Unlocking the Genetics of GLP-1 Medications Through Crowdsourced Science at 23andMe
Published in Genetics & Genomics, General & Internal Medicine, and Pharmacy & Pharmacology
The Mystery of GLP-1 Variability
The development of GLP-1 receptor agonists like semaglutide and tirzepatide has transformed the clinical management of obesity. But as these medications have surged in popularity, a clear mystery emerged in the data: there is large variability in how patients respond. While some individuals experience life-changing weight loss of up to 20%, others lose as little as 5% or even gain weight [1, 2]. Similarly, side effects are unpredictable, with some users experiencing various adverse events, while others feel completely fine[3].
At the 23andMe Research Institute, we suspected genetics played a role in this variability, just as it does in other medication responses. In order to investigate, we needed to gather data to build a large cohort of individuals actively taking these drugs. So, Institute scientists conceived of a new study to investigate 23andMe research participant experiences on these treatments, aiming to advance personalized medicine while making any resulting insights available to the community.
A Unique Model: The Research Flywheel
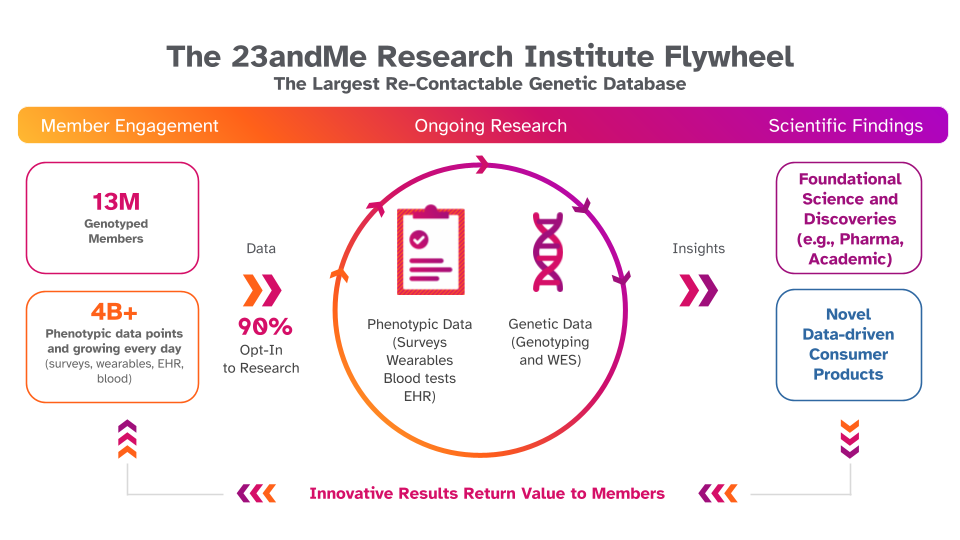
Traditional genomic studies can take years to recruit a few hundred participants. At the 23andMe Research Institute, we are proud to have built a model we call the "research flywheel." The flywheel is a cycle of research engagement and creation of novel genetic reports. 23andMe+ members receive insights into how their genetics impact many aspects of their lives, such as the likelihood of developing serious conditions, like coronary artery disease. Not only do we provide these insights, but millions of our members have consented to participate in research overseen by an institutional review board (IRB), allowing us to deploy surveys and to receive thousands of responses in a matter of days. Through this engagement, we are able to perform additional research and generate new genetic insights that are made available to our members; and it is this feedback loop that we refer to as the research flywheel. Using this process, 23andMe scientists and research collaborators have published over 300 studies and white papers spanning many topics in health and population genetics, with the average participant contributing to over 200 research papers. These scientific findings, primarily polygenic risk scores, are then translated into over 50 member reports, completing the cycle by delivering personalized insights back to the community and fueling the next turn of the research flywheel.
For this study, we launched the GLP-1 survey in the late summer of 2024, asking participants about their experiences with these medications, including drug brand, dosage, efficacy, and side effects. Given that some patients in the U.S. obtain access to GLP1 medications through telemedicine services rather than through traditional healthcare providers, our survey-based approach included data that would not be part of a large medical provider’s database. In this way, we were uniquely able to gather data from over 27,000 consented participants and to conduct a large-scale genetic study just one year after starting to collect data.
