The Pristine Myth: We Still Don’t Know What Most Nanoparticles Become in Real Water
Published in Earth & Environment, Ecology & Evolution, and Materials

The Particle You Release Is Not the Particle the Environment Receives
Nanotoxicology has historically relied on a deceptively simple assumption: if we know an engineered particle’s size, morphology, surface charge, and composition, we can accurately predict its environmental behavior.
But environmental systems do not preserve engineered perfection.
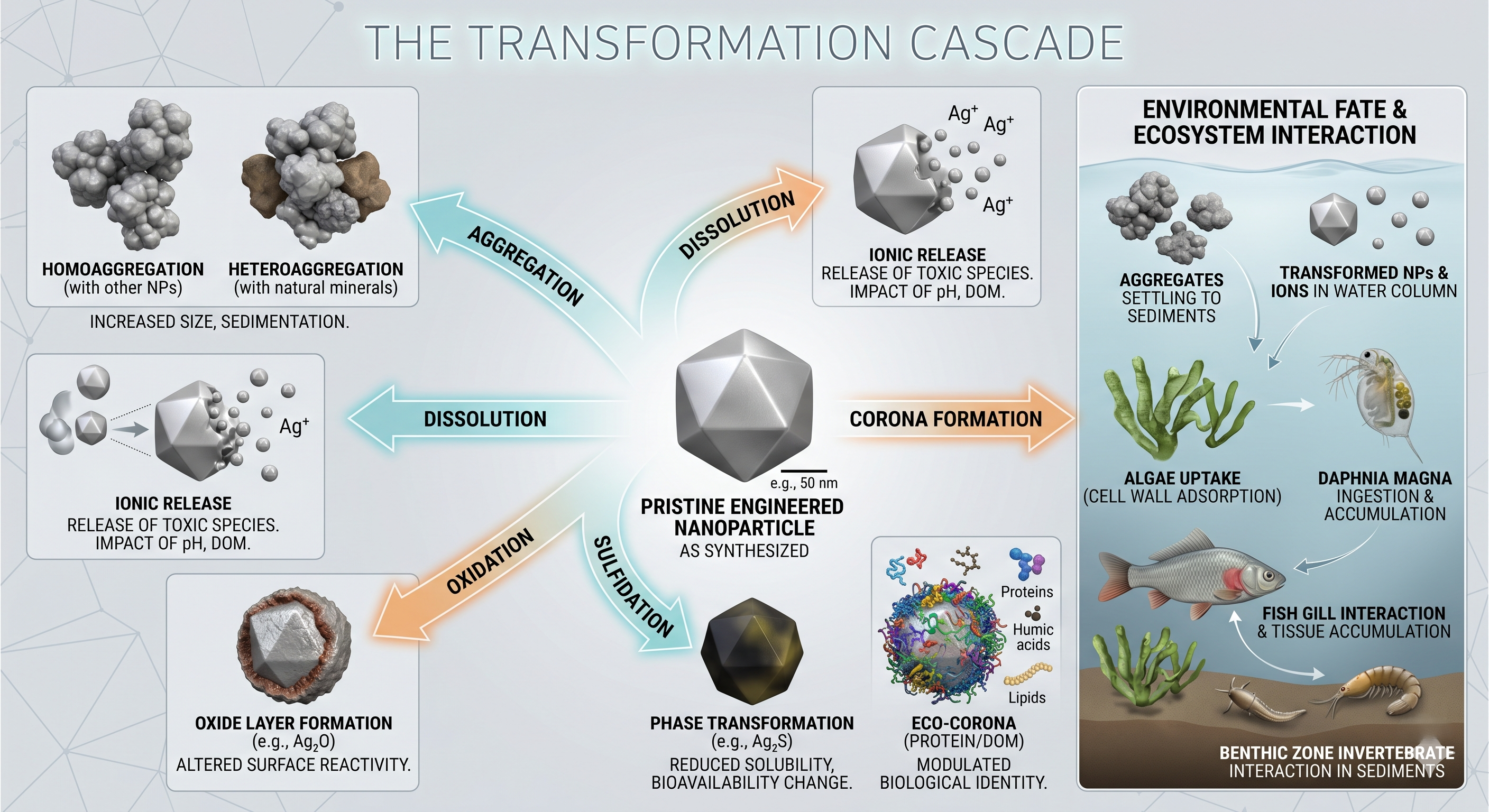
The moment a nanoparticle enters a river, groundwater aquifer, or wastewater effluent, it begins transforming. A silver nanoparticle released into wastewater rapidly interacts with sulfides, chlorides, and dissolved organic matter. Copper oxide nanoparticles dissolve into ionic species. Zero-valent iron oxidizes into complex iron oxyhydroxide phases. Titanium dioxide aggregates onto colloidal sediments.
