Profound sympathetic neuropