Changing Conversations: Immune Cells in the Aging Ovary
Published in Protocols & Methods, Cell & Molecular Biology, and General & Internal Medicine

The mammalian ovary is a highly dynamic and cellularly complex reproductive organ responsible for housing and cyclically maturing female germ cells, called oocytes, for fertilization. The ovary is partitioned into follicles: spherical units that contain a central oocyte surrounded by populations of nurse cells specialized to support the oocyte’s unique needs as it grows and prepares for ovulation1. As an oocyte matures, the somatic cells of the follicle must grow, divide, and differentiate to meet the oocyte’s changing needs1. Not only does the follicle have to change, but the surrounding stromal tissue of the ovary must remodel to accommodate growing follicles2. Once the oocyte is fully matured and ready for fertilization, the follicle ruptures, creating an opening through which the oocyte is released. The remaining somatic cells of the follicle then regress into a hormone-producing structure that supports early pregnancy, if the oocyte is fertilized3.
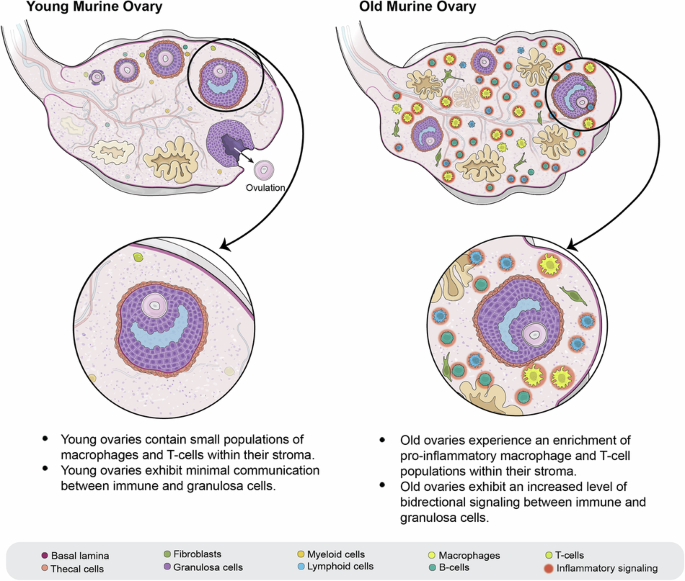
The constant cyclicity of follicular growth, destructive rupture, and restoration, creates a highly dynamic tissue environment. A diverse population of ovarian immune cells work tirelessly to remodel ovarian tissue to encourage healthy follicle growth as well as clear debris upon ovulation and follicular regression4, 5, 6. However, with age, these constant cycles contribute to a dysregulated, chronically inflamed tissue environment, a phenomenon that has been termed inflammaging. It has been speculated that inflammaging plays a major role in ovarian dysfunction with age, but the aging ovary is a complex cellular environment with many unknowns.
Major remaining open research areas in the field include understanding the full repertoire of immune cell types in young and aged ovaries, the changing character of follicular and stromal cells with age, and the complex signaling interactions between cell types. Our study tackled these questions by coupling single-cell and spatial transcriptomics to better define the cell identities of the immune milieu as well as their organization and interaction with the follicular unit. In this publication we identified many immune cell types, including new types of proinflammatory immune cells in the aged ovary and the marker genes that define them. We identify inflammatory signatures in immune cells with age, and how this alters inter-cellular communication between the granulosa and immune cells of the stroma. Furthermore, we find that with age, the granulosa cells increase their inflammatory immune signaling. Our work provides additional evidence for aging-related immune populations, novel insight into how these aging-associated immune cells communicate with each other and with the stroma, and evidence that they are significant contributors to the inflammatory signaling driving inflammaging. Together, these findings will help guide future studies targeting these cell types for in-depth functional characterization.
This study also provided significant training opportunities for the authors. For Dr. Joe Varberg, who trained as a cell biologist, the project provided foundational experience in the analysis and visualization of single cell datasets that helped launch a new direction in his career as a bioinformatician. Anna Galligos participated in the Chan-Zuckerberg Initiative Reproductive Annotation Jamboree, a consortium of reproductive scientists that gathered to collectively streamline reproductive single-cell data and standardize reproductive cell ontology.
References:
- Richards JS, Pangas SA. The ovary: basic biology and clinical implications. J Clin Invest 120, 963-972 (2010).
- Kinnear HM, et al. The ovarian stroma as a new frontier. Reproduction 160, R25-R39 (2020).
- Stocco C, Telleria C, Gibori G. The molecular control of corpus luteum formation, function, and regression. Endocr Rev 28, 117-149 (2007).
- Field SL, Dasgupta T, Cummings M, Orsi NM. Cytokines in ovarian folliculogenesis, oocyte maturation and luteinisation. Mol Reprod Dev 81, 284-314 (2014).
- Matsuura T, Sugimura M, Iwaki T, Ohashi R, Kanayama N, Nishihira J. Anti-macrophage inhibitory factor antibody inhibits PMSG-hCG-induced follicular growth and ovulation in mice. J Assist Reprod Genet 19, 591-595 (2002).
- Kirsch TM, Friedman AC, Vogel RL, Flickinger GL. Macrophages in corpora lutea of mice: characterization and effects on steroid secretion. Biol Reprod 25, 629-638 (1981).
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
Mechanistic insights into human host and microbiome interactions
Publishing Model: Open Access
Deadline: May 31, 2026
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Jun 30, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in