Tumor-promoting immune cells retrained to fight most aggressive type of brain cancer
Published in Cancer
https://www.nature.com/articles/s41467-021-22885-8
GBM is the most aggressive primary brain tumor. Most glioblastoma patients survive < two years, despite surgery, and chemo-radiation. Cancer treatments that work by engaging the immune system to attack tumors have proven effective against many cancers, but not for glioblastoma.
An important feature of the immune system in clearing cancers is its ability to distinguish normal cells from cancerous (mutated) cells. For the immune system to attack cancer cells while leaving the normal cells intact, one requirement is the expression of immune “checkpoints” – molecules on certain immune cells that act as the “brakes” on an immune response. Cancer cells often hijack these checkpoints to stop an attack by the immune system. A class of drugs that removes these “brakes” – known as immune checkpoint blockers – are expected to “step on the gas pedal” of the immune system to help it fight against glioblastoma as effectively as in other cancers. However, these blockers have failed to improve survival in randomized clinical trials conducted to date in this disease 1-3.

Steele Laboratories for Tumor Biology
Our laboratory led by Dr. Rakesh K. Jain, has shown over the past 30 years that every element of the tumor microenvironment is abnormal. Such an abnormality and the brain's unique features make brain tumors the only primary tumors that still kill patients. We asked these questions in our research toward finding a cure for glioblastoma: (1) What are the cellular subtypes that render ICB therapy ineffective? (2) If we cannot efficiently deliver immune cells to GBM, can we reprogram the cells that contribute to immunotherapy resistance and are located inside GBM to kill cancer cells? (3) How can we target and reprogram these cells? and (4) Finally, would such a strategy cure GBM in mice, providing us hope to move toward a trial in patients?
We showed that tumor microenvironment abnormalities promote a suppressive environment for the immune system, which blocks tumor-fighting immune cells, mainly CD8 T cells, at the tumor border while allowing infiltration of tumor-promoting immune cells known as regulatory T cells (Tregs). Among elements of the tumor microenvironment, Tregs are untapped resources for efficient GBM immunotherapy. We have two main reasons to target Tregs. First, Tregs are abundantly present in GBMs, but classically anti-tumor CD8 T cells are excluded in GBM. Therefore, our idea of inducing Treg conversion to effector cells would generate large numbers of anti-tumor effector cells from existing GBM Tregs independent of new recruitment. Second, T-cell based therapy in GBM has been focused on enabling CD8 T cell function. Because CD8 T cells require a neo-antigen epitope to recognize tumor cells, their function can be reduced if tumor cells have low level of mutations as GBM. We exploited the inherent capacity of Tregs, i.e. their ability to recognize self-antigens and by reprogramming these cells we benefit from their specialized T cell receptor (TCR) to recognize self-antigens while changing their behavior from “recognize and protect” tumor cells to “recognize and kill” tumor cells.
Regulatory T cells in the brain can be reprogrammed from guarding glioblastoma tumors to attacking them from within. In our work, we exploited the preferential accumulation of Tregs in the tumor by therapeutically altering their function – a strategy known as “reprogramming” – to make them kill cancer cells instead of protecting them. We collaborated with Dr. Hye-Jung Kim, an immunologist at Dana-Farber Cancer Institute, toward finding an appropriate Treg reprogramming strategy tailored to GBM. Based on our genetic and proteomic profiling of GBM Tregs and systemic Tregs, we targeted glucocorticoid-induced TNFR-related receptor (GITR) that is preferentially expressed in GBM Tregs with an agonistic antibody (αGITR). Upon antibody-mediated engagement of GITR, Tregs converted to an anti-tumor T cell phenotype. Combination of αGITR and anti-programmed cell death 1 antibody (αPD1), the most frequently used checkpoint blocker in the clinic, resulted in a strong survival benefit in mice. Importantly, some of these mice not only rejected tumors but also developed a long-term immunity to GBM.
We think that the unique phenotype of GBM Tregs allows their selective targeting and reduces immune-related adverse events (irAEs). GBM infiltrating Tregs are different in their phenotype from peripheral Tregs. This unique feature allowed us to design tumor-specific immunotherapy targeting specifically GBM-infiltrating Tregs while avoiding systemic autoimmunity by interfering with systemic Tregs that maintain immune homeostasis. This approach is likely to alleviate irAEs which often accompany systemic Treg depletion strategies in the clinic resulting in treatment discontinuation and therefore loss of therapeutic benefit.
