A serendipitous path to discovering a potent monoclonal antibody (mAb) for the treatment of human metapneumovirus (HMPV) infections
Published in Biomedical Research

Back in 2021, during the heart of the COVID-19 pandemic, our lab at the Fred Hutchinson Cancer Center in collaboration with industry partners embarked on a discovery campaign for highly potent mAbs that could be used as therapy for HMPV infections. HMPV has two major surface proteins: the G glycoprotein mediates attachment, whereas the F fusion protein mediates viral entry. In contrast to F, the G protein is highly variable, and others had previously found that vaccination with G protein failed to induce robust neutralizing antibodies or protective immunity in animals. Therefore, we focused our attention on the F protein. The F protein transitions between a metastable prefusion (preF) conformation and a stable postfusion (postF) conformation. Since preF is the major conformation on infectious virions, antibodies to preF tend to be the most potent at neutralizing virus.
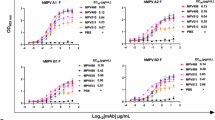
To identify highly potent neutralizing antibodies to HMPV, we decided to take a deep dive into the human B cell repertoire. Virtually all adults have been infected with HMPV and experience repeated infections over their lifetime. We started our search with almost one billion immune cells from healthy adult donors. We leveraged a ‘bait and switch’ strategy to enrich for B cells that could neutralize multiple subtypes of HMPV. This strategy is based on the rationale that B cells capable of binding to one subtype, while neutralizing another subtype, are more likely to cross-neutralize both subtypes. We used fluorescent probes to identify individual B cells able to bind the recombinant F protein of one HMPV subtype, which was stabilized in the preF conformation by introduction of a GCN4 trimerization domain. This was followed by magnetic enrichment and sorting of individual B cells into culture to stimulate antibody secretion. To identify candidate B cells expressing cross-neutralizing antibodies, culture supernatants were screened for their ability to neutralize a different subtype of HMPV. The 4F11 antibody was the most potent antibody we isolated in our antibody discovery campaign.
In the course of characterizing 4F11's mechanism of neutralization, we uncovered several unexpected findings. First, whenever we mixed 4F11 with the HMPV preF trimer that was partially stabilized by a GCN4 trimerization domain, the trimer dissociated into monomers. In an attempt to obtain a structure with an intact HMPV preF trimer, we turned to alternative versions of prefusion-stabilized trimers. Many of these newer constructs contain a proline mutation at amino acid 185 (Pro185) at the apex of the F protein (called site Ø ) to confer local rigidity and disfavor the transition to the postfusion conformation. However, 4F11 was unable to bind to these stabilized trimers. We noticed that the Pro185 mutation was located within the 4F11 epitope and indeed, reverting proline 185 back to an alanine (P185A) rescued binding of 4F11. However, even after solving this conundrum, mixing 4F11 Fab with the reverted P185A trimer still led to trimer dissociation. We tried mixing the trimers with other antibodies to help stabilize them, but to no avail. We then moved on to even newer, stabilized versions of HMPV preF, including those that are stabilized with interprotomer disulfide bonds. Unfortunately, we still ended up with trimer dissociation after mixing with 4F11. As a last ditch effort, we turned to the most recently published, stabilized version of the HMPV preF trimer, MPV-2c which does not rely on stabilization with Pro185. Fortunately, the MPV-2c trimer remained intact when co-transfected with 4F11 Fab, and we were able to obtain a cryoEM structure of this complex. We found that 4F11 binds site Ø and only one Fab is bound per trimer. Modeling the trimer with two or three 4F11 Fabs revealed steric clashes between Fabs. Based on these data, we think that 4F11 likely facilitates trimer dissociation when more than one Fab tries to bind the trimer, because the Fabs clash with each other.
The saying "hindsight is 20/20" rings true here. We had serendipitously used HMPV F without the Pro185 stabilizing mutation in our 'bait and switch' strategy. GCN4-stabilized HMPV F was first described in 2012 and newer, more stable constructs of HMPV F have since been reported, but we used GCN4-stabilized HMPV as the probe in our B cell sorting experiments, because this was the only recombinant version of HMPV F reported at the time of our experiments to adopt a prefusion conformation. Structural analysis of GCN4-HMPV F has revealed that a mixture of prefusion and postfusion conformations is present, indicating that GCN4-HMPV F is relatively less stable compared to newer versions of recombinant HMPV F. However, if newer constructs with the Pro185 mutation had been used in our experiments to sort B cells, then 4F11-like antibodies would have been missed.
In the interest of brevity, I will just list five other surprising observations from this study: (i) 4F11 accommodates the glycan shield at site Ø by approaching vertically, dodging the Asn57 glycan, and interacting with the Asn172 glycan; (ii) The light chain of 4F11 interacts with two protomers of the trimer; (iii) An escape mutation with a lysine to glutamic acid substitution at amino acid 179 (K179E) in the F protein can arise in vitro but is highly attenuated; (iv) No viruses with a K179E substitution have been observed in nature; and (v) a relatively low dose of 4F11 administered one day after infection blocked viral replication in the lungs of most animals and significantly reduced replication in the nose.
There are no available treatment options for HMPV infection, and no mAbs are currently approved for clinical use against this virus. 4F11's high potency, neutralizing activity against diverse strains, unique glycan-dependent binding mechanism, low resistance susceptibility, and significant in vivo therapeutic efficacy support its further advancement. 4F11 may provide a much-needed therapeutic option for vulnerable populations, including infants, the elderly, and immunocompromised individuals, who are most at risk for severe HMPV disease.
In summary, this collaborative study that began in 2021 establishes 4F11 as a promising candidate for clinical development against HMPV infection. The lead author Evelyn Harris is pictured presenting these results at the NSV meeting in Montpellier, France in 2025.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in