Harvesting Safe Water from Air: A Bioinspired Journey from Lab to Life
Published in Materials
As researchers working at the intersection of materials science and environmental engineering, we’ve always been fascinated by a simple yet profound question: How can we sustainably produce clean, safe drinking water in places where it’s most needed?
Atmospheric water harvesting (AWH), the process of capturing water vapor from air offers a promising solution, especially in arid or resource-limited regions. But there’s a hidden challenge: the air isn’t just carrying water vapor; it’s also full of microbes, including harmful bacteria like E. coli and S. aureus. These pathogens can be absorbed together with moisture, posing a contamination risk to the harvested water unless effectively inactivated.
In our recent study published in Nature Water, we set out to tackle this dual challenge: not only harvesting water efficiently from the air, but also ensuring it’s free from harmful bacteria, safe enough even for cell cultures and living organisms.
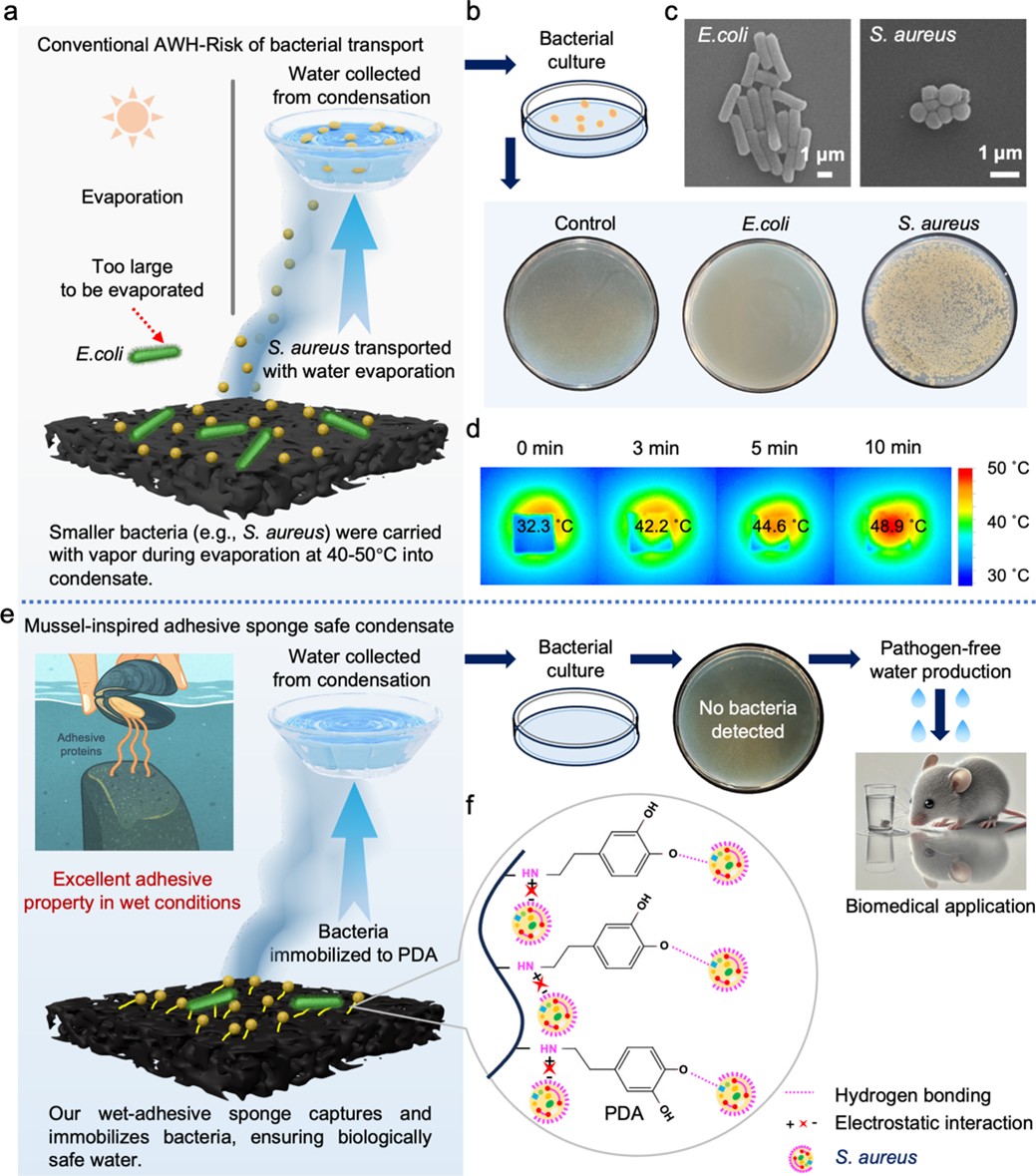
Fig.1 Comparative evaluation of conventional AWH and mussel-inspired PDA sponge for pathogen-free water harvesting. a, Schematic of bacterial transport during AWH. b, Pathogen assessment of AWH-collected water. c, SEM images of E. coli (left) and S. aureus (right). Scale bars, 1 µm. d, Thermal images of a conventional AWH sample following water uptake, illustrating a surface temperature rise from 32.3 °C to 48.9 °C over 10 min under one-sun irradiation. e, Schematic of the mussel-inspired PDA sponge capturing bacteria for pathogen-free water harvesting. f, Mechanism of bacterial capture by PDA-coated sponge under AWH conditions.
The Inspiration: Mussels and Sunlight
Our journey began with two unlikely inspirations: mussels and sunlight.
Mussels cling tightly to wet, slippery surfaces thanks to a special protein rich in catechol groups. This “wet adhesion” ability inspired us to use polydopamine (PDA), a synthetic polymer that mimics mussel proteins, to create a material that could “grab” and immobilize bacteria during water capture.