Within seconds, organic matter adsorbs onto the surface. Within minutes, oxidation and dissolution pathways reshape the crystal interface. Within hours, the material no longer resembles the engineered nanoparticle originally synthesized in the laboratory. The environmental identity of the material becomes fundamentally divorced from its manufactured identity.
Recent reviews across environmental nanoscience have reached a strikingly consistent conclusion: transformation processes alter nanoparticle behavior so profoundly that pristine-particle toxicity data often fail to predict real-world ecological outcomes. This is not a minor methodological issue; it is a foundational flaw in environmental risk assessment.
The Eco-Corona: Nature’s Invisible Rewrite Layer
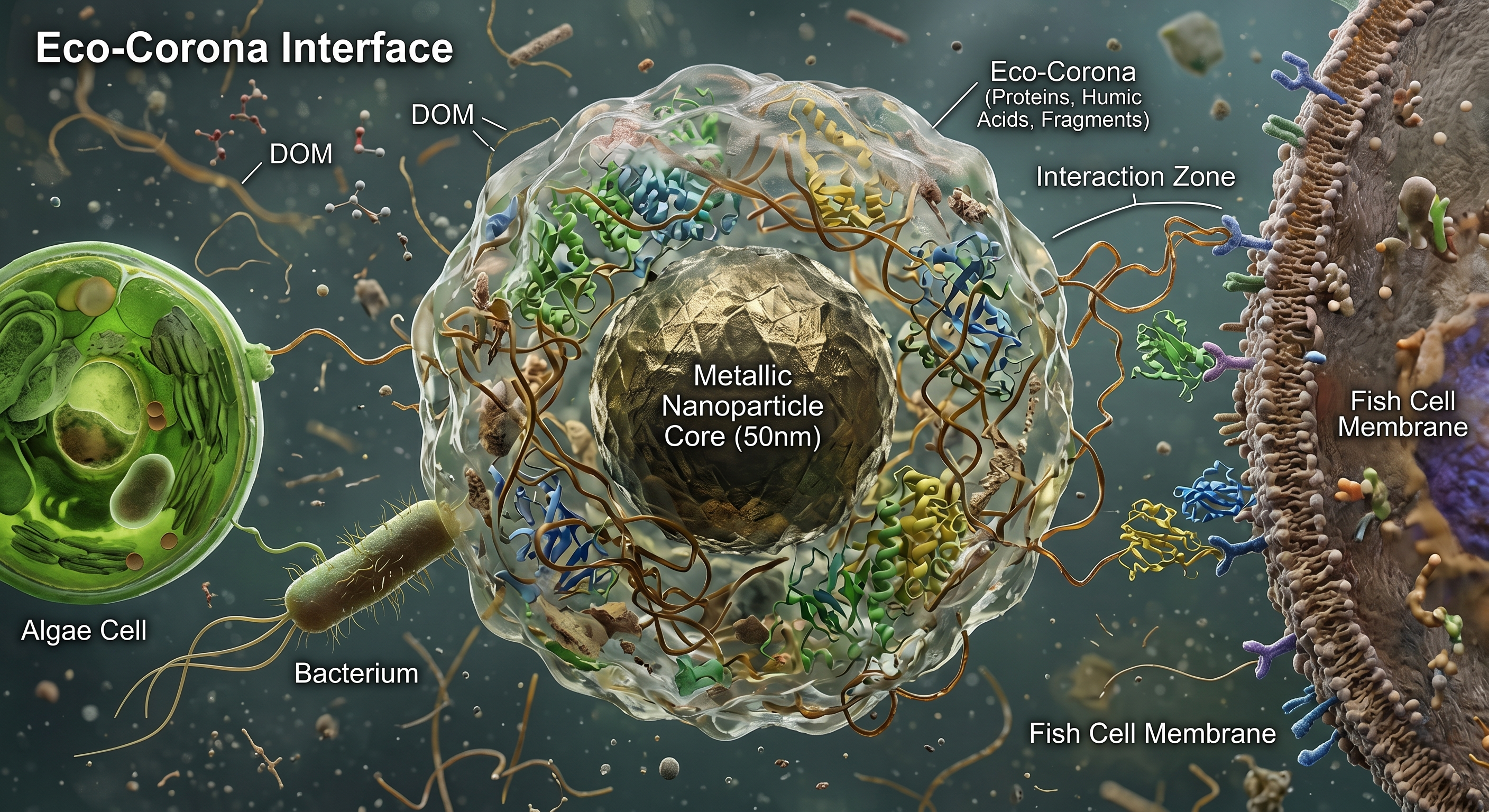
Perhaps the most important transformation is also the hardest to see. When nanoparticles enter environmental media, they acquire an eco-corona—a dynamic shell of adsorbed biomolecules, including humic acids, proteins, polysaccharides, and microbial metabolites.
