Zero‑Energy Urine Mining: How Hydrogen‑Bonded Covalent Organic Frameworks Can Selectively Harvest Urea Without Heat, Pressure, or Power
Published in Earth & Environment, Materials, and Sustainability

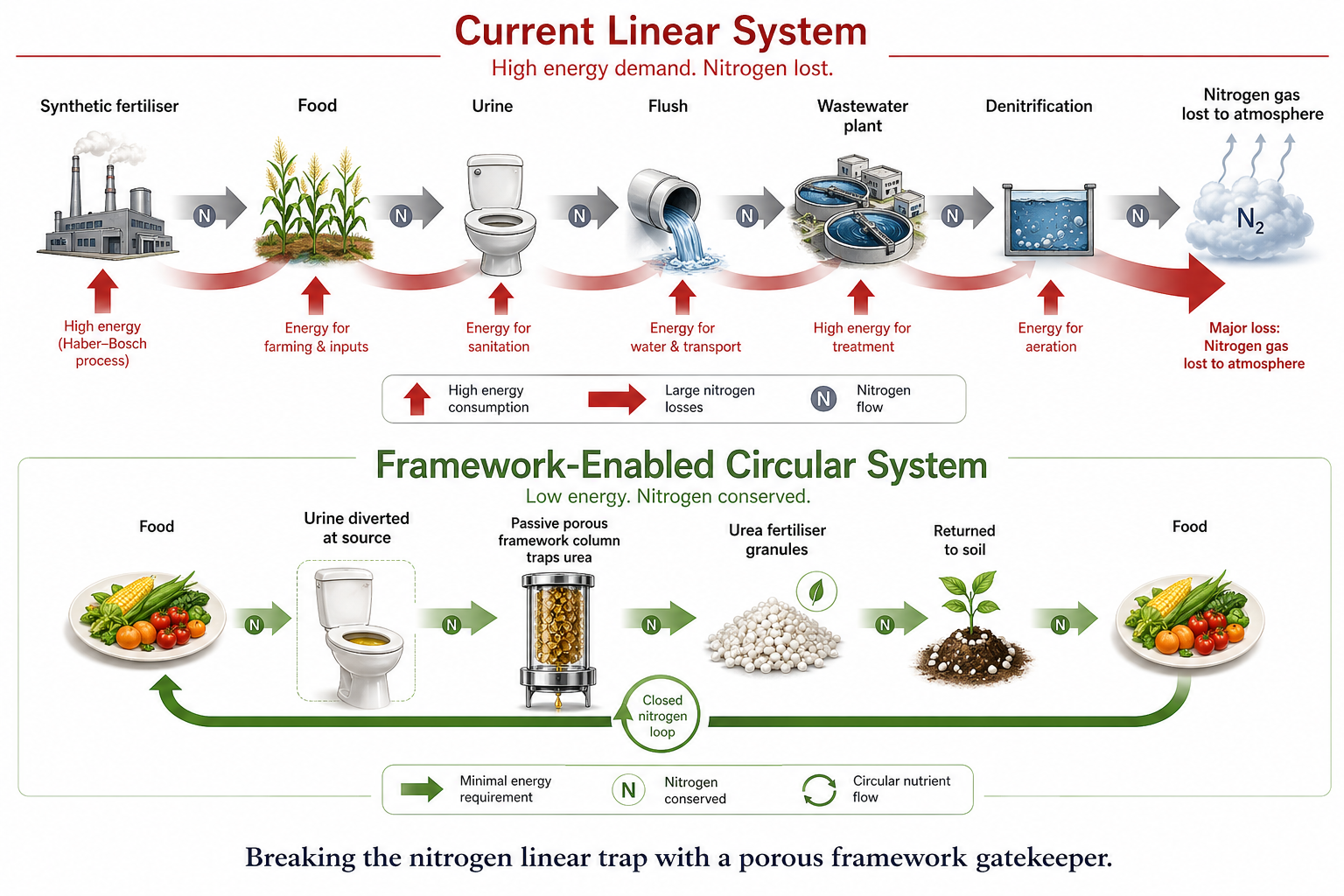
The Absurdity of the Modern Nitrogen Cycle
Sanitation infrastructure operates on a fundamentally flawed linear logic: collect, treat, and discard. This approach has turned the global nitrogen cycle into a costly, energy-intensive contradiction.
