Standardized lung function reference values in rats for translational respiratory research
Published in Biomedical Research and Anatomy & Physiology
Why a “normal” lung function value in rats might not be normal at all
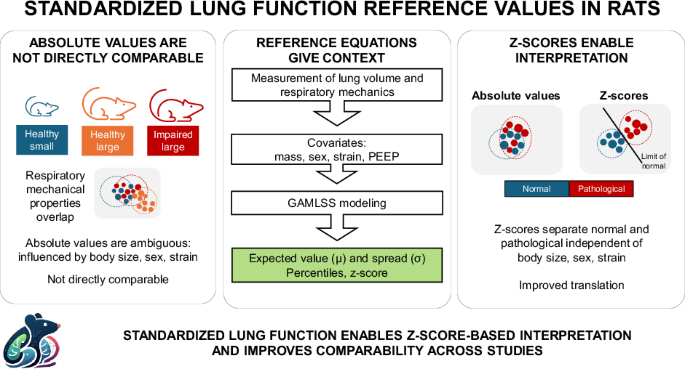
In clinical medicine, a single lung function value rarely stands on its own. If a patient's airway resistance or lung volume is measured, the number only becomes meaningful when compared to what is expected for someone of the same age, size, and sex, typically expressed as a z-score that captures how far a measurement deviates from normal biological variation.
Preclinical research has not followed the same logic. Studies in animal models, including rats, typically report absolute values: a number is presented, groups are compared, conclusions are drawn. Without a reference framework, it is often genuinely unclear whether a measured value is abnormal or simply reflects natural variability between animals.
This gap is what motivated the work behind our paper.
The problem
Rats are widely used to study respiratory diseases and test new therapies. Modern oscillometry techniques (including the forced oscillation technique used in several commercially available devices) can capture detailed information about lung mechanics and generate large amounts of high-quality data. Interpretation, however, has remained surprisingly limited.
Two animals with different lung function values may or may not differ meaningfully. Differences in body mass, sex, or strain can easily obscure real biological effects. In clinical practice, reference equations and z-scores solved this problem decades ago. In preclinical respiratory research, no comparable system has existed.
Building reference equations for rat lung function
We studied 182 healthy rats from two commonly used strains, including both males and females across a range of body masses, measuring airway resistance, tissue damping, tissue elastance, and end-expiratory lung volume.
Critically, we did not only model the average expected value for each parameter. We also modelled the expected variability, accounting for body size, sex, strain, and ventilatory settings. This allows any individual animal's measurement to be placed within the expected biological distribution and expressed as a z-score. In practical terms: we can now ask whether a value is actually abnormal, not just whether it differs from a group mean.
When the signal was hidden in plain sight
Applying this framework to existing experimental data produced a striking result.
In a separate study, rats had been treated with bleomycin to induce lung fibrosis. Looking at raw lung function values, the results were difficult to read; there was substantial overlap between healthy and diseased animals, and differences in body size and sex further complicated direct comparison. Once the same data were expressed as z-scores, the picture changed substantially. Most diseased animals fell outside the normal range; nearly all healthy controls remained within it.
