The evolving clone
Published in Ecology & Evolution

Clones—large asexual populations of plants or animals producing offspring through branching, budding or fragmentation—should rather not exist. The emerging modules are supposed to be genetically identical replicates. Hence, their very existence is hard to reconcile with concepts of genetic diversity as prerequisite for stress tolerance and resilience. How is it possible that clones are the foundation of some of the most stable and productive ecosystems such as coral reefs or seagrass meadows?

These questions intrigue me since a quarter of a century. In eelgrass Zostera marina, the most widespread seagrass species throughout the northern hemisphere, clones are stretching for thousands of square meters (Fig. 1). In the Baltic Sea, these were discovered in the late 1990s (Reusch et al. 1999) with the advent of the first microsatellite markers that allowed for an unambiguous assignment of ramets (the independent modules) to genets (the "clone" that arose from a single recombination event). When I showed the autoradiograph of the invariant banding patterns to my then postdoctoral mentor, Jeanine Olsen at the University of Groningen-Netherlands, where I had developed the required microsatellites, she was as struck as myself. All plants at a single site of Åland islands archipelago were composed of the same multi-locus genotype, thus is not a population, but a single "individual". Subsequent work along with long-term colleague, Christoffer Boström at Åbo Akademi University (Finland) showed that extreme clonality is widespread (Reusch & Boström 2011). Shortly thereafter, similar surveys by others of additional seagrass species (notably the Mediterranean Posidonia oceanica) revealed millennial-old clones covering tens of kilometers (Arnaud-Haond et al. 2012).
The next critical discovery occurred while genotyping large clones. We found strange allelic patterns with three or four fragments per microsatellite locus, where maximally two alleles were expected in our diploid plants. At first, we dismissed the results as sample spill-over and excluded those genotypes, but later confirmation revealed that we were looking at genetic mosaics, the mixture of cell lineages having two or more microsatellite genotypes, owing to somatic mutations during mitosis (Reusch & Boström 2011).
Fast forward another eight years, and the time was finally ripe for a more systematic survey of the phenomenon of somatic genetic variation. Since 2016, we not only have a high-quality genome of eelgrass at hand (Olsen et al. 2016), but also the required sequencing power to re-sequence a sample of putative clone members to sufficient depth (>1000x) so as to detect low frequency variants. Fortunately, we were able to recruit PhD student and skillful molecular ecologist, Lei Yu (funded by the Chinese Scholarship Council CSC), who was able to carry out the indispensable, large-scale and non-standard analyses of variant calling required to control against false positive hits.
