Skin Neglected Tropical Diseases: Why One Health is the way forward
Published in Microbiology

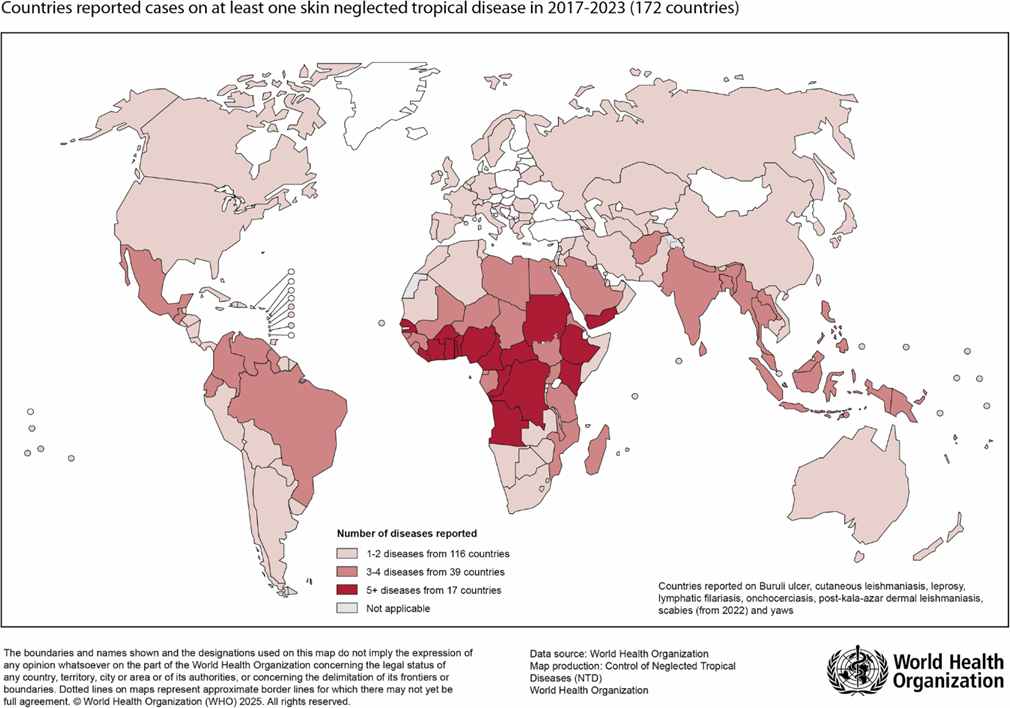
Skin-related diseases are the fourth leading cause of disease burden worldwide, yet a significant subset (the skin neglected tropical diseases (skin NTDs)) remain chronically neglected. An estimated one billion people are at risk of, or are affected by, at least one skin NTD, although the true scale is almost certainly larger given how poorly many of these conditions are captured by existing surveillance networks.
A recent review in Nature Communications by Mosi and colleagues (2026) makes the case that improved prevention and control of these diseases demands more than clinical interventions alone, and that a comprehensive One Health approach connecting human medicine, animal science, and environmental research is needed.
The problem in plain sight
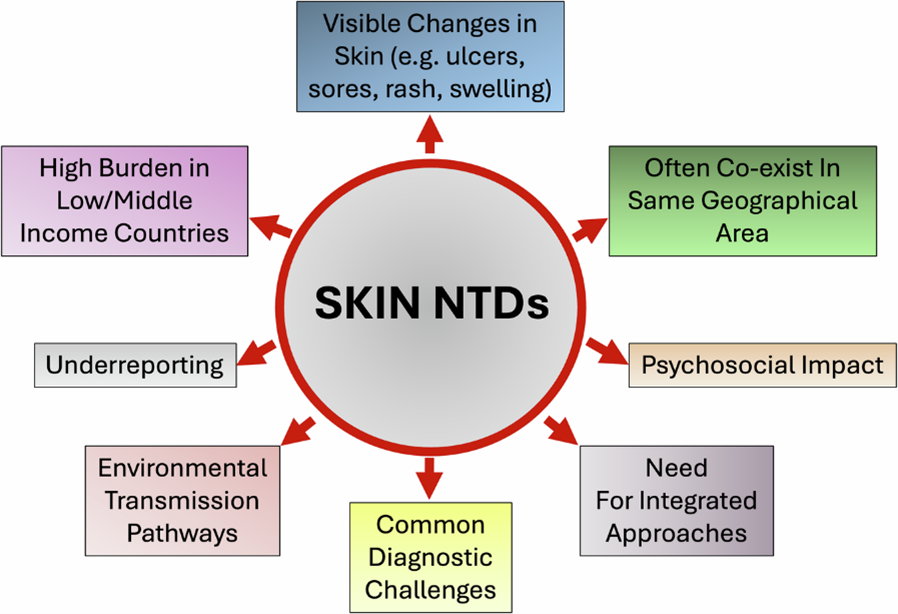
At least 10 of the identified NTDs are skin diseases, and include conditions such as Buruli ulcer, leprosy, cutaneous leishmaniasis, mycetoma, yaws, scabies, tungiasis, onchocerciasis, and lymphatic filariasis (elephantiasis). These diseases share several defining features - visible skin surface lesions, endemicity in the same communities, a heavy burden in low- and middle-income countries, and severe psychosocial consequences, including stigma, financial hardship, and barriers to seeking care.
