Why Recovery, Not Stress, May Be the Missing Biology of Resilience
Published in Social Sciences, Neuroscience, and Cell & Molecular Biology
Stress is inevitable. Recovery is not !
This simple idea became the starting point for our paper, “Stress Is Inevitable; Recovery Is Conditional: Bioenergetic Limits of Resilience in Aging and Disease.” For many years, research on stress, aging, chronic disease, nutrition, and mental health has described the consequences of persistent biological challenge. We know a great deal about allostatic load, inflammation, mitochondrial dysfunction, nutrient sensing, frailty, metabolic disease, and functional decline. Yet one question continued to trouble us: why do some individuals recover from repeated stressors, while others gradually lose resilience even when no single disease process fully explains their symptoms?
The more we examined this question, the more it seemed that the missing piece was not simply stress exposure itself. It was the biological cost of completing recovery.
From stress response to recovery capacity
Much of modern stress biology focuses on activation: cortisol, sympathetic tone, immune signaling, metabolic mobilization, and adaptive behavioral changes. These are essential responses. The body must respond to challenge, mobilize energy, defend itself, and adapt.
But activation is only half of the cycle.
After stress, the organism must repair, rebuild, restore redox balance, replenish substrates, resolve inflammation, remodel tissue, and return physiological systems toward a functional baseline. These processes are not passive. They require ATP, mitochondrial processing capacity, time, and adequate nutritional and metabolic support.
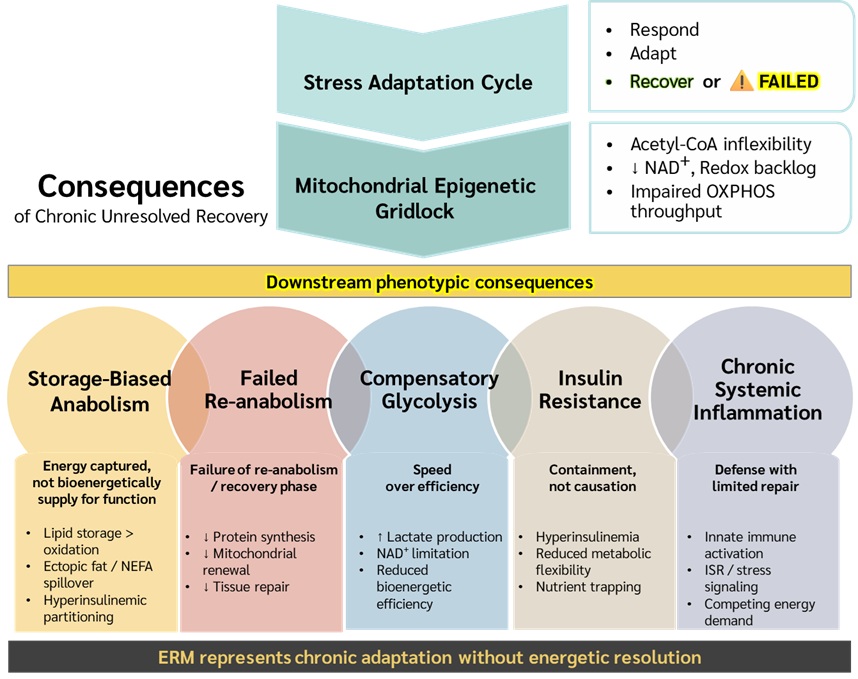
This led us to reframe stress adaptation as a three-part cycle: respond, adapt, recover. The first two phases have been studied extensively. The third phase—recovery as an active, bioenergetically funded process—has received less explicit attention.
In our view, this matters because a person may still be capable of mounting a stress response while no longer having enough bioenergetic reserve to complete recovery. This creates a subtle but clinically important asymmetry: stress adaptation can continue, while recovery becomes progressively incomplete.