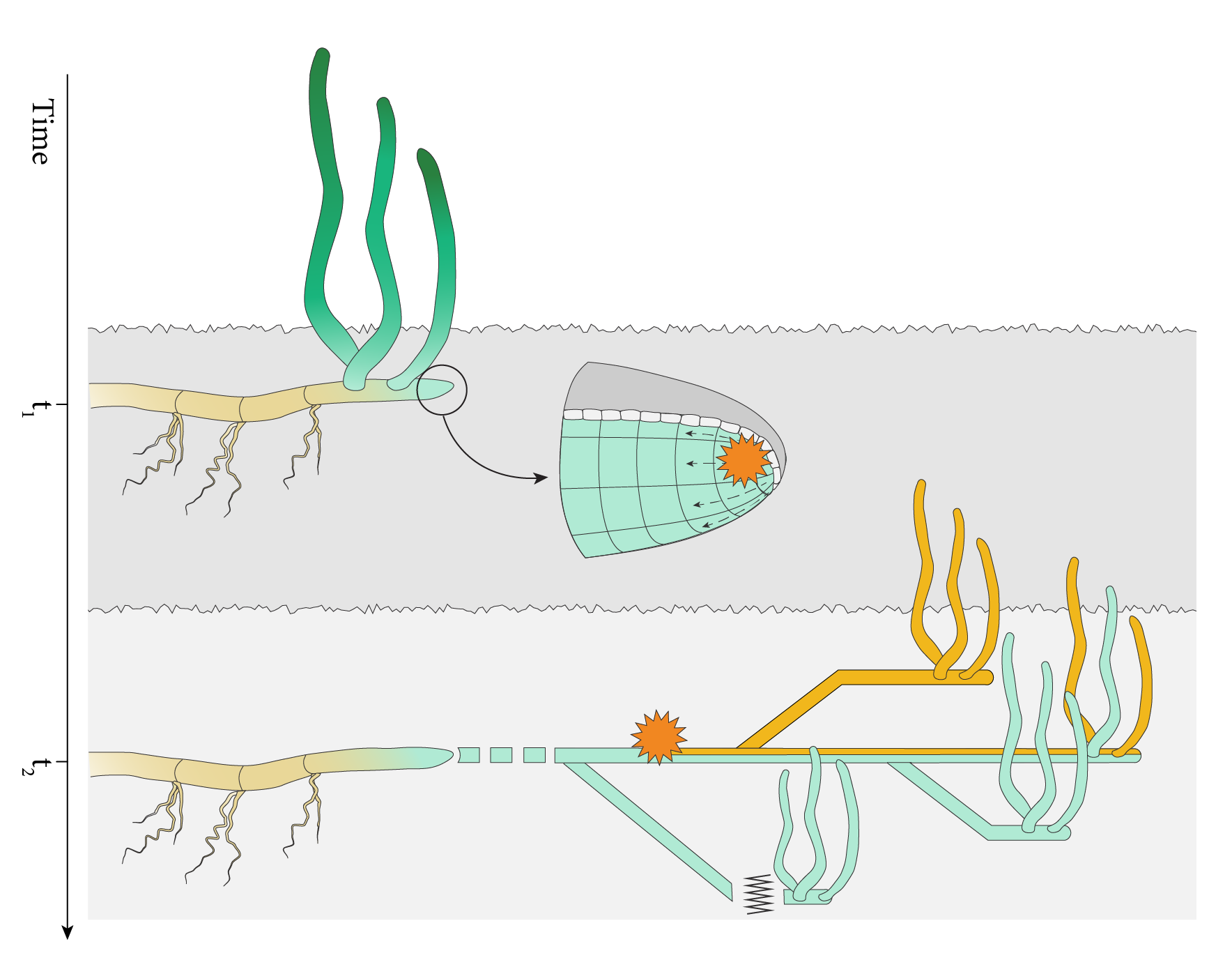
The amount of somatic genetic variation that we detected was unexpectedly high (Yu et al. 2020), with hundreds of genes affected by polymorphisms at non-synonymous positions, thus demonstrating their potential to contribute to adaptation and thus, improved resilience to environmental stress. As this is the first investigation, we focused on neutral genetic processes and found that the branching process during module formation constitutes a genetic bottleneck that can fix particular somatic variants in different modules. This process of somatic genetic drift is key to partition somatic genetic variation into differentiated modules so that they can be subject to selection (Fig. 2). It possibly applies to many clonally growing species, including algae, corals and fungi. Because any selected genetic variants will drag along neutral variation via genetic hitchhiking, it will be challenging to identify the causal gene variants responsible for such selective sweeps.
Ultimately, we have learned that clones are not perfectly, genetically uniform. And this, in turn, may partially solve the riddle why genetically homogenous clones exist for thousands of years - precisely because they may not be genetically homogeneous after all.
References
Arnaud-Haond, S. et al. Implications of Extreme Life Span in Clonal Organisms: Millenary Clones in Meadows of the Threatened Seagrass Posidonia oceanica. PLOS One 7, e30454 (2012).
Olsen, J. L. et al. The genome of the seagrass Zostera marina reveals angiosperm adaptation to the sea. Nature 530, 331–335 (2016).
Reusch, T. B. H., Boström, C., Stam, W. T. & Olsen, J. L. An ancient eelgrass clone in the Baltic Sea. Marine Ecology Progress Series 183, 301-304 (1999).
Reusch, T. B. H. & Boström, C. Widespread genetic mosaicism in the marine angiosperm Zostera marina is correlated with clonal reproduction. Evol Ecol 25, 899-913 (2011).
Yu, L., C. Boström, S. Franzenburg, T. Bayer, T. Dagan, T. B. H. Reusch (2020): Somatic genetic drift and multilevel selection in a clonal seagrass. Nature Ecology & Evolution, doi.org/10.1038/s41559...
Follow the Topic
-
Nature Ecology & Evolution
This journal is interested in the full spectrum of ecological and evolutionary biology, encompassing approaches at the molecular, organismal, population, community and ecosystem levels, as well as relevant parts of the social sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Biodiversity and ecosystem functioning of global peatlands
Publishing Model: Hybrid
Deadline: Jul 27, 2026
Understanding species redistributions under global climate change
Publishing Model: Hybrid
Deadline: Jun 30, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in