Left unmanaged, they can progress to secondary systemic infections, non-traumatic amputation, and premature death, and many cause chronic disabilities for life if not diagnosed early. The WHO's 2021-2030 NTD roadmap and a landmark 2025 resolution recognise skin diseases as a global public health priority.
Many skin NTDs cannot be prevented through mass-scale drug administration alone. Instead, they require individual-level diagnosis and often long-term, resource-intensive treatments (e.g. 8-week course of dual antibiotic therapy combined with wound care for Buruli ulcer). This kind of care is hardest to deliver, particularly in remote, under-resourced settings where these diseases are most prevalent, making skin NTDs particularly neglected.
The case for integration
Historically, NTD control has been organised around disease-specific programmes, an approach that can concentrate expertise but also duplicates effort and fragments access to limited resources. The WHO's 2022 strategic framework for skin NTDs marked a deliberate shift, whereby integrated approaches were promoted that exploit the shared feature all these conditions have in common - the skin itself.
Intiutively, as many skin NTDs are often co-endemic, a single community screening visit can detect multiple conditions simultaneously - post-kala-azar dermal leishmaniasis during a leprosy case-finding campaign, for instance, or scabies alongside tungiasis in school-based surveys.
Integration improves understanding of the combined disease burden, strengthens advocacy and funding cases, and makes better use of trained personnel. This integration can take multiple forms, from combining two or three related treatment programmes, to embedding skin NTD diagnosis within primary care more generally.
Two transmission categories: known and unknown
There is a clear distinction between skin NTDs whose transmission is well characterised and those where critical knowledge gaps persist. Onchocerciasis (transmitted by blackflies), cutaneous leishmaniasis (sandflies), lymphatic filariasis (mosquitoes), scabies (person-to-person contact), yaws (skin-to-skin contact), and tungiasis (Jigger (sand) fleas) all have broadly understood transmission routes, though even here, questions remain about strain diversity, non-human reservoirs, and the influence of climate and land-use change.
However, for Buruli ulcer, leprosy, and mycoses, the picture is far murkier:
- Buruli ulcer is associated with aquatic environments, and has been linked to possums and mosquitoes in Australia for example, yet the precise mechanism by which Mycobacterium ulcerans (the causative agent of Buruli ulcer) reaches human skin remains debated.
- Leprosy persists, despite effective multi-drug therapy, partly because of environmental reservoirs in the soil and water, and zoonotic hosts such as armadillos (in the Americas), red squirrels (in Europe), and primates, sustaining bacterial circulation outside the human population.
- Mycoses (fungal infections) are known to be acquired through skin trauma, but the ecological niches, climatic preferences, and geographic distributions of their causative fungi and bacteria are poorly mapped.
Why One Health matters
This is where the One Health framework becomes indispensable - Mosi and colleagues outline how skin NTD transmission sits at the intersection of landscape disturbance, pathogen ecology, animal reservoirs of infection, human behaviour, and individual susceptibility.
Addressing any one of these dimensions in isolation will only result in partial control success. For example, primates harbour the yaws pathogen (Treponema pallidum subspecies pertenue) in West Africa, meaning elimination strategies focused solely on human treatment may fail to interrupt transmission. Landscape modelling with integrated case mapping and remote sensing data has already proven valuable for understanding Buruli ulcer distribution, and the success of an EcoHealth approach to Chagas disease in Guatemala (which broke transmission for over two decades), demonstrates that multi-disciplinary, environmentally informed interventions can deliver lasting results.
Laboratory networks, such as LABNET, are also highlighted as highly successful initiatives at creating standardised, combined, next generation diagnostic protocols that are effective at diagnosing multiple skin NTDs – this is particularly important as many skin NTDs are co-endemic, and it cuts the costs of diagnosing any single disease.
Looking ahead
Moving forward, the authors emphasise that prioritisation of funding for integrated studies linking human cases to animal and environmental reservoirs is essential, along with expanding standardised diagnostic networks beyond Africa, and systematic collection of data related to the financial and social costs of skin NTDs in order to underpin robust cost-effectiveness modelling.
The skin offers something unusual in infectious disease control - a visible, accessible entry point for detection, diagnosis, and management across multiple co-endemic conditions. Realising that potential, however, means looking well beyond the skin itself, into the ecosystems, animal populations, and social structures that drive transmission. As with many other zoonotic diseases, One Health may be the only realistic path to meaningful, lasting control.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
-
BugBitten
A blog for the parasitology and vector biology community.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Healthy Aging
Publishing Model: Open Access
Deadline: Dec 31, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in