The origin of the ERM concept
The idea of Exposure-Related Malnutrition (ERM) emerged from this clinical and conceptual gap. Classical malnutrition is usually associated with inadequate intake, deficiency, wasting, or overt changes in body composition. Yet in practice, many patients present with fatigue, poor recovery, low stress tolerance, reduced functional reserve, metabolic inflexibility, or subtle biomarker patterns despite apparently adequate intake and laboratory values that may remain within conventional reference ranges.
ERM is not proposed as a replacement for frailty, sarcopenia, cachexia, metabolic syndrome, or classical malnutrition. Rather, we propose it as a cautious, hypothesis-generating framework for a different level of the problem: a state in which cumulative exposure burden and recovery demand exceed the body’s capacity to fund repair and restoration.
In this sense, ERM is not simply “not enough food.” It is a mismatch between biological demand and recovery capacity. It asks whether the body has enough usable energy, mitochondrial throughput, redox flexibility, substrates, and time to complete the work of recovery.
Mitochondria as the bottleneck
A central idea in the paper is that mitochondria are not merely energy producers. They are rate-limiting infrastructure for recovery.
When substrate inflow exceeds downstream oxidative processing capacity, the system may enter what we describe as mitochondrial congestion. Glucose, fatty acids, and amino acids may remain available, but their effective use becomes constrained by the capacity of the TCA cycle, electron transport chain, NAD⁺ regeneration, and oxidative phosphorylation. Under these conditions, ATP production may appear preserved at a basal level, while reserve capacity and recovery flexibility decline.
This distinction is important. A cell or organism may maintain enough ATP for short-term survival while losing the surplus capacity required for repair, remodeling, and resilience. In the paper, we interpret this as a shift from efficient recovery toward compensatory maintenance.
If this state persists, mitochondrial congestion may become reinforced by redox backlog, altered acetyl-CoA handling, NAD⁺ depletion, impaired sirtuin activity, lipid storage, and epigenetic lock-in. We refer to this as mitochondrial epigenetic gridlock—a state in which the system remains viable but less flexible, less efficient, and less able to re-enter a fully restorative program.
Why this matters for aging and chronic disease
Aging and chronic disease are often studied as outcomes of damage accumulation, inflammation, metabolic dysregulation, or loss of homeodynamic capacity. These are all important. Our contribution is to suggest that many of these downstream features may also be understood as consequences of repeated, incomplete recovery.
From this perspective, central adiposity, insulin resistance, chronic low-grade inflammation, anabolic resistance, cognitive symptoms, fatigue, pain, and reduced physical reserve may not be independent phenomena. They may represent coordinated adaptations to persistent bioenergetic constraint. Initially, these adaptations may protect survival. Over time, however, they can undermine recovery, plasticity, and long-term function.
This framing also helps explain why many clinical signals of vulnerability appear before overt pathology. A patient may not yet meet criteria for frailty, sarcopenia, diabetes, cachexia, or inflammatory disease, but may already show delayed recovery, reduced reserve, poor anabolic responsiveness, or subtle multidomain biomarker shifts. These early signals may be easy to dismiss when viewed one at a time. When viewed as a pattern, they may reveal a system struggling to fund recovery.
A shift in clinical and research priorities
One of the most important implications of this paper is that resilience should not be measured only by the ability to withstand stress. It should also be measured by the ability to recover from it.
This suggests several research directions.
First, we need better ways to measure recovery kinetics. Static biomarkers are useful, but they may miss dynamic vulnerability. Longitudinal patterns, response-to-challenge tests, recovery curves, and integrated biomarker profiles may provide more meaningful information than isolated thresholds.
Second, mitochondrial reserve capacity, NAD⁺ availability, redox flexibility, and intercellular bioenergetic support deserve more attention as determinants of resilience. Emerging evidence on extracellular vesicles, mitochondrial transfer, and mitochondrial communication suggests that recovery may be coordinated across cells and tissues, not only within individual cells.