This layer is not passive contamination. It fundamentally rewrites the particle’s environmental identity. The eco-corona controls colloidal stability, aggregation behavior, cellular recognition, dissolution kinetics, and biological uptake. In effect, nature immediately “re-engineers” our engineered material.
As recently framed in Accounts of Chemical Research, the eco-corona is a double-edged environmental interface. In some systems, it stabilizes nanoparticles, increasing long-range transport through aquatic ecosystems. In others, it passivates highly reactive surfaces, suppressing acute toxicity. The outcome depends on a dizzying matrix of water chemistry, ionic strength, and microbial ecology.
Critically, these concepts are now expanding beyond conventional metallic nanoparticles into emerging porous systems like MOFs and hybrid nanocomposites. For the porous materials community, this poses a defining question: What does a MOF actually become after six months in a real aquatic system? We still do not fully know.
The Evidence Gap: What We Test vs. What Actually Exists
One of the most uncomfortable realities in environmental nanoscience is that the majority of ecotoxicology experiments are still performed under conditions specifically designed to exclude environmental complexity.
Many studies continue exposing model organisms to pristine nanoparticles dispersed in ultrapure water, simple electrolytes, or highly simplified laboratory buffers. These systems deliberately avoid the very transformations that dominate real environmental behavior. As a result, we have created a widening gap between the particles used in hazard testing and the particles that actually exist in rivers, soils, and wastewater systems.
Several international nanosafety initiatives—including the MISTRA Environmental Nanosafety consortium—have already proposed standardized methods for studying eco-corona formation and environmentally aged nanomaterials. The protocols exist. The instrumentation exists. Yet, widespread adoption remains stubbornly slow.
The Regulatory Blind Spot
The science of nanoparticle transformation is advancing rapidly, but regulation is not. Current regulatory frameworks—including REACH, OECD nanosafety guidelines, and national hazard assessment protocols—still largely operate within a pristine-particle paradigm.
Critical transformation pathways like sulfidation, eco-corona formation, and redox aging are rarely integrated into mandatory hazard evaluation workflows. This creates a dangerous disconnect. We are making real-world regulatory decisions using environmentally unrealistic material states.
Conclusion: Expanding the Definition of Performance
This is not an anti-nanotechnology argument. Nanomaterials remain essential to water purification, catalysis, energy storage, and environmental remediation. But the materials community must expand its definition of performance.
A nanoparticle’s environmental afterlife is not a downstream regulatory inconvenience; it is a fundamental property of the material itself. The future of advanced materials design will increasingly depend on answering not just, "What can this nanoparticle do?" but also, "What does this nanoparticle become?"
Treating environmental transformation as a central design parameter—rather than a post-hoc complication—will fundamentally redefine how next-generation nanomaterials are designed, tested, and approved.
I open this question to the community: Should journals begin requiring environmentally aged particle data for nano-enabled water treatment studies? Or would that create regulatory barriers that slow innovation and translation?
#Nanotechnology #Nanomaterials #EnvironmentalScience #WaterTreatment #NanoSafety #Ecotoxicology #MOFs #AdvancedMaterials #EnvironmentalEngineering #ResearchCommunities




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in