The "Aha!" Moment: Breakthrough at the GLP-1 Receptor
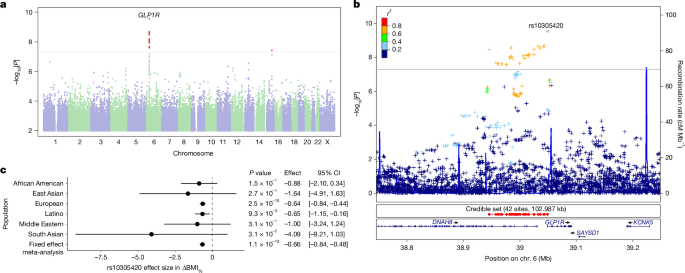
Few moments in genomic research match the thrill of finding a genetic signal that can plausibly explain differences in medication response. To our delight, our findings mapped specifically to the gene responsible for the cell-surface receptor targeted by GLP-1 drugs. The variant, rs10305420, is a missense variant within the gene itself, meaning that it changes the protein encoded by the gene. This finding suggests that some of the clinical variation in efficacy and side effects seen in GLP-1 patients stems from a fundamental change in the receptor’s own biology rather than other complex factors that can affect drug response indirectly.
To validate our findings, we set out to replicate them in the real-world data of the All of Us Research Program. On All of Us Research Workbench, we assembled each of our discovery model's covariates. The moment of truth arrived: a statistically significant association with the identified single nucleotide polymorphism (SNP), pointing in the same direction. This electronic health record (EHR) based validation bridged the gap between patient-reported data and formal clinical records, confirming that we had indeed pinned down the genetic signals for GLP-1 efficacy and nausea likelihoods.
The GIPR Discovery
Our research provides a great example of how collaborative peer review can improve research findings. A reviewer suggested we perform a GWAS for each medication class separately to avoid masking signals, given that tirzepatide targets both GLP-1 and GIP receptors. Re-running the analysis within drug-specific datasets revealed a second hit in the GIPR gene. This variant was associated with nausea and vomiting specifically in tirzepatide users, and was entirely absent in the semaglutide (pure GLP-1 receptor agonist) cohort. This suggests that while GLP1R it isn't the only driver when it comes to understanding the gastrointestinal side effect of dual-agonist therapies.
Integrating into Clinical Care
The final, and perhaps most important, piece of our research flywheel is providing an opportunity to return insights back to the broader community. We have published our research in an open-access format so that anyone can go and read the paper directly. However, in addition to advancing the scientific literature, we also aim to translate this research in a manner that can directly improve the health of the individuals we serve.
Using the data gathered from this study, we have translated our scientific findings into a new interactive report, available through the physician-supervised 23andMe+ Total Health™ service. By combining the two newly discovered genetic variants with non-genetic factors like age, sex, and medical history (such as type 2 diabetes status), individuals can receive personalized estimates for their likelihood of weight loss and side effects.
Discoveries like this simply would not exist without our research participants. Their willingness to share their experiences is paving the way for a new era of precision medicine in obesity treatment.
References
1. Wilding, J. P. H. et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. New Engl J Med 384, 989–1002 (2021). https://pubmed.ncbi.nlm.nih.gov/33567185/
2. Ryan, D. H. et al. Long-term weight loss effects of semaglutide in obesity without diabetes in the SELECT trial. Nat Med 30, 2049–2057 (2024). https://pubmed.ncbi.nlm.nih.gov/38740993/
3. Joy, Bethany, et al. Exploring the rates of Gastrointestinal adverse effects among five GLP-1 receptor agonists: A systematic review and Meta-Analysis of randomized controlled trials. Endocrine 91(1), 23 (2026). https://pubmed.ncbi.nlm.nih.gov/41489846/
Follow the Topic
-
Nature
A weekly international journal publishing the finest peer-reviewed research in all fields of science and technology on the basis of its originality, importance, interdisciplinary interest, timeliness, accessibility, elegance and surprising conclusions.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in