Human urine carries up to 80% of the reactive nitrogen entering municipal wastewater. Yet, treatment plants spend massive amounts of energy and capital converting that useful nitrogen into inert dinitrogen gas, often releasing nitrous oxide (N₂O)—a greenhouse gas 300 times more potent than CO₂—in the process. Simultaneously, the agricultural sector burns fossil fuels at extreme temperatures to synthesise urea fertiliser via the Haber-Bosch process.
We literally dismantle useful nitrogen molecules in one facility, only to painstakingly reassemble them in another. The obvious circular alternative—intercepting urinary urea at the source—has long been stalled by urea’s rapid enzymatic hydrolysis into volatile ammonia.
The Selectivity Bottleneck: Why Water Usually Wins
The central obstacle to source-separated urine mining is not availability; it is selectivity.
Urea is a small, uncharged molecule present at roughly 0.3 M in fresh urine, drowning in water at a concentration of 55 M. Any sorbent deployed here must distinguish urea from water with extraordinary fidelity, while ignoring competing salts, creatinine, and organic metabolites.
Traditional materials fail this test. Ion-exchange resins lack supramolecular precision; activated carbons and zeolites offer terrible selectivity; and many conventional metal-organic frameworks (MOFs) suffer hydrolytic degradation under prolonged water exposure. A material that can operate passively, reject water, and reversibly bind urea has, until recently, been purely theoretical.
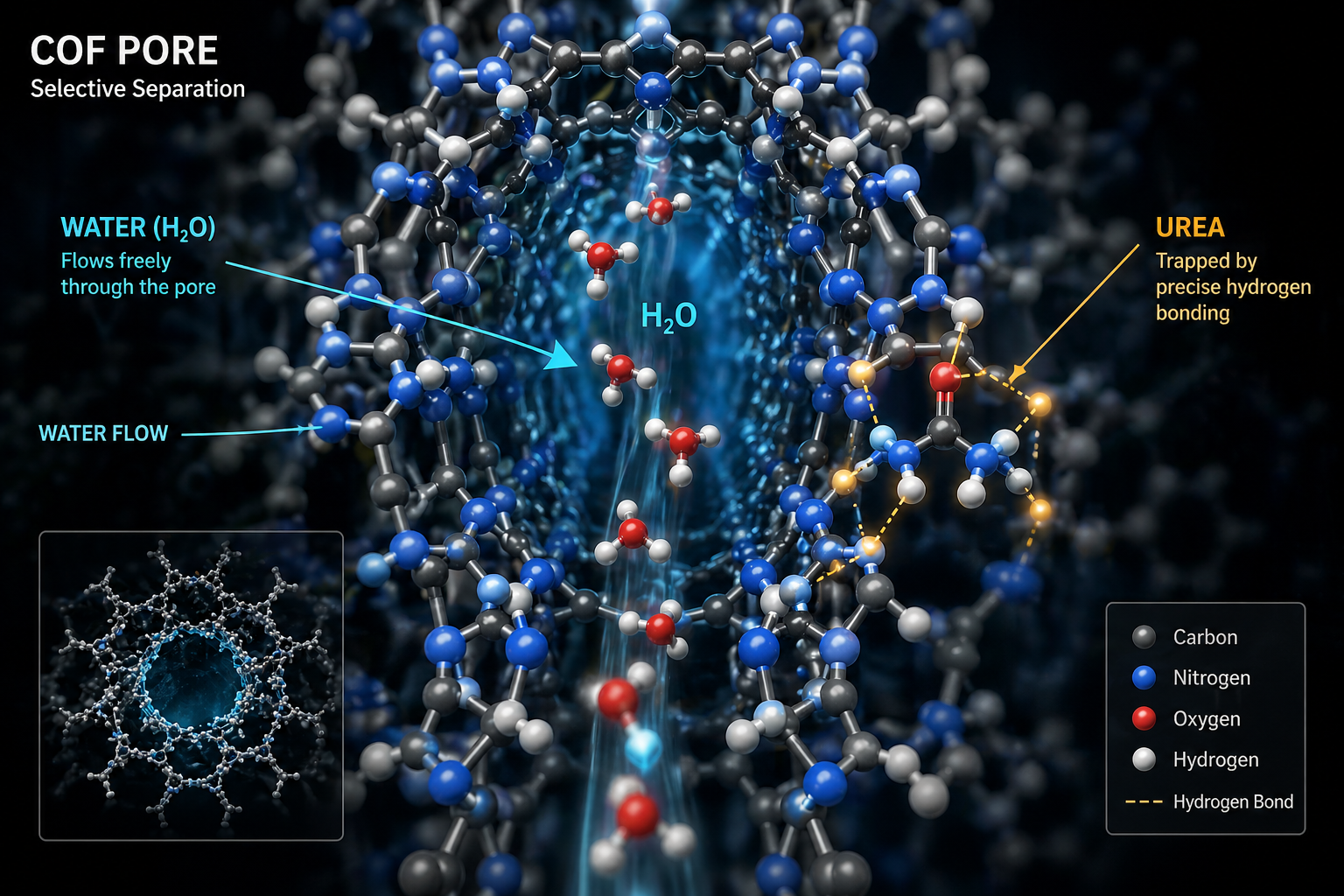
Hydrogen-Bonded COFs: The Supramolecular Sieve
Covalent Organic Frameworks (COFs) have entirely rewritten the separation playbook. Constructed from lightweight organic monomers linked by robust covalent bonds, COFs combine permanent microporosity with exceptional hydrothermal stability.
But their true power lies in atomic-level tunability. By integrating complementary hydrogen-bond donors and acceptors—like amide, urea, carboxyl, and hydroxyl groups—into the framework, we can line pores with dimensions perfectly matching urea’s kinetic diameter.
This creates a multidentate, hydrogen-bonded "pocket." It recognizes urea through cooperative supramolecular interactions, snapping it into place, while the high-dielectric water molecules simply slip through unimpeded. The framework functions as a shape- and hydrogen-bond-selective gate, capturing urea with efficiencies exceeding 98% in real urine matrices.
