Doubling down on carbon capture
Published in Chemistry
It all began September 26, 2017, in Houston, Texas, where the principal investigators were attendees at the Mission Innovation summit in Houston, Texas, USA. It was here where Professor Leclaire and Dr. Heldebrant first met as attendees helping set international R&D priorities for carbon capture, utilization, and mineralisation for the 24 participating countries. Here, we and 258 other attendees helped draft the report, titled Accelerating Breakthrough Innovation in Carbon Capture, Utilization, and Storage.1 During this engaging workshop, over dinner and drinks, we realized a shared research interest in Green Chemistry, and the perspective offered by the coordination chemistry of amines and captured forms of CO2 in integrated CO2 capture and utilization.
We began our initial collaboration by authoring a perspective in Green Chemistry where we issued “A Call to Green Arms” to begin applying the tenets of atom and energy efficiency into the CCUS field.2 It was here that we identified key areas of need for our field and even provided some avant garde engineering concepts for advanced CO2 capture systems.
From that initial perspective, our groups began to meet frequently holding discussions on each other’s research, and the synergies and complimentary nature of our respective works. Here, Heldebrant work on designing and testing water-lean solvents and designing new concepts around reactive carbon capture for CO2 utilization were aligned with Leclaire who focused on supramolecular chemistry to enable simultaneous CO2 capture and metal purification from waste streams. The international collaboration was growing until the COVID pandemic arrived and shut the world down for the better part of a year.
Fortuitously, during a lull in global case rates in the fall of 2021, one of Professor Leclaire’s students, Francesca Marocco was defending her PhD at the Université Claude Bernard in Lyon, France. Dr. Heldebrant was invited to serve as an external committee member. Handling the logistics of international travel during the early years of the pandemic was challenging to say the least. In part due to the creation and enforcement of vaccination passports, demonstration of proof of negative tests both directions, the coordination of travel and getting requisite travel approvals from the governments of France and the United States.
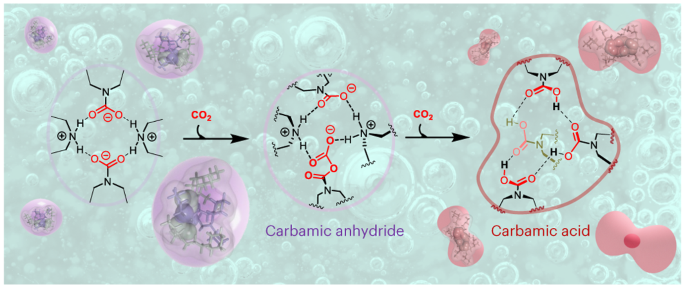
At (now) Dr. Marocco’s successful defense, Leclaire and Heldebrant spent days in the office, going over each other’s most recent results and to tour the technology incubator that housed Leclaire’s newly launched CO2/amine battery recycling company MECAWARE. It was during these discussions where Heldebrant showed his most recent publications on the performance of the single-component water-lean carbon capture solvent, EEMPA.3 Upon asking how we were able to design the amine from the molecular-level, Heldebrant pointed out his previous work designing amines to promote high degrees of internal hydrogen bonding to achieve low solution viscosities.4,5 It was previously thought that one side effect of a high degree of internal hydrogen bonding was a dynamic equilibrium between conjugate acid and base in the molecule, where the zwitterionic CO2-containing carbamate would be in equilibrium with a charge-neutral carbamic acid.6 It was here that Leclaire first asked to see yet to be published 1H NMR data on EEMPA, and a discovery was made. EEMPA was not capturing CO2 as a charge-neutral carbamic acid as theory had predicted, it was a conventional carbamate. Something else was amiss, as this carbamate did not act like others before it. Leclaire’s assessment of the NMR data upended our understanding of carbamates and carbamic acids and led us to posit how EEMPA was truly capturing CO2 to learn how EEMPA and analogues showed promise in carbon capture.
After departing Lyon, Heldebrant and team began gathering more NMR data, working in tandem with Leclaire’s group. It was during this time, that Dr. Septavaux lead a matlab modelling campaign on isotherms constructed from the extensive NMR measurements done at Lyon and PNNL on solvent made by Dr. Malhotra. It was here that the thermodynamic analysis confirmed a conventional carbamate binding mechanism as posited by Leclaire. Digging deeper into the modeling, it was then that Septavaux saw that the data could only be fitted with a tetrameric system, and everything started to make perfect sense.
