2-DG reprograms mitochondrial metabolism to rescue CD8 T cells impaired by HIV-1
Published in Biomedical Research
Type I interferons (IFN-I) are essential for early antiviral defense, yet their role in chronic infection is paradoxical. While transient IFN-I signaling restricts viral replication, sustained activation has been associated with immune dysfunction. In our previous studies, we showed that blockade of IFN-I signaling or depletion of plasmacytoid dendritic cells, the major IFN-I producing cells, in HIV-infected humanized mouse models restores CD8⁺ T cell function and reduces viral reservoirs. However, the mechanistic basis for this effect has remained unclear.
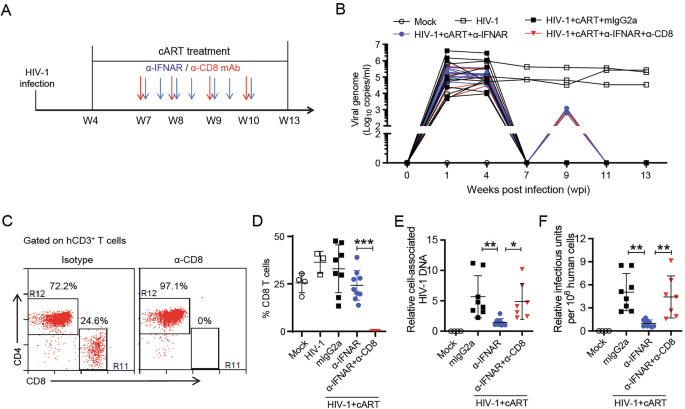
Using HIV-infected humanized mice under ART, we first confirmed that the reduction of viral reservoirs following IFN-I blockade is dependent on CD8⁺ T cells, highlighting their central role as effector cells in reservoir clearance.
To investigate the underlying mechanisms, we performed transcriptomic analyses of CD8⁺ T cells and observed a striking upregulation of mitochondrial metabolic pathways—including the tricarboxylic acid cycle, electron transport chain, and oxidative phosphorylation—upon IFN-I blockade. These findings suggested that IFN-I signaling may impose a metabolic constraint on CD8⁺ T cells.
Functional analyses supported this hypothesis. HIV infection led to a marked reduction in mitochondrial membrane potential in CD8⁺ T cells, a defect that was not corrected by ART alone. In contrast, combined ART and IFN-I blockade restored mitochondrial function. In vitro experiments further demonstrated that sustained IFN-I signaling directly suppresses mitochondrial metabolism in CD8⁺ T cells. Importantly, analysis of single-cell transcriptomic datasets from individuals living with HIV revealed a similar pattern: elevated interferon response signatures coupled with diminished oxidative phosphorylation, which persisted despite ART.
Together, these results identify a previously unappreciated mechanism whereby chronic IFN-I signaling drives CD8⁺ T cell dysfunction through suppression of mitochondrial metabolism.
We next asked whether this metabolic impairment could be therapeutically targeted. Screening a panel of metabolic interventions revealed that low-dose 2-deoxy-D-glucose (2-DG) uniquely restored CD8⁺ T cell function. Treatment with 2-DG enhanced the production of IL-2, IFN-γ, and TNF-α, and improved HIV-specific CD8⁺ T cell responses across multiple experimental systems.
In vivo administration of 2-DG (5mg/ml in drinking water) to ART-treated, HIV-infected humanized mice further demonstrated its therapeutic potential. 2-DG restored mitochondrial membrane potential, reduced aberrant activation markers, and rebalanced T cell subsets toward a central memory phenotype. Notably, expression of exhaustion markers such as PD-1 was decreased, accompanied by enhanced antigen-specific T cell responses.
Given that functional restoration of CD8⁺ T cells alone may be insufficient to eliminate latent reservoirs, we combined 2-DG with the latency-reversing agent poly(I:C). This combinatorial approach led to a significant reduction of viral reservoirs in both spleen and bone marrow, supporting a cooperative strategy in which reservoir reactivation is coupled with enhanced immune clearance.
To assess clinical relevance, we examined the effects of 2-DG on peripheral blood mononuclear cells from ART-treated individuals with HIV. 2-DG treatment enhanced CD8⁺ T cell effector function under both TCR-dependent and -independent stimulation conditions and upregulated transcriptional programs associated with mitochondrial biogenesis and effector cytokine signaling. Moreover, 2-DG suppressed ex vivo expansion of viral reservoirs, further supporting its translational potential.
Collectively, our findings establish a mechanistic link between chronic IFN-I signaling, mitochondrial dysfunction, and CD8⁺ T cell exhaustion. By identifying immunometabolic reprogramming as a key determinant of antiviral immunity, this study provides a conceptual framework for targeting metabolism to restore immune function in chronic infection.
Importantly, 2-DG has been previously evaluated in clinical settings, particularly in oncology, where it has demonstrated favorable safety and tolerability profiles. This raises the possibility of repurposing 2-DG as part of combination strategies aimed at achieving functional cure of HIV.
Looking ahead, integrating metabolic interventions with approaches that reactivate latent reservoirs and enhance immune effector function may represent a promising path toward durable HIV control.
Follow the Topic
-
Cellular & Molecular Immunology
A monthly journal from the Chinese Society of Immunology and the University of Science and Technology of China, covering both basic immunology research and clinical applications.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in