The biological signal had been there throughout. It was obscured by variability that the z-score framework was able to account for.
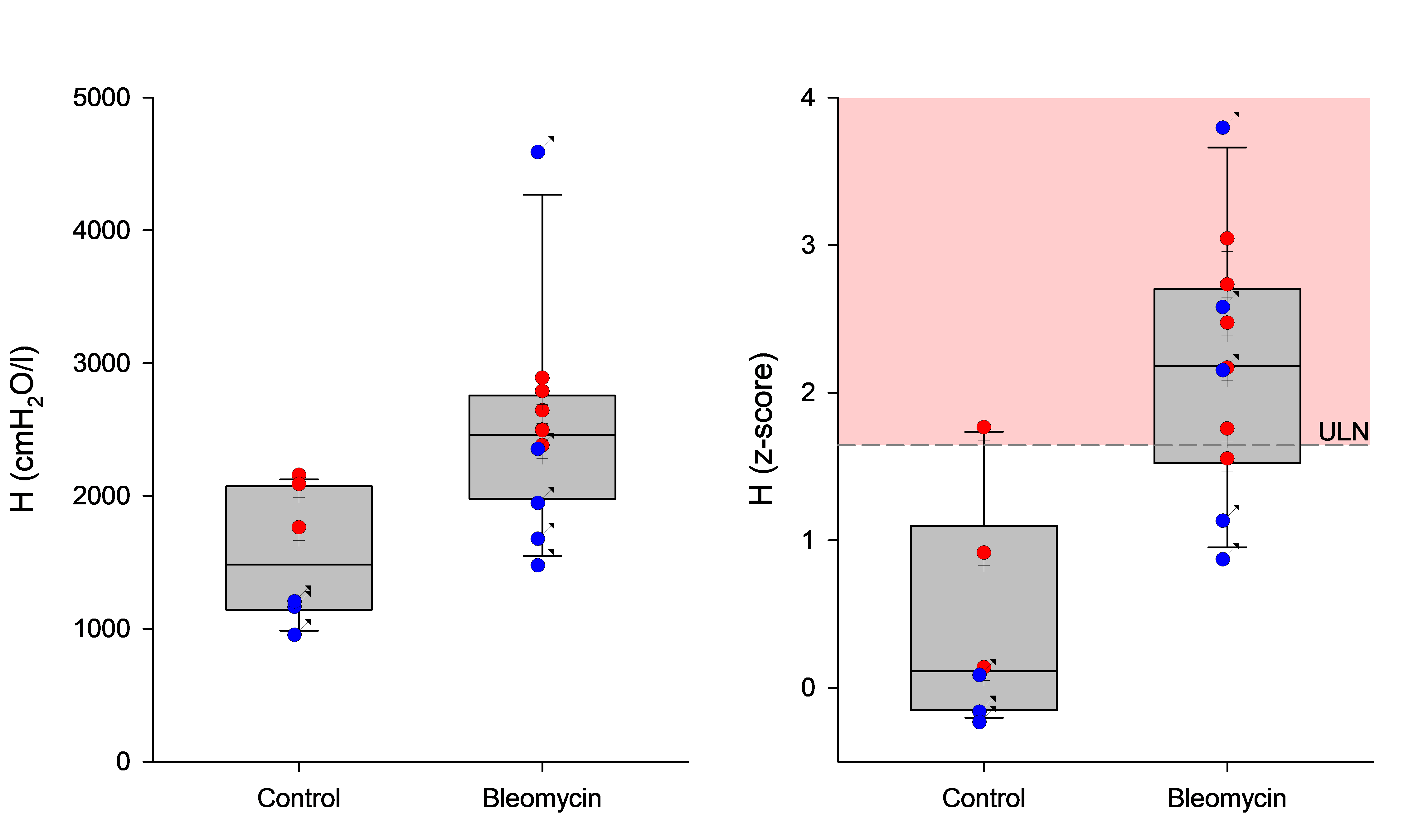
Variability is structured, not just noise
One of the more important findings was that variability itself is not uniform. Sex and strain influence not only average lung function values but also the degree of within-group spread. Two animals with identical measurements may not be equally "normal" if their expected variability differs. Modelling both the expected value and its dispersion is therefore essential for accurate interpretation; it is not a statistical nicety.
Practical implementation
To make the framework usable, we developed open-access tools tfor calculating predicted values and z-scores: an Excel-based calculator and R scripts that can be integrated into existing workflows. The aim was to make interpretation in rats as straightforward as it is in the clinic.
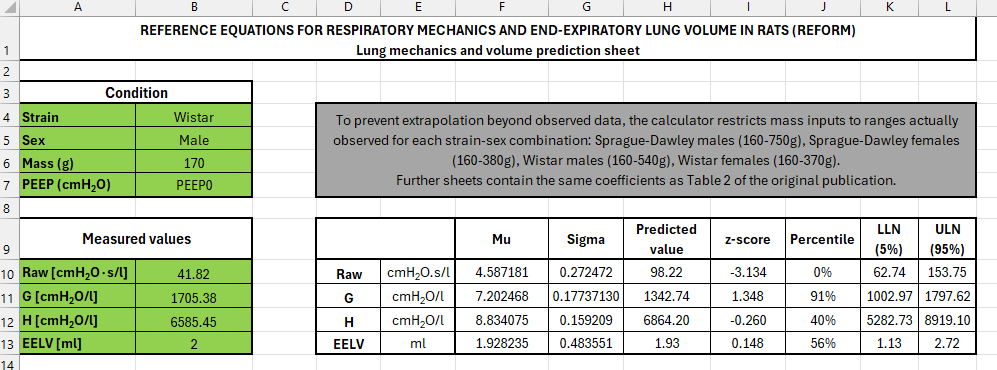
Broader implications
The immediate application is analytical, but the consequences extend further. A standardised reference framework for rat lung function supports reproducibility across laboratories, reduces ambiguity in experimental results, and brings preclinical reasoning closer to clinical practice. This alignment matters most in translational research, where the gap between animal models and human disease is often where studies succeed or fail.
There are limitations. The current models cover two rat strains, and extension to other strains, species, and experimental conditions will require additional data. The underlying principle, however, is not strain-specific: physiological measurements are more informative when interpreted relative to expected biological variation than as raw numbers alone. This has been standard in clinical medicine for decades. Applying it systematically to preclinical research is long overdue.
This work was supported by the National Research, Development and Innovation Office of Hungary (grants FK134274, K138032, and NKKP-152553).
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
🔍 Ask the Editor – Clinical medicine, Respiratory physiology, and Cardiology
I’m excited to connect with a global network of specialists through the Research Communities – how will you get involved?
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
Mechanistic insights into human host and microbiome interactions
Publishing Model: Open Access
Deadline: May 31, 2026
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Jun 30, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in