We also asked if Treg reprogramming can enhance GBM response to the standard of care (SoC). To obtain clinically relevant information, we also need an animal model mimicking SoC in the clinic. We established a SoC regimen for orthotopic mouse GBM consisting of surgery, radiation and chemotherapy. We found that indeed our targeted strategy tailored to the GBM specific TME converts it to anti-tumor immune-TME and thus synergizes with the SoC.
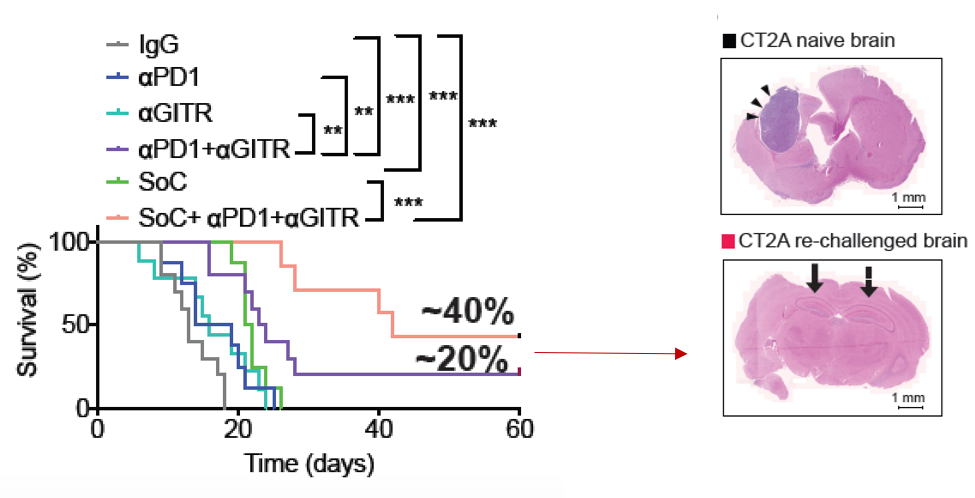
Our study is both mechanistic and translational. We revealed novel cellular and molecular mechanisms including the switch in the function of Tregs to produce IFNγ and turn on MHCs in tumor cells, enabling the recognition of tumor cells and tumor cell eradication. Because these cellular and phenotypical changes govern immunotherapy, decoding them leads to treatment strategies tailored to the patients’ specific immune profile (e.g., the response in Treg versus CD8 T cells enriched Tumor microenvironment). This novel combination of ICB therapy and Treg reprogramming represents a paradigm shift in understanding, managing and eventually curing GBM. Our work offers a possible solution to overcome GBM resistance to immunotherapy, and can be translated to patients. Our data can also be used for patient stratification. Because we are using αGITR antibodies that are currently being evaluated in a number of clinical trials, our study provides mechanistic insights and compelling preclinical data in support of testing the combination of αGITR with αPD1/PDL1 – with or without the standard of care therapy – in GBM patients with high intratumoral accumulation of Treg cells.
References:
1 Lim, M., Xia, Y., Bettegowda, C. & Weller, M. Current state of immunotherapy for glioblastoma. Nat Rev Clin Oncol 15, 422-442, doi:10.1038/s41571-018-0003-5 (2018).
2 Reardon, D. A. et al. Effect of Nivolumab vs Bevacizumab in Patients With Recurrent Glioblastoma: The CheckMate 143 Phase 3 Randomized Clinical Trial. JAMA Oncol, doi:10.1001/jamaoncol.2020.1024 (2020).
3 Nayak, L. et al. Randomized phase II and biomarker study of pembrolizumab plus bevacizumab versus pembrolizumab alone for recurrent glioblastoma patients. Clinical Cancer Research (2020).
4 Reardon, D. A. et al. Glioblastoma Eradication Following Immune Checkpoint Blockade in an Orthotopic, Immunocompetent Model. Cancer Immunol Res 4, 124-135, doi:10.1158/2326-6066.CIR-15-0151 (2016).
5 Saha, D., Martuza, R. L. & Rabkin, S. D. Macrophage Polarization Contributes to Glioblastoma Eradication by Combination Immunovirotherapy and Immune Checkpoint Blockade. Cancer cell 32, 253-267 e255, doi:10.1016/j.ccell.2017.07.006 (2017).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in