Technology in the treatment of type 1 diabetes
Published in Biomedical Research
In the last decade, there have been significant advances in how technology is used by people living with type 1 diabetes (T1D). The primary aim of these technologies is to help achieve target blood sugar levels. As technology has become more automated, there is the potential to reduce the burden of living with this chronic medical condition. To explore this, a systematic literature review was conducted on papers describing the use of diabetes technologies, including continuous glucose monitors , insulin pumps, sensor-augmented pump therapy, low glucose suspend, predictive low glucose suspend, and automated insulin delivery systems.
Continuous glucose monitors have largely become standard of care in T1D management, as they provide glucose readings every 1-5 minutes or at least 288 data points a day as compared to intermittent fingerstick checks where most living with diabetes strive to do 4 checks per day. While checking blood sugar levels is important, how insulin is delivered has also been impacted by technological advances. Insulin pumps were first introduced in the late 1970s and provide a more physiologic methods of insulin delivery. Bridging glucose sensing and insulin delivery, a sensor augmented pump allows the glucose sensor to “talk” to the pump; thereby displaying the glucose reading on the pump screen. Low glucose suspend and predictive low glucose suspend systems go a step further; these systems have either an algorithm which pauses insulin delivery when a low glucose threshold (e.g., 70 mg/dL) is breeched or shuts off insulin delivery when a low blood sugar is predicted to occur soon (e.g., if predicted to be <80mg/dL in 30 minutes time). Automated insulin delivery, also known as closed-loop control or artificial pancreas, not only pauses delivery but can also increase insulin delivery in tiny amounts; however, these systems are not completely autonomous as users need to enter carbohydrates or announce meals prior to eating.
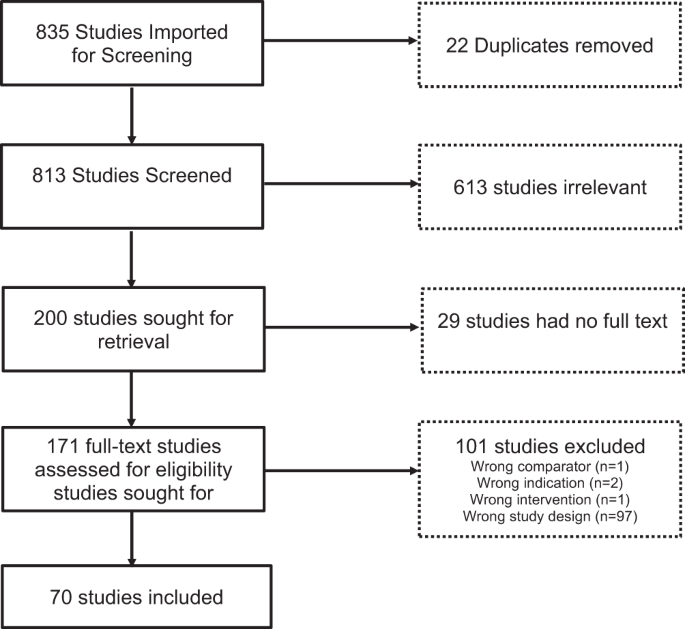
In order to evaluate the impact of technologies on T1D treatment over the past decade, we implemented a search strategy using PubMed and EMBASE with terms specific to T1D technology devices like continuous glucose monitors, insulin pumps with low glucose suspend and predictive low glucose suspend features, and automated insulin delivery systems. 813 studies were imported into the review system, Covidence, and screened for eligibility. Screening occurred in duplicate, with disagreements resolved by consensus by a third reviewer. Of the 813 studies, 200 full-text studies were retrieved, and based on a priori specifications 70 studies met eligibility criteria to be included in the final analysis. Inclusion criteria included papers published after Jan 1, 2012, in English, with sample sizes greater than 50 participants with
We find that several of these novel technologies helped improve important diabetes-related measures, including lowered hemoglobin A1c (HbA1c), which provides a 3-month assessment of average glucose levels with consensus guidelines recommending a target of <7%, and reduced hypoglycemia. Recognizing that people with diabetes may come to technology from different starting points, understanding the benefits derived from a broad range of user characteristics will allow clinicians to use a more individualized approach to care. Baseline HbA1c subgroup analyses in most trials produced consistent results except in three studies. Children with HbA1c > 7.5% (vs. <7.5%) on continuous glucose monitors had improved HbA1c at 3 months. Adults with HbA1c > 7.5% (vs. <7.5%) on SAP had improved time in target range, a measure derived from continuous glucose monitoring data and indicating the percent of time spent between glucose levels of 70-180 mg/dL. Adolescents and adults with HbA1c < 8% (vs. >8%) on low glucose suspend had reduced nocturnal hypoglycemic events. The presence or absence of detectable C-peptide had no bearing on metabolic outcomes.
Several studies focused on adults with impaired awareness of hypoglycemia and demonstrated improvements in time in range, HbA1c, and rates of hypoglycemia with continuous glucose monitors. In addition, continuous glucose monitor trials often performed subgroup analyses based on insulin delivery modality used by participants (multiple daily injections vs. pump), yet this did not affect the results. Unsurprisingly, higher wear time of continuous glucose monitor and automated insulin delivery systems were often tied to increased efficacy.
Importantly, studies showed a positive impact of technologies on quality of life in people living with diabetes through questionnaires and interviews. Ease of use and benefit were measured, and if technology is effective but overly burdensome persistence of device use can be impacted. Studies that were reviewed generally demonstrated that diabetes devices have a positive impact on these patient-reported outcomes. The goal is to utilize these measures to aid in the future application of a precision medicine approach when prescribing diabetes technology.
Evaluation of advanced diabetes technologies across different subgroups is essential given the diverse population living with the condition. By examining factors like glycemia at enrollment, duration of diabetes, body mass index, daily insulin requirements, previous use of diabetes devices, education, comorbidities, and inclusion of racial/ethnic minorities, will allow for more informed decision making about how to best pair glucose monitoring and insulin delivery modality to the needs of an individual. Despite the studies in this analysis including people with T1D from 2 to over 70 years of age, and most studies including nearly equal male and female participants, the need for real-world data collection and the use of registry data that includes more diverse populations remains. Real-world evidence from heterogeneous groups outside of the focus of pivotal randomized controlled trials may provide critical information regarding the frequency of device errors/malfunctions or clinically relevant outcomes, like frequency of skin irritations from device adhesives noted with long-term routine use of devices. Indeed, it has been well established that disparities exist regarding access to diabetes technologies for those of minority groups; how we overcome these disparities is of critical importance.
Our findings highlight the benefits of novel technologies in the treatment of T1D and identify areas for further research to optimize and personalize diabetes care. While there is evidence of benefit, there also remains a substantial need to ensure health equity in the use of advanced diabetes technologies for glycemic and personal benefits across the population to reduce the existing disparities. The use of advanced diabetes technologies remains an ongoing challenge in low and middle-income countries and even for those who are uninsured or underinsured. Despite these many challenges, diabetes technology has revolutionized T1D care with the ability to attain more targeted glycemia, reduce hypoglycemia, and preserve well-being without additional burdens for people living with T1D and their families. There is more work to do to understand and implement a precision medicine model, but progress is underway.
Follow the Topic
-
Communications Medicine

A selective open access journal from Nature Portfolio publishing high-quality research, reviews and commentary across all clinical, translational, and public health research fields.
Related Collections
With Collections, you can get published faster and increase your visibility.
Reproductive Health
Publishing Model: Hybrid
Deadline: Mar 30, 2026
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in