Gut microbiota and metabolome among pregnant women with HIV and their infants
Published in Microbiology, Protocols & Methods, and General & Internal Medicine
In June 2016, our study team in Pune, India, began the PRACHITi longitudinal cohort study (Pregnancy Associated Changes in Tuberculosis Immunology) with the goal of understanding how stages of pregnancy and postpartum affect immunity in women living with and without HIV. Nested within this cohort was a secondary analysis related to the following question: how does maternal HIV status affect the gut microbiome and plasma metabolome in mothers throughout pregnancy and postpartum, and in their uninfected infants? Our paper recently published in Nature Communications addresses this research question.
Gut microbiome research is rapidly growing in low- and middle-income countries (LMICs) like India, where maternal and child morbidity and mortality remain high. We hoped that examining the microbiome and metabolome, separately and together, could provide insights on why uninfected children born to mothers with HIV face higher rates of adverse birth and child health outcomes. These findings may inform new prevention and treatment strategies to healthier pregnancies, improving birth outcomes, and promoting early childhood growth and development.
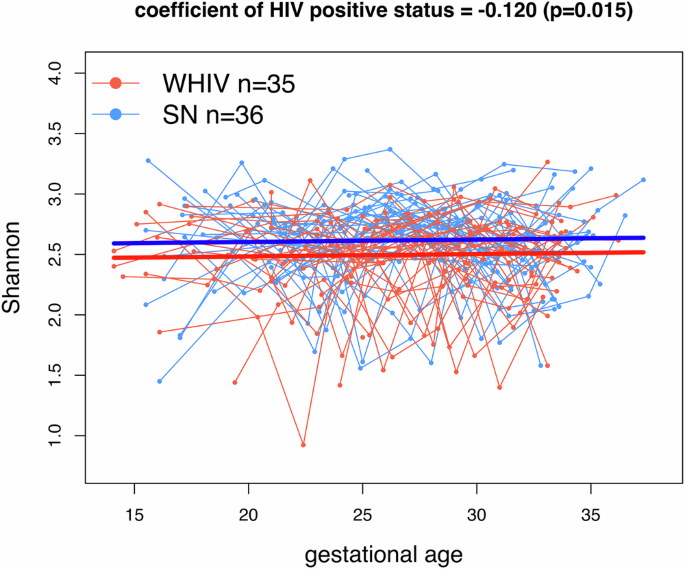
During data analyses, we observed several interesting findings. We had initially hypothesized that mothers with HIV would have a more variable/less consistent microbiota α-diversity over the course of gestation compared to mothers without HIV. However, our results, using intensive longitudinal sampling during pregnancy, showed that α-diversity was consistent through the second and third trimester for both mothers with and without HIV, although mothers with HIV had consistently lower α-diversity compared to mothers without HIV.
Beyond α-diversity, there were also interesting findings related to taxa abundance. Analyzing microbiome data, however, comes with unique challenges. Microbial communities are highly variable between individuals and often containing rare species or “structural zeros” (bacteria present in one group but completely absent in the other). To compare differences between the two HIV groups, we used a statistical method called Analysis of Compositions of Microbiomes with Bias Correction (ANCOM-BC). This analytical approach addresses the compositional nature of microbiome data and seeks to identify bacteria that are different between groups. We noted several differences, some as differential abundance and others as structural zeros, between mothers with and without HIV during pregnancy and postpartum, with differences also noted in their children. Importantly, some of these bacteria have been linked to the adverse outcomes experienced by mothers with HIV and their children, suggesting potential connections between HIV status, microbes, and clinical outcomes.
