Targeting the bromodomain and extra-terminal domain (BET) proteins in cancer
Published in Cancer
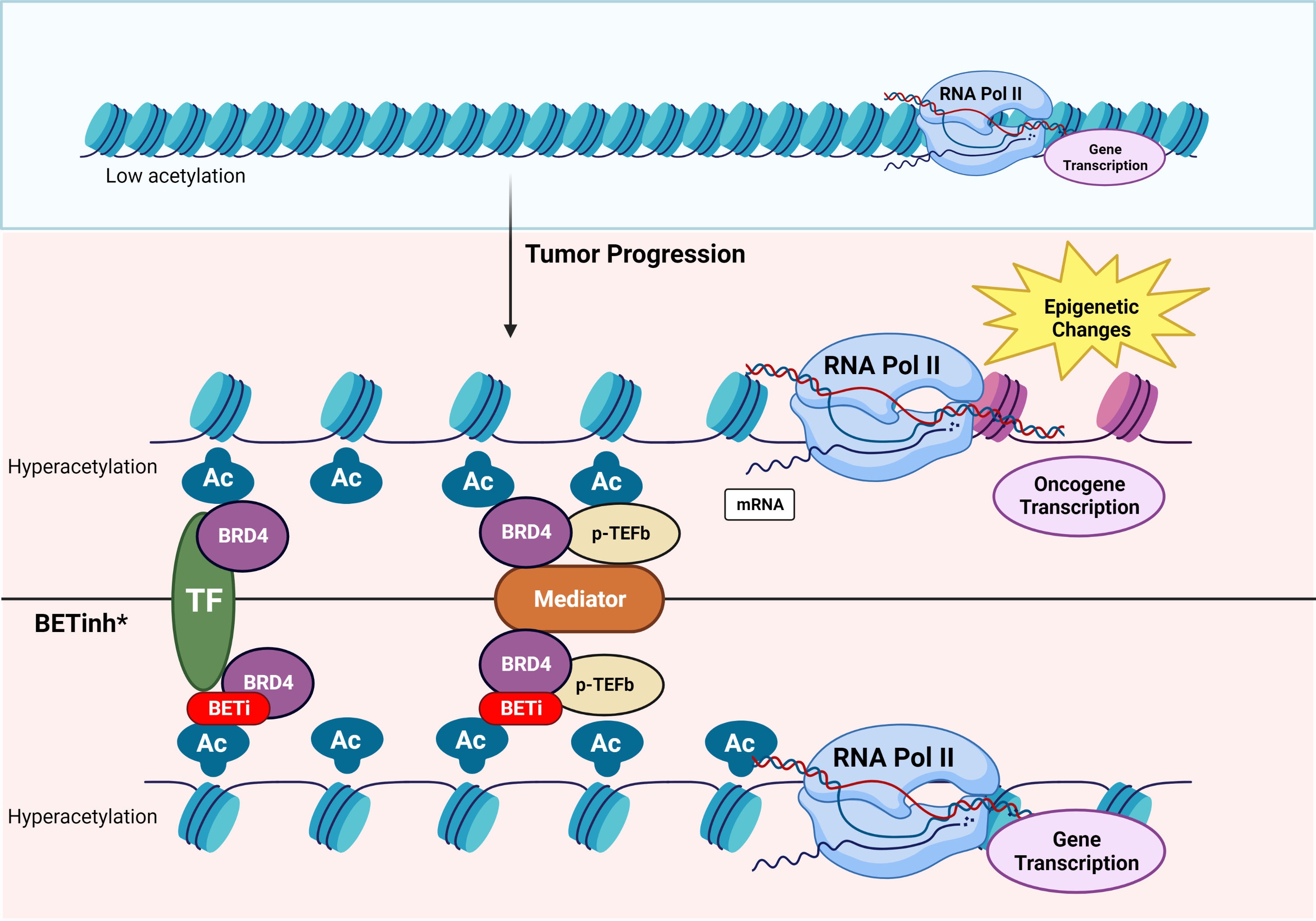
The bromodomain and extra-terminal domain (BET) proteins are epigenetic readers that recognize and bind acetylated lysine residues involved in cancer proliferation and metastases 1. Several molecules targeting this family of proteins have been tested in early phase clinical trials, although no one has been approved yet. Limitations on the development include their narrow therapeutic index with limited target engagement compromising the efficacy in patients 2.
Trotabresib (CC-90010, BMS-986378) is a potent, orally available, and reversible dose-dependent BET inhibitor targeting BRD2, BRD3, BRD4 and BRDT, with higher affinity towards BRD4, with a mean IC50± value of 15.0nM ± 6.6nM. In preclinical studies demonstrated a significant antiproliferative activity, particularly in hematological tumors with IC50± ranging from 0.02 ± 0.006 μM in acute myeloid leukemia (AML) and 0.10 ± 0.31 μM in diffuse large B-cell lymphoma (DLBCL) as well as glioblastoma cells (IC50± 0.98 ± 1.06 μM). Cytotoxic and antiproliferative activity was further described in patient-derived xenograft models of different solid tumors including glioblastoma as monotherapy (with an antiproliferative activity IC50 ranging from 34 nM to 1608 nM), and in combination with temozolomide (IC50 ranging from 26 nM to 2828 nM), confirming modulation of MYC, FOSL1 and GLI expression as pharmacodynamic markers, highly consistent with potential blood-brain-barrier penetration3.
These promising preclinical results on potency and improved pharmacological properties of the drug led to the first in human study. We had prior experience with other BET inhibitors 4 and trotabresib has the potential to achieve robust target engagement and thus significant antitumor activity although on the other side, potentially would induce profound hematological toxicity, especially in heavily pretreated patients. This would prove a challenge in the clinical development of the molecule.
Trotabresib dose escalation clinical trial
We recently reported the results of the phase 1 dose escalation and expansion study of trotabresib 3,5(which included 69 patients with different cancers, including DLBCL and central nervous system (CNS) progressing tumors). The toxicity profile (considering 56 patients evaluable for dose limiting toxicities) was mainly characterized, as expected, by thrombocytopenia, elevated transaminases, fatigue and gastrointestinal symptoms such as nausea/vomiting, and diarrhea. Adverse events could be well managed with support medication and in some rare cases, dose adjustments. Interestingly, some patients presented arthralgias, erythematous skin rash, and minor signs of seronegative arthritis suggesting a potential immunomodulatory effect of the compound. This was supported by previous evidence that BET inhibitors could positively affect CD8+ T cells, directly or indirectly, through reduction of anti-programmed death ligand-1 (PDL1) expression 6–9. It was later shown that, in fact, BET inhibitors have the capacity to rescue T cell exhaustion by restoring proliferation and expansion of precursor T cells, with a synergistic effect when administered in combination with PD1 inhibitors on reduction of tumor cells in AML. A combination of anti-PD1 therapy with BET inhibition could have a role in overcoming the natural resistance of glioblastoma cells to conventional immune checkpoint inhibition. Trotabresib also showed early signs of antitumor activity, two patients had objective responses: one patient with a grade 2 diffuse astrocytoma achieved complete response, sustained after >19 cycles, and one patient with endometrial carcinoma achieved partial response. Interestingly, disease control was evident for over 20% of patients. One patient diagnosed with thymoma achieved 18% tumor reduction lasting over one year. Pharmacokinetic (PK) studies showed a dose-proportional increase in trotabresib plasma exposure and confirmed central nervous system presence with cerebrospinal fluid concentrations reaching the concentration for preclinical activity. This was later confirmed in a window of opportunity study. 5 . Pharmacodynamic activity was also shown through modulation of CCR1, particularly with the monthly dosing schedule (4 days on/24 days off), achieving the most profound CCR1 suppression. Longer follow-up for the expansion phases supported these findings, with long lasting clinical effect in five patients who received over 38 cycles of treatment, including one patient with a high-grade astrocytoma. The longer half-life of trotabresib compared to other BET inhibitors, and the interesting PK and clinical activity seen in CNS tumors, led to its further development in patients with glioblastoma, in combination with Temozolomide, also supported by preclinical studies. As further clinical data is generated, time will finally say if a BET inhibitor is available to increase our armamentarium of anticancer agents.
REFERENCES
- Taniguchi, Y. The Bromodomain and Extra-Terminal Domain (BET) Family: Functional Anatomy of BET Paralogous Proteins. Int J Mol Sci 17, 1849 (2016).
- Singh, M. B. & Sartor, G. C. BET bromodomains as novel epigenetic targets for brain health and disease. Neuropharmacology 181, 108306 (2020).
- Moreno, V. et al. Phase I study of CC-90010, a reversible, oral BET inhibitor in patients with advanced solid tumors and relapsed/refractory non-Hodgkin’s lymphoma. Annals of Oncology 31, 780–788 (2020).
- Shorstova, T., Foulkes, W. D. & Witcher, M. Achieving clinical success with BET inhibitors as anti-cancer agents. Br J Cancer 124, 1478–1490 (2021).
- Moreno, V. et al. BET inhibitor trotabresib in heavily pretreated patients with solid tumors and diffuse large B-cell lymphomas. Nat Commun 14, (2023).
- Zhu, H. et al. BET Bromodomain Inhibition Promotes Anti-tumor Immunity by Suppressing PD-L1 Expression. Cell Rep 16, 2829–2837 (2016).
- Hogg, S. J. et al. BET-Bromodomain Inhibitors Engage the Host Immune System and Regulate Expression of the Immune Checkpoint Ligand PD-L1. Cell Rep 18, 2162–2174 (2017).
- Wang, N., Wu, R., Tang, D. & Kang, R. The BET family in immunity and disease. Signal Transduct Target Ther 6, 23 (2021).
- Kagoya, Y. et al. BET bromodomain inhibition enhances T cell persistence and function in adoptive immunotherapy models. Journal of Clinical Investigation 126, 3479–3494 (2016).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in