Meanwhile, we knew that sunlight could be more than just a source of energy, it could also be a disinfectant. Photothermal materials convert light into heat, and that heat can kill bacteria without chemicals. Could we combine these two ideas into one material?
Designing the “Smart” Aerogel
We developed a novel aerogel, a lightweight, porous material, made from sodium alginate (a natural polymer from seaweed), carbon black (for absorbing sunlight), polydopamine (our mussel-inspired adhesive), and lithium chloride (a salt that absorbs moisture from the air).
The result was a material that doesn’t just passively absorb water; it actively captures and neutralizes bacteria. The PDA coating acts like a sticky net, trapping bacteria on its surface. When sunlight hits the aerogel, the carbon black and PDA work together to rapidly heat up, killing over 90% of captured bacteria within minutes.
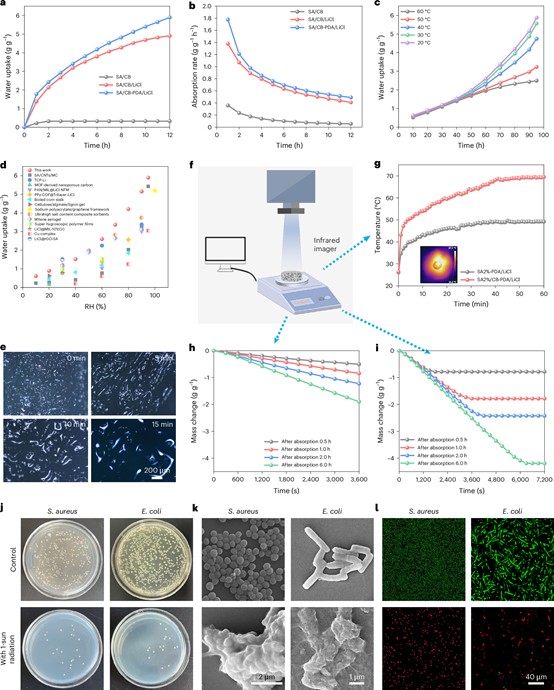
Fig. 2 Water adsorption, desorption, and antibacterial performance of SA/CB-PDA/LiCl aerogels. a, Dynamic water sorption process of SA/CB, SA/CB/LiCl, and SA/CB-PDA/LiCl aerogels with salt contents of 30% at 25 °C and 95% RH. b, Water absorption rate of SA/CB, SA/CB/LiCl, and SA/CB-PDA/LiCl aerogels with salt contents of 30% at 25 °C and 95% RH. c, Water absorption isotherms of SA/CB-PDA/LiCl aerogels at different temperatures. d, Comparison of the adsorption capacity at various RH of SA/CB-PDA/LiCl aerogels with those of several reported AWH materials. e, Optical images of the water droplets growth, coalescence, and migration in SA/CB-PDA/LiCl aerogels. Scale bar: 200 µm. Photograph of the SA/CB-PDA/LiCl aerogels. f, Schematic diagram of the setup for the water evaporation performance test. g, Time-temperature curves of SA-PDA@LiCl aerogel and SA/CB-PDA/LiCl aerogels at 24 °C and 70% RH under 1.0 Sun irradiation. h, i, The desorption processes of SA-PDA/LiCl and SA/CB-PDA/LiCl aerogels at 24 °C and 70% RH under 1.0 Sun irradiation. j, The images of bacterial colonies on the agar plates. k, SEM images of S. aureus and E. coli of co-culture with different hydrogels for 4 h. l, Confocal images of S. aureus and E. coli co-incubation with different hydrogels by using SYT09/PI fluorescent staining.
