Behind the Paper: When the Matrix Speaks Up During Radiotherapy
Published in Biomedical Research
Scientific insights do not always emerge at a defined moment; they often arise from persistent “inconsistencies” that resist simple explanations. In oncology, one such puzzle stood out: despite being a cornerstone of treatment for head and neck cancers, why does radiotherapy so often fail to achieve tumor control?
From Tumor Cells to the Microenvironment
In clinical practice, radiotherapy shrinks tumors, reduces symptoms, and prolongs survival in many cases. Yet, relapse remains common. For years, research focused on tumor cells themselves with DNA damage, repair mechanisms and intrinsic resistance. Increasingly, however; evidence points beyond cancer cells to their surroundings.
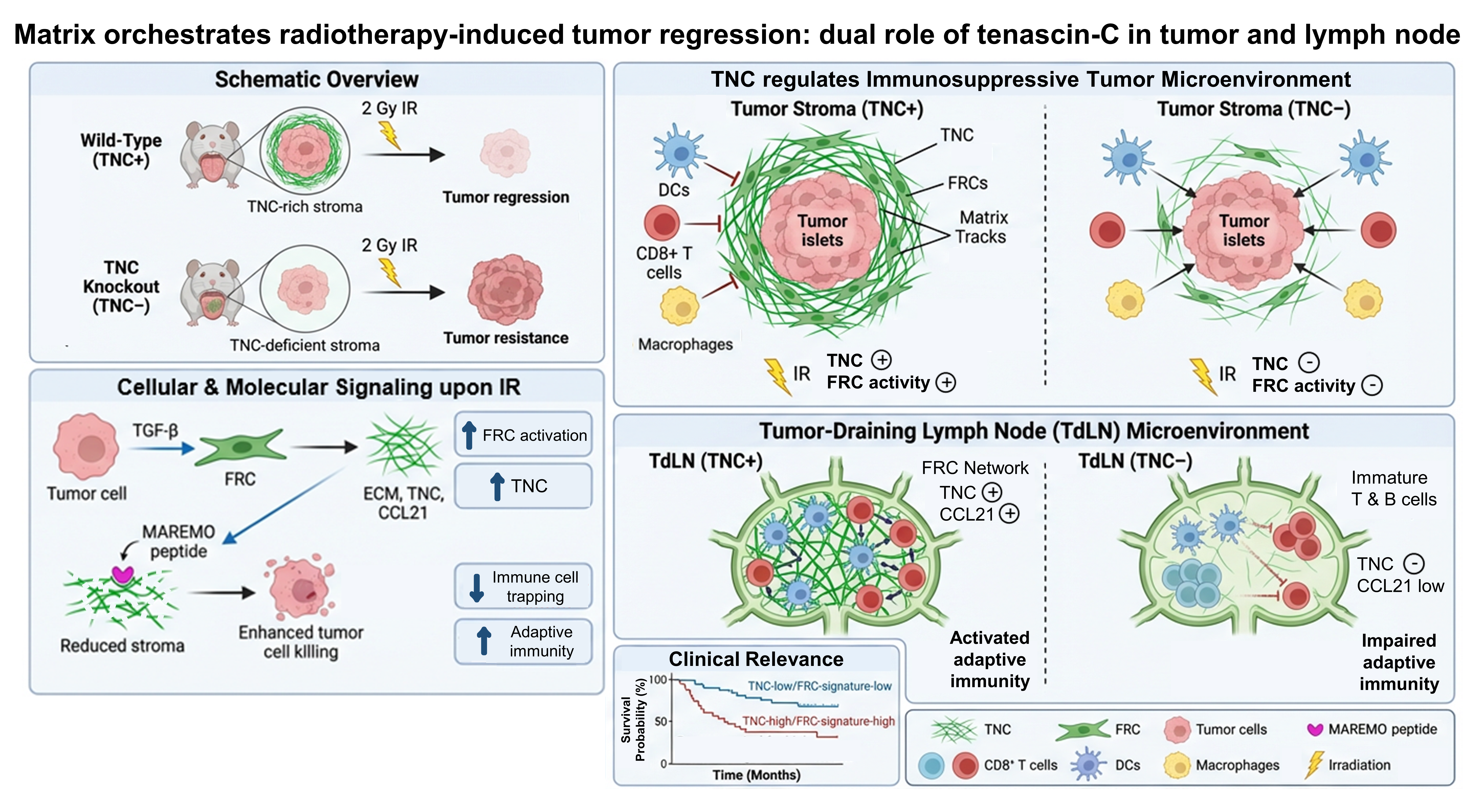
Our attention turned to the tumor microenvironment, specifically the extracellular matrix, a complex, dynamic network often seen as mere scaffolding but now recognized as biologically active. Among its components, tenascin-C is highly expressed in many cancers, induced by stress signals and implicated in immune modulation and tumor progression. Its role in radiotherapy, however, remained unclear.
A Dual Role for Tenascin-C Revealed
The project crystallized around a hypothesis: could tenascin-C regulate the balance between tumor regression and persistence post-radiotherapy? Using a murine model of oral squamous cell carcinoma we compared tumors with and without tenascin-C.
Radiotherapy’s effect were not uniform, it hinged on the presence of tenascin-C in the microenvironment. Surprisingly, tenascin-C played a dual compartmentalized role. Within tumors, tenascin-C contributed to an immunosuppressive environment. Immune cells were present, but restrained, trapped within the stromal compartment and unable to effectively reach and eliminate cancer cells. This spatial organization, shaped by the matrix, appeared to limit the efficiency of the anti-tumor immune response.
In the tumor-draining lymph nodes, key sites for immune priming, tenascin-C supported immune response organization. Its depletion led to dysregulation.
This paradox resolved spatially: tenascin-C’s function depends on location and cellular partners. Fibroblast reticular cells, known for structuring lymph nodes, their function within tumors is less well understood. In tumors, Fibroblast reticular cells produce tenascin-C and irradiation activates surviving Fibroblast reticular cells to remodel the microenvironment, promoting tumor persistence.
Translational Promise
If tenascin-C contributes to an environment that limits the efficacy of radiotherapy, could targeting tenascin-C improve treatment response? Targeting tenascin-C with a MAREMO peptide, disrupting its function, enhanced tumor radiosensitivity and reduced features associated with tumor plasticity and regrowth. Clinically, high levels of tenascin-C and a strong Fibroblast reticular cells signatures correlated with poorer radiotherapy outcomes, positioning tenascin-C as a potential prognostic marker.
This work reflects a broader shift in cancer research. Tumors are no longer seen as isolated masses of malignant cells, but as complex ecosystems shaped by constant interactions between cancer cells, immune cells, and the surrounding matrix. What makes this story particularly meaningful is the convergence of perspectives that made it possible. It required input from basic researchers, clinicians, imaging specialists, and bioinformaticians, as well as patience, since many of the key insights emerged only 10 years later after integrating molecular, cellular, and tissue-level data. Behind this paper, there was no single defining moment, but rather a gradual realization that the extracellular matrix is not just a scaffold, but is an active participant in therapy response. This opens new possibilities. By targeting components such as tenascin-C, we may be able to tilt the balance in favor of durable tumor regression. More broadly, it highlights the importance of designing therapeutic strategies that take into account not only cancer cells, but also their environment. Ultimately, the goal remains the same: to improve patient outcomes. And sometimes, this means listening not only to the cells we see, but also to the matrix that surrounds them.
Follow the Topic
-
EMBO Molecular Medicine
EMBO Molecular Medicine (EMM) publishes breakthrough research in translational and biomedical sciences in the field of experimental medicine.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in