Why is Singapore Identified in Global Research as Number One? How Physical Activity and Education Excellence Created a Global Leader

Published in Neuroscience
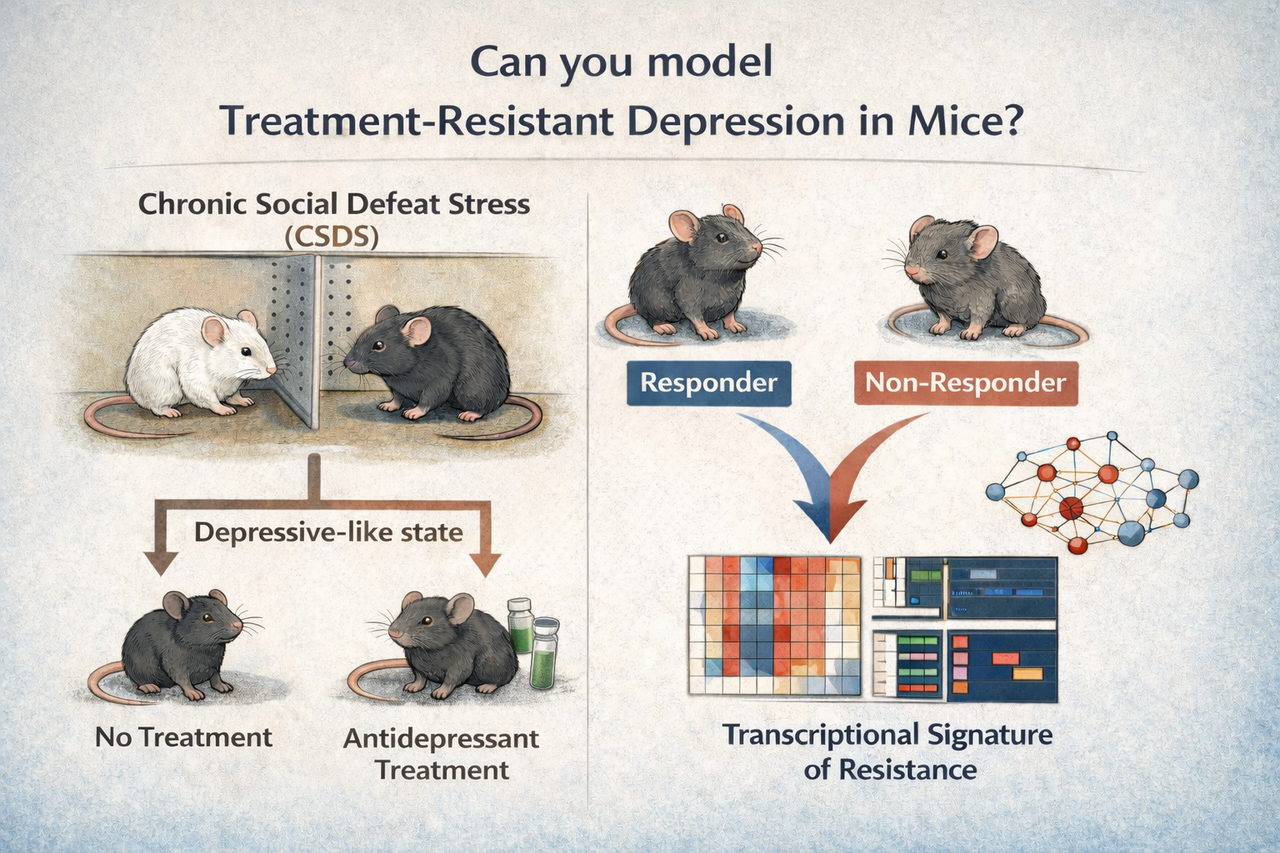
When someone is living with depression, finding a treatment that actually helps can take time, often through painful trial and error. For some people, that process is even harder: after trying more than one antidepressant at an appropriate dose and duration, symptoms still don’t meaningfully improve. Clinicians call this treatment-resistant depression (TRD), and for patients and families it can feel like running out of options.
We wanted to ask a simple, but surprisingly tricky, question: can you model TRD in a mouse?
TRD in humans is often defined as failure to respond to two adequate antidepressant trials. But what does “adequate” mean in a mouse? In people, treatment can last months. In rodents, depressive-like behaviors induced by chronic social defeat stress (CSDS) may not persist long enough to realistically model that timeline. The biology and the clock don’t align neatly.
So we had to make a decision: do we try to replicate the clinical definition exactly, or do we model the core concept of treatment resistance?
We chose the latter.
After CSDS, we identified susceptible mice using the social interaction test. We then treated one group with fluoxetine (FLX) for 30 days, long enough to approximate chronic SSRI exposure in rodents. About 65% responded. Roughly 35% did not. That number caught our attention: it mirrors the proportion of patients who do not benefit from first-line antidepressant treatment.
But here’s where the story became interesting. We next gave an acute injection of ketamine to all of the fluoxetine non-responders. In parallel, we gave ketamine to a separate group of susceptible mice that had never received fluoxetine. The results surprised us. None of the mice that received ketamine as a first-line treatment responded at the 4-week post-stress time point. But 50% of the fluoxetine non-responders did respond to ketamine.
Ketamine has repeatedly been shown to reverse stress-induced behavioral deficits in mice when given within two weeks after CSDS. By four weeks, that window may close. We expected ketamine to fail across the board at that later time point.
It didn’t.
Fluoxetine exposure, even when behaviorally ineffective, appeared to “prime” the system to respond to ketamine. That raised a new possibility: perhaps treatment resistance is not simply the absence of drug effect. Perhaps antidepressants still induce molecular changes in non-responders—changes that alter how the brain responds to subsequent treatments.
To explore that idea, we performed bulk RNA sequencing. We wanted to know whether mice that fail to respond to antidepressants share a molecular signature. Are non-responders biologically similar to one another? Or is “treatment resistance” simply noise?
What we found suggests that non-response is not random. There is a transcriptional signature associated with treatment resistance, a pattern that distinguishes mice that fail multiple treatments from those that respond.
Of course, we are cautious. This is still a rodent model. We cannot replicate the full clinical trajectory of TRD, with months-long treatments and washout periods. Single housing our mice is necessary to track fluoxetine consumption, and it adds another layer of complexity because it can be viewed as an additional stressor.
But perhaps modeling TRD does not require perfect replication of the human definition. Perhaps it requires capturing the biological logic: that some individuals fail first-line treatment, some respond to second-line therapies, and that these outcomes reflect underlying molecular differences.
This study began with skepticism: can we really model treatment resistance in rodents? It ended with a more nuanced answer.
Maybe not perfectly.
But perhaps closely enough to start asking the right biological questions.
I am a neuroscientist and PhD candidate at the Icahn School of Medicine at Mount Sinai, where I study the molecular mechanisms underlying stress resilience and antidepressant response. My work focuses on transcriptional and gene network changes in the nucleus accumbens using RNA-seq and computational approaches to better understand treatment-resistant depression.
I am particularly interested in how individual differences in stress susceptibility and treatment response are encoded at the transcriptomic level, and how these signatures can inform more precise therapeutic strategies. My research integrates behavioral neuroscience, bioinformatics, and systems-level analyses to uncover biological pathways that distinguish responders from non-responders to antidepressant treatments.

An international scientific journal and the official publication of the American College of Neuropsychopharmacology (ACNP). This journal focuses on clinical and basic science contributions that advance our understanding of the brain and behaviour
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcement Read more
We use cookies to ensure the functionality of our website, to personalize content and advertising, to provide social media features, and to analyze our traffic. If you allow us to do so, we also inform our social media, advertising and analysis partners about your use of our website. You can decide for yourself which categories you want to deny or allow. Please note that based on your settings not all functionalities of the site are available.
Further information can be found in our privacy policy.
Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in