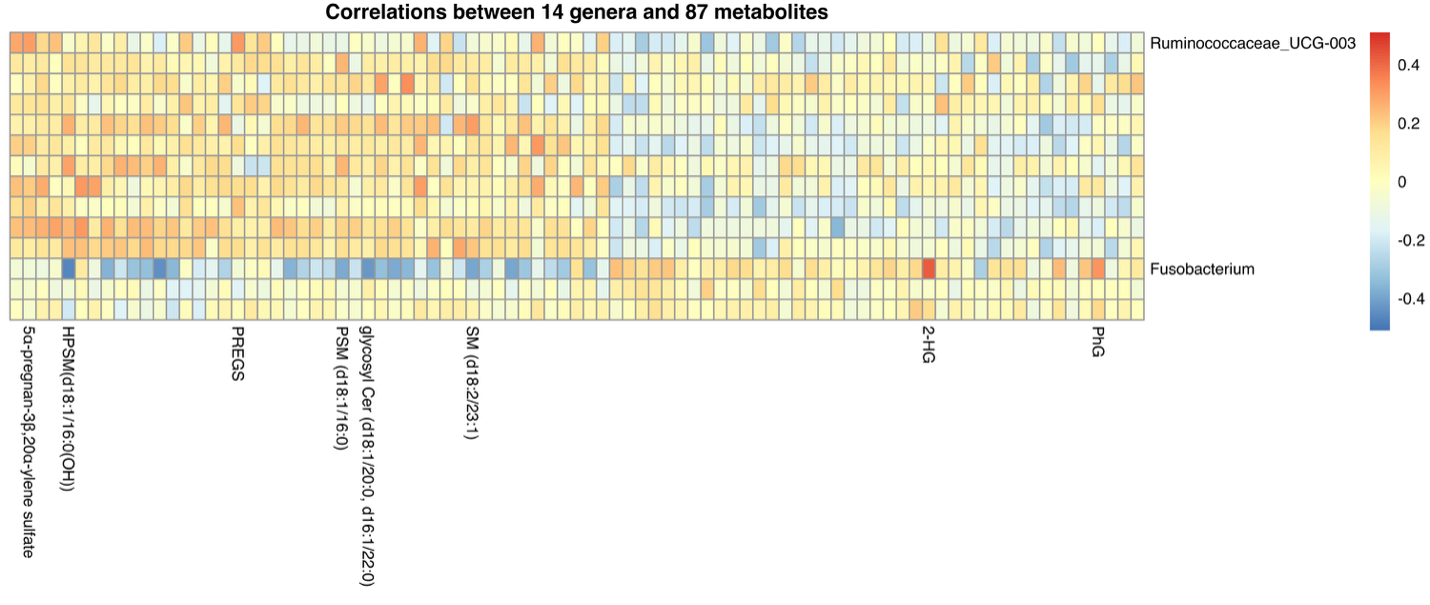
We also conducted a third trimester metabolomics analysis among third trimester plasma samples, and observed several differences by maternal HIV status, with some metabolites also linked to adverse maternal-infant outcomes. Based on helpful suggestions by reviewers, we also assessed how changes in the gut microbiome could be linked to the systemic metabolome. We looked at the correlation between microbes and metabolites (figure below) to try to better understand their connections, and whether these interactions may help explain adverse clinical outcomes. This new analysis broadened the scope of our manuscript and led to additional findings that helped us better understand the connection between the gut microbiome and systemic metabolome in pregnant women with and without HIV.
In summary, our study provides important insights and implications for improving maternal and infant health outcomes. By exploring both the microbiome and metabolome, and their connection in HIV-affected populations during pregnancy, postpartum and infancy, we highlight possible new directions for research and potential interventions such as probiotics or prebiotics to modulate the microbiota that could ultimately improve maternal-child health outcomes. Yet, our study also identified gaps that remain in our understanding of how the microbiome and metabolome, and their interactions influence the health of populations affected by HIV. We hope that future efforts continue to explore these complex interactions and identify potential interventions. Studies like PRACHITi can help further encourage filling the gaps in global maternal and child health. Seeing this work published has been both rewarding and meaningful for our team, as it represents years of collaboration between Indo-US scientists from multiple institutions, the extensive efforts of the study team in data collection, NIH funding support, and, especially, the dedication and efforts of the study staff and study participant. Together, these contributions have helped generate and relay important findings and provide suggestions for the next steps in this field of research.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in