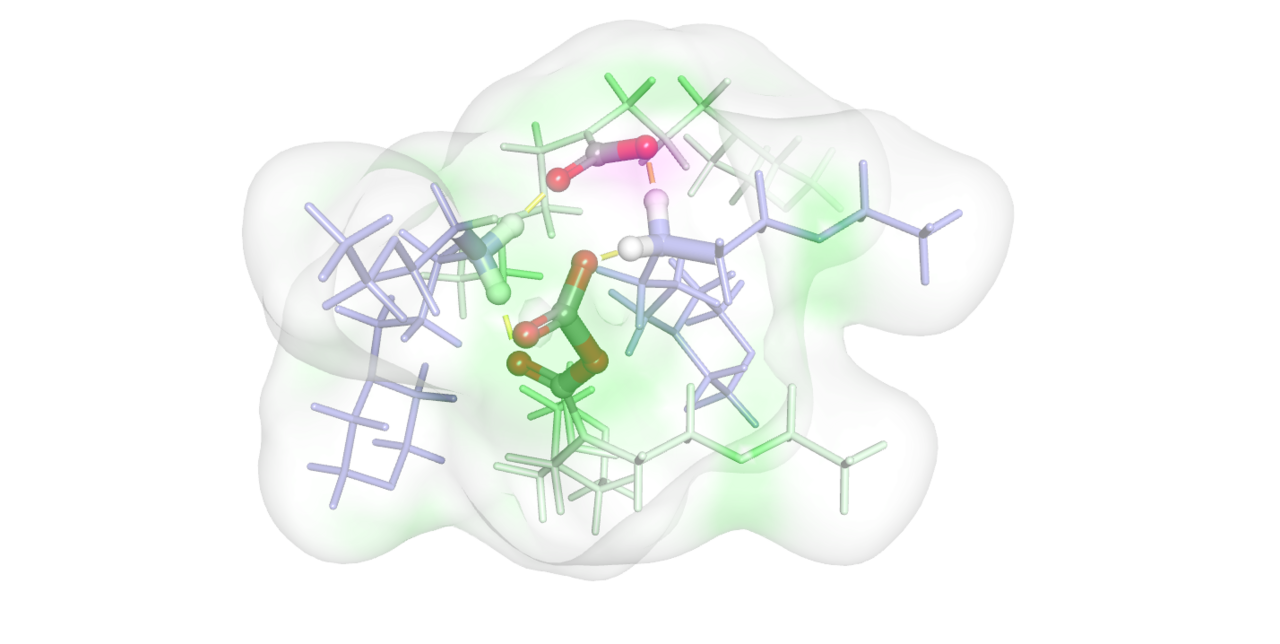
From there, Dr.’s Zhang and Nguyen began to build DFT and CMD computational models to study a conventional dimeric and assumed tetrameric systems. It was here that the first visuals of the tetrameric system began to emerge, and computational data confirmed a higher degree of stability for the tetramer over a conventional dimer. The team engaged Professor Bañuelos at the University of Texas at El Paso to perform X-ray scattering and diffraction measurements to study the orientation and speciation of EEMPA. It was here that the team gained the first direct experimental evidence of the structures of tetrameric clusters, which serendipitously matched the structures predicted from DFT.
Upon learning about the need for pressurization to capture CO2 as carbamic acids, it was Dr.’s Grubel, Walter, Chen and King who used their high-pressure NMR and IR capabilities to study the interconversion of carbamate into carbamic acid, where we first gained evidence of a transient second carbamate-like moiety in EEMPA under elevated pressures. From here, Grubel and Dr. Joo at PNNL and Dr. Hannebelle at Lyon were able to isolate this transient carbamate-anhydride and to make synthetic analogues on which to compare. With these species confirmed, Zhang and Nguyen were able to elucidate the mechanism of formation being attributed to an active site inside the tetramers that acted like an enzyme and activated CO2 to coax it into making new forms of doubly-bound CO2.
From there, our teams have worked tirelessly together to piece together various elements of this puzzle and craft the results of this long and complex story into the manuscript before you. The multidisciplinary team of physicists, computational chemists, organic chemists, supramolecular chemists, chemical engineers all came together, combining their varied expertise to complete the comprehensive 2-year study. We hope that you enjoy its story.
References
2) Green Chem. (2018), 20, 5058-5081.
3) Energy. Environ. Sci. (2020), 13, 4106-4113.
4) ChemSusChem., (2020), 13, 3429-3438.
5) ACS Sustainable Chem. Eng., (2019), 7 (8), pp 7535–7542.
6) J. Phys. Chem. Lett. (2016), 7, 1646−1652.
Follow the Topic
-
Nature Chemistry

A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in