Dual-vector rAAVrh8 gene therapy for GM2 Gangliosidosis: a Phase I/II trial
Published in Neuroscience, Genetics & Genomics, and Pharmacy & Pharmacology
Tay-Sachs disease was described in 1881 by Warren Tay, an ophthalmologist who described a unique finding in the retina of infants and children, the so-called “cherry red spot”. Bernard Sachs, in 1887, united this finding with that of “arrested cerebral development”, in which infants and children with this disorder cease normal neurologic development and progressively experience a global loss of cerebral function. Early clinical descriptions highlighted the high incidence of Tay-Sachs among those of Ashkenazi Jewish ancestry. According to the Norton & Elaine Sarnoff Center for Jewish Genetics, the first Community carrier screening for Tay-Sachs carriers took place in 1971 at a synagogue in Maryland. Since that time, there has been a 90% reduction in the number of cases in the US. However, Tay-Sachs disease and the closely related Sandhoff disease cases continue to arise with tragic consequences for the affected families.
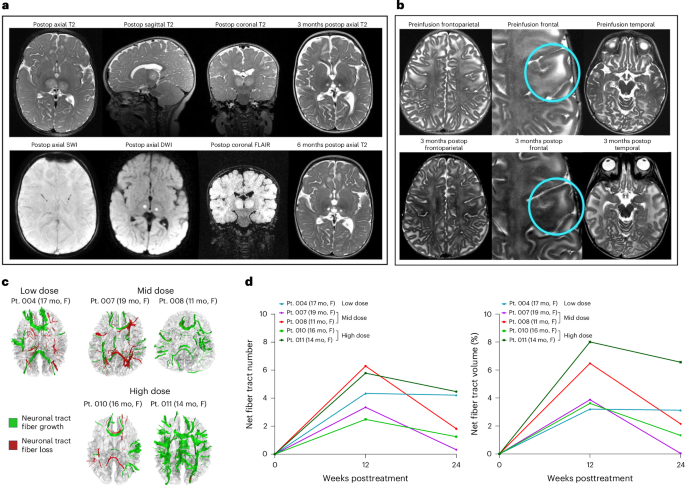
Our recent paper arose from a collaboration between Drs. Miguel Esteves, Heather Gray-Edwards, Oguz Cataltepe, myself and other research labs at UMass Chan Medical School, and Dr. Florian Eichler at Massachusetts General Hospital. Drs. Esteves and Gray-Edwards developed a dual adeno-associated virus (AAV) based approach to correction of defects in the two subunits of the beta-hexosamindase A enzyme which is responsible for breaking down GM2 gangliosides. The genes for the alpha and beta subunits of this enzyme are HEXA and HEXB. Biallelic defects in HEXA result in Tay-Sachs disease while defects in HEXB result in Sandhoff disease. The challenge for gene therapy in these conditions is the global nature of CNS dysfunction. Some benefit can be gained because of cross-correction, where lysosomal enzymes secreted from a highly expressing corrected cell can export a portion of the enzyme made in that cell to neighboring cells. However, this requires over-expression of both the HEXA and HEXB genes in the same cell. Furthermore, cross-correction alone is not sufficient to effectively deliver functional enzyme to the huge number of neurons requiring correction throughout the brain. While intravenous (IV) AAV gene therapy has the potential to achieve global correction of the CNS, the currently available vectors cross the blood-brain barrier (BBB) inefficiently and pushing the doses of IV AAV too high has resulted in severe adverse events and deaths in patients with other genetic diseases.
The approach that our group took was to take advantage of the broad connectivity of the thalamus to the entire cerebral hemisphere. In previous work, Drs. Esteves, Gray-Edwards and Martin showed that directed injection of AAV vectors expressing HEXA and HEXB genes into the thalamus of genetic models of the disease resulted in high levels of expression of the functional enzyme, which then underwent anterograde axonal transport out to the entire ipsilateral hemisphere. This intrathalamic infusion technique was adapted to human infants in work that was published in Nature Medicine in 2022. That patient showed a clear increase in beta-hexosaminidase A levels in the CSF. The current study continued that effort in a 9-patient dose escalation trial. CSF enzyme activity progressively increased over the 4 dosage levels, GM2 levels were reduced, myelination improved and macrocephaly was ameliorated. At the highest dose level, patients were noted to have improved oral feeding and fewer less severe seizures, both important endpoints for patient quality of life. Overall, however, the patients continued experiencing major neurologic declines over time, indicating that this dual-vector approach was not going to be sufficient. A next generation bicistronic AAV vector (expressing both HEXA and HEXB genes) has been developed by the Esteves and Gray-Edwards labs. Preparations for a first-in-human trial with that vector are ongoing.
The broader question remains as to how one might achieve global CNS delivery of a therapeutic transgene. Newly modified AAV capsids could achieve sufficient BBB penetration to be efficacious in treating global CNS defects. One serious caution has arisen from a recently described fatal toxicity in a trial of a BBB-crossing AAV capsid. In that context, it is possible that the bilateral intrathalamic approach used in this study, while not efficient enough to treat Tay-Sachs disease patients could still prove useful in other genetic neurologic disorders.
Follow the Topic
-
Nature Medicine
This journal encompasses original research ranging from new concepts in human biology and disease pathogenesis to new therapeutic modalities and drug development, to all phases of clinical work, as well as innovative technologies aimed at improving human health.
Related Collections
With Collections, you can get published faster and increase your visibility.
Clinical Research in Respiratory Medicine
Publishing Model: Hybrid
Deadline: Feb 18, 2027
Microbiome and energy metabolism
Publishing Model: Hybrid
Deadline: Dec 06, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in