The Ambient Advantage: Defeating Haber-Bosch
The operational elegance of this approach cannot be overstated.
Fresh urine is introduced into a gravity-fed column. The COF retains the urea while the stripped effluent drains away passively. Once saturated, a simple warm-water wash disrupts the hydrogen-bond network, releasing a highly concentrated urea stream ready for crystallization.
The framework returns to its active state, repeating the cycle hundreds of times without capacity loss. Zero applied voltage. Zero extreme heat. Zero chemical regenerants.
When juxtaposed with Haber-Bosch synthesis—which requires 400–500 °C and 150–300 bar of pressure—the energy ledger swings violently in favor of COFs. This is passive, selective recovery of high-purity nitrogen from an unavoidable waste stream.
The Reality Check: Bench to Basin
However, the research community must confront the translation gap. We cannot solve global infrastructure problems with milligram-scale chemistry.
-
Synthesis Scale-Up: Current COF syntheses are predominantly solvothermal and expensive. Transitioning to mechanochemical and continuous-flow routes is mandatory for multi-kilogram production.
-
Biological Fouling: Real urine is biologically active. Extracellular polymeric substances (EPS) and microbial biofilms will inevitably challenge long-term pore integrity and sorption kinetics.
-
Infrastructure: Source-separated collection systems (like modern urine-diverting toilets) are still in their infancy globally.

Conclusion
Hydrogen-bonded COFs offer something that environmental engineering rarely sees: the opportunity to entirely eliminate an energy demand rather than merely optimizing it. By shifting the paradigm from "waste treatment" to "nutrient mining," these materials turn a century-old linear problem into a profitable, closed-loop solution.
The question for our community is no longer whether urea can be captured at the source with precision.
The question is: Who will build the first scalable pilot that proves this can survive the biological realities of a municipal basin?




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
I've laid out the materials case—what's the biggest non‑technical barrier you see? Public acceptance? Supply chain? Regulatory inertia? I'd genuinely like to hear from water engineers, sanitation specialists, and behavioural scientists.