Third, interventions should be studied not only for their ability to suppress symptoms or reduce downstream risk markers, but for their ability to restore recovery capacity. This includes strategies that reduce mitochondrial congestion, improve temporal rhythm, support sleep and circadian alignment, provide adequate protein and micronutrients, improve metabolic flexibility, and appropriately dose physical activity.
In this model, exercise is not simply another stressor. When properly timed and scaled, it may expand mitochondrial throughput and improve the capacity to process substrates. Similarly, nutrition is not only about intake quantity, but also about timing, substrate quality, and whether the system has the capacity to use what is supplied.
What we hope the paper contributes
This article is conceptual and hypothesis-generating. It does not claim that ERM is a validated diagnostic entity, nor that mitochondrial constraint is the only cause of resilience failure. Genetic, immune, psychosocial, environmental, developmental, and behavioral factors all shape stress exposure and recovery capacity.
Our goal is to provide a unifying framework that helps connect these influences through a common biological question: can the system complete recovery?
If the answer is no, then stress adaptation may remain chronically engaged. Repair may be postponed. Maintenance may be underfunded. Functional symptoms may emerge before disease thresholds are crossed. Over time, unresolved adaptation may become biology’s quiet pathway from resilience to decline.
The central message of the paper is therefore simple:
Patients may not be failing to adapt to stress.
They may be failing to recover because recovery is energetically underfunded.
For the research community, we hope this perspective stimulates empirical work on recovery kinetics, mitochondrial reserve, redox flexibility, multidomain biomarker patterns, and early reversible vulnerability states. For clinicians, we hope it encourages a shift from asking only “What disease does this patient have?” toward also asking, “What is preventing this patient from completing recovery?”
Stress will always be part of life. The more important question may be whether biology still has the resources, rhythm, and mitochondrial capacity to resolve it.
Tippairote, T., Hoonkaew, P., Suksawang, A. et al. Stress is inevitable; recovery is conditional: bioenergetic limits of resilience in aging and disease. Biogerontology 27, 96 (2026). https://doi.org/10.1007/s10522-026-10445-w
Follow the Topic
-
Biogerontology
Biogerontology is a peer-reviewed journal dedicated to exploring the biological basis and mechanisms of ageing, with an aim of promoting healthy old age.
Related Collections
With Collections, you can get published faster and increase your visibility.
Viral Etiology in Brain Ageing and Longevity
Ageing is increasingly recognized as a state of heightened vulnerability to the reactivation of latent viruses and the lingering effects of systemic viral insults. This phenomenon, termed ‘Viral-induced Neuroinflammageing,’ involves the persistent activation of microglia and astrocytes in response to viral ligands, leading to chronic neuroinflammation, DNA damage, and accelerated neuronal senescence. The call for this special issue on ‘Viral Etiology in Brain Ageing and Longevity’ invites review and research articles exploring the role of neurotropic viruses (such as HSV, VZV, and Retroviruses) and systemic viral infections (including SARS-CoV-2) in modulating brain health. We seek contributions that investigate how viral persistence alters the blood-brain barrier and triggers neurodegenerative cascades. A particular focus will be placed on the disruption of glymphatic clearance—the brain’s essential waste-clearance pathway—and the impairment of cell-to-cell communication.
Crucially, this issue will explore the alteration of junctional proteins (such as Connexins) and the subsequent breakdown of gap junctional communication within glial networks. Such dysregulation of metabolic and electrical coupling is a significant contributor to the neuropathology of the ageing brain and may be exacerbated by viral interference.
Furthermore, this issue aims to highlight the importance of experimental animal models in deciphering the etiology of viral-induced neuroinflammageing. By bridging the gap between virology, cell biology, and biogerontology, this issue intends to identify how targeting viral pathways, junctional integrity, and host-virus interactions may provide novel therapeutic avenues for promoting healthy brain ageing and extending longevity.
Publishing Model: Hybrid
Deadline: Nov 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in