How Water Drives Deep Oxidation in Copper
Published in Materials
In this research, we focused on copper oxidation. Copper is widely used in electronics, catalysis, and energy technologies, and its surface structure and chemistry are well understood. At the same time, it responds strongly to its environment, making it an ideal system for studying how water influences oxidation processes.
A Simple Question That Isn’t Simple
At first glance, oxidation in humid air seems easy to explain: oxygen reacts with the metal, and water accelerates the process. However, when oxygen and water are both present, their reaction pathways overlap, which makes it difficult to determine whether water is merely present during oxidation or actively changing how it proceeds.To disentangle these effects, we compared copper oxidation under two controlled environments: dry oxygen and a mixture of oxygen and water vapor.
Watching Oxidation as It Happens
Understanding this process required combining multiple techniques, because no single method can capture the full picture. We used environmental transmission electron microscopy (ETEM) to observe copper surfaces while they reacted with gases at high temperature — essentially filming oxidation in real time. We complemented this with reactive force field molecular dynamics simulations, which track how atoms move and react over time, and density functional theory calculations, which explain why certain chemical species are stable at the interface. Together, these approaches allowed us to determine not only what happens, but also how and why.
Two Very Different Outcomes
As shown in Figure 1, oxidation proceeds differently under dry oxygen and water vapor. Under dry oxygen, oxidation remained largely confined to the surface. Oxide formation occurred mainly at step edges, where atoms are less tightly bound and more reactive. The process was relatively orderly, and the interface between metal and oxide stayed structured. When water vapor was introduced, the behavior changed dramatically. Oxidation began to penetrate Cu beneath the surface, and the interface became rough and disordered. In microscopy images, the oxide appeared to invade the metal rather than simply forming a surface layer. Simulations showed increased movement of copper atoms and easier incorporation of oxygen below the surface.These observations indicated that water fundamentally alters the oxidation mechanism.
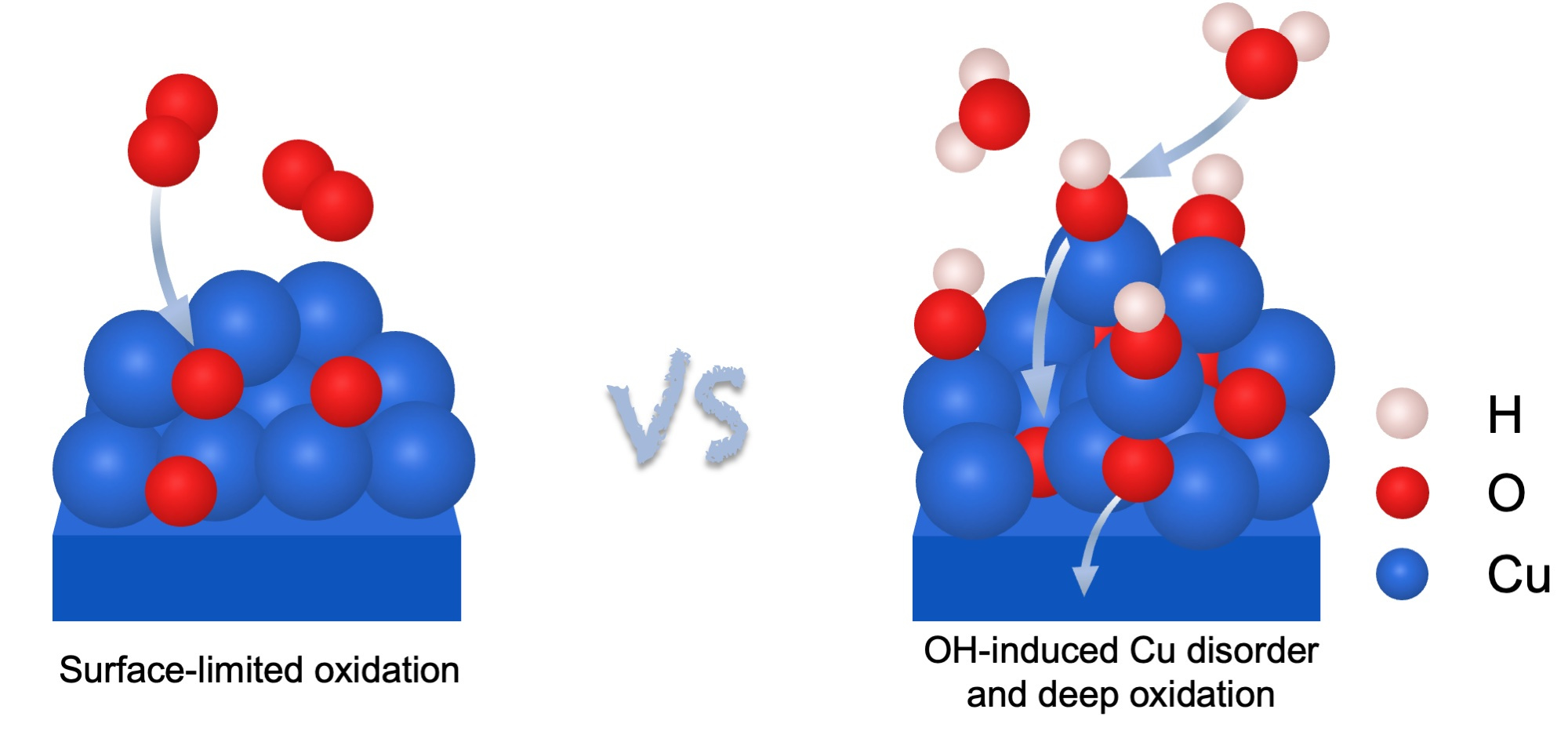 Figure 1. Comparison of the oxidation processes under dry oxygen and water vapor.
Figure 1. Comparison of the oxidation processes under dry oxygen and water vapor.
The Key Role of Hydroxyl
The crucial factor turned out not to be water itself, but hydroxyl (OH), formed when water molecules dissociate on the copper surface. OH groups accumulated in disordered regions where metal and oxide met. There, they interacted strongly with copper atoms, disturbing the surface structure and exposing more reactive sites. This disruption made it easier for oxygen to move into the subsurface region. In effect, OH groups loosened the interface, transforming a surface-limited reaction into one that enables deeper oxidation.
A Self-Reinforcing Process
Our calculations suggested an additional feedback mechanism. Initial oxidation creates mixed metal–oxide regions that stabilize OH species. These stabilized OH groups then promote further atomic displacement and roughening, which in turn facilitates deeper oxygen penetration. Once initiated, this cycle can sustain continued oxidation. One of the most encouraging aspects of the study was that different methods — experiments, simulations, and electronic calculations — all pointed to the same mechanism.
Why It Matters
Although our work focused on copper, the implications extend beyond a single material. In real environments, oxidation rarely occurs under perfectly dry conditions. Water vapor is almost always present, whether in air, industrial reactors, or electrochemical systems.
If OH species destabilize interfaces in other materials as they do in copper, they may play a key role in corrosion, catalysis, and energy technologies more broadly. Understanding this mechanism could help scientists design materials that perform better and last longer in humid environments.
The Take-Home Message
The central insight of our work is simple but important:
Water changes oxidation not just by supplying oxygen, but by reshaping the interface through hydroxyl groups.
In the presence of water, the copper surface becomes more disordered, more mobile, and more vulnerable to deep oxidation. What might otherwise remain a surface reaction can evolve into a subsurface invasion.
This study highlights how even the most familiar substance — water — can profoundly influence material behavior at the smallest scales, reminding us that complex processes often depend on subtle interactions we cannot see directly.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in