal through the ear. That’s an elegant and exciting idea.
IL-6 emerged as the clearest signal because it also happens to be a cytokine with strong clinical relevance. Prior studies suggest that even a modest early rise in IL-6 within the first 24 hours is associated with a 13% higher odds of poor 3-month functional outcome and a 16% higher odds of mortality. So reducing IL-6 isn’t just a laboratory curiosity — it may have real consequences for how patients do.
Q3. Most patients in your cohort underwent thrombectomy, while some also received thrombolysis. Do you see taVNS primarily as an adjunct to thrombectomy, or more broadly as an adjunct strategy for acute large vessel occlusion stroke care?
We think about it as a broadly applicable adjunct for the acute LVO stroke population, rather than something tightly coupled to thrombectomy alone. Thrombolytics were administered to 43% and thrombectomy was attempted in 94% of the cohort. So in the real world, the vast majority of these patients are getting both reperfusion therapies, and taVNS fits naturally alongside that standard-of-care approach.
What’s particularly encouraging — and perhaps counterintuitive — is that taVNS still appeared to work even in patients who had more challenging strokes. The taVNS treatment group had more proximal LVO, longer times from last known normal to recanalization, and worse reperfusion scores than the sham group. Despite these baseline imbalances, we still observed an effect on inflammatory biomarkers — highlighting the potential robustness of this intervention. That’s really heartening, because it suggests this isn’t just a therapy for the ‘easy’ cases. If anything, the patients with harder-to-treat strokes — incomplete reperfusion, more proximal occlusions — may be exactly the population with the most to gain from an anti-inflammatory adjunct.
Q4. Your study showed biologic effects on circulating inflammatory markers, with IL-6 emerging as the clearest overall signal and several cytokine differences appearing most prominently around day 3. How do you interpret that temporal pattern biologically?
The timing is fascinating, and we think it reflects the natural arc of the post-stroke immune response. In general, cytokine trajectories appeared U-shaped — an inverted U for sham-treated patients and an upright U for taVNS-treated patients. While IL-6 trajectories were significantly different between groups overall, post-hoc analysis also found potential differences in Day 3 cytokine levels for IL-1β, IL-6, and IL-17α.
The reason Day 3 stands out is that this is roughly when the acute inflammatory surge — triggered by ischemia and reperfusion — tends to peak before the immune system begins to recalibrate. There is substantial evidence that VNS reduces systemic inflammation in multiple pre-clinical models, and in animal models of cerebral ischemia and reperfusion injury, VNS not only reduced infarct size but also improved neurological outcomes. What taVNS appears to be doing is attenuating that peak — essentially blunting the inflammatory crescendo at precisely the moment when it would otherwise be doing the most damage to vulnerable but potentially recoverable brain tissue. It’s a bit like turning down the volume on a loudspeaker at the exact moment it would otherwise distort.
There’s also an interesting observation about IL-10, which is classically considered anti-inflammatory but behaved similarly to IL-6 in our cohort. This is consistent with prior studies showing that the physiological inverse correlation between IL-6 and IL-10 is disrupted after stroke, with both cytokines rising within shared post-injury pathways. taVNS produced reductions across multiple cytokines, including IL-10, suggesting its effects may reflect a broader attenuation of post-stroke immune responsiveness rather than a selective shift in pro- or anti-inflammatory cytokines. That broader immunomodulatory effect — rather than selective cytokine targeting — may ultimately be one of its greatest strengths.
Q5. Looking ahead, what do you see as the most important next step: validating the anti-inflammatory signal in a larger cohort, identifying the patients most likely to benefit, or integrating biomarker effects with imaging and clinical endpoints?
If we had to sequence them, we’d say the most urgent priority is validating and extending the anti-inflammatory signal in a larger, adequately powered cohort. Although cytokine changes were consistent and statistically significant, replication in a larger cohort is warranted. The current study provides pilot data to allow us to perform the larger pivotal trial to test efficacy and clinical significance.
That said, the question of who benefits most is deeply intertwined with that validation work. Early changes in IL-6 correlated with 90-day mRS scores, with higher IL-6 levels correlating with higher mRS values; there was also a statistically significant relationship between IL-6 and NIHSS scores, with higher IL-6 levels correlating with higher NIHSS. This tells us that IL-6 trajectory isn’t just a biological curiosity — it tracks with real clinical outcomes. That opens the door to using IL-6 as a biomarker to identify which patients are most inflamed and therefore most likely to benefit from intervention — a precision medicine approach for neuromodulation in stroke that we find tremendously exciting.
The NUVISTA findings support further investigation of taVNS as a potential adjunctive therapy through larger, multicenter trials. We believe the field is ready for that next chapter, and this trial gives us the scientific and logistical foundation to pursue it with real confidence.
EDITORIAL NOTE
This interview was conducted by the Translational Stroke Exchange Editorial Team as part of TSE’s ongoing series highlighting translational advances in stroke science. The NUVISTA trial is published in Translational Stroke Research: Leuthardt et al., 2025 (DOI: 10.1007/s12975-025-01405-6).
Responses have been lightly edited for length and clarity.
Follow the Topic
-
Translational Stroke Research
A journal committed to the development of new strategies for prevention, assessment, treatment, and repair after stroke and neurotrauma.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in