From Lab Bench to Real World
In the lab, our aerogel showed impressive numbers: it could absorb up to 6 grams of water per gram of material at high humidity, and release it quickly under sunlight. But we wanted to go further to test not just if the water was clean, but if it was truly safe.
We collected water produced by our aerogel in a custom-built hybrid device that runs on solar, wind, and electric power. We then used this harvested water for human cell culture. Not only did the cells survive, they thrived, with over 90% viability after a week.
The ultimate test came when we gave this water to live rats for two weeks. Their organs showed no signs of damage, and their behavior and weight remained normal. This was a critical milestone: we had produced water from air that was not just drinkable, but biocompatible.
Why This Matters
In many parts of the world, clean water isn’t just scarce, it’s also a vector for disease. Traditional water purification often relies on chemicals like chlorine, which can leave residues, promote antibiotic resistance, or be unavailable in remote areas.
Our approach offers a chemical-free, energy-efficient alternative. The aerogel is scalable, made from low-cost materials, and functions even in arid conditions. It’s a step toward decentralized, sustainable water solutions for communities, disaster relief, remote healthcare, and even laboratory settings where ultra-pure water is essential.
Behind the Scenes: Challenges and Teamwork
This project was a true interdisciplinary effort. Materials scientists, chemists, biomedical researchers, and engineers all collaborated to move from concept to prototype to validation. There were challenges, optimizing the pore structure to prevent salt leakage, fine-tuning the photothermal response, and ensuring the aerogel remained stable through dozens of cycles.
One memorable moment was watching the first droplets of water condense in our outdoor device under the Singapore sun. It was a simple yet powerful reminder of why we do this work: to turn science into something that can tangibly improve lives.
Looking Forward
While our study demonstrates a promising proof-of-concept, the journey isn’t over. We’re now exploring how this technology performs with mixed bacterial communities, how it scales for larger modules, and how it integrates into real-world water systems.
We hope this work inspires more innovation at the nexus of sustainability, materials design, and public health, because access to safe water isn’t just a scientific challenge; it’s a human right.
If you’re interested in the details, you can read our full paper here (https://www.nature.com/articles/s44221-026-00592-2).
Acknowledgments
This work was made possible by funding from the China Scholarship Council, Heilongjiang Provincial Natural Science Foundation, National Natural Science Foundation of China, and Singapore’s Ministry of Education. We are grateful to our collaborators and institutions for their support.
Author:
Feng Cheng, Hongbin Li, and the research team
Correspondence to: Lin Yang and Swee Ching Tan
Follow the Topic
-
Nature Water
This journal publishes research on the evolving relationship between society and water resources on a monthly basis. It covers the natural sciences, engineering, and social sciences, with a particular interest in regards to interdisciplinary research.
Related Collections
With Collections, you can get published faster and increase your visibility.
Remote sensing and forests
Publishing Model: Hybrid
Deadline: Jul 31, 2026
Tropical cyclones impacts and preparedness
Publishing Model: Hybrid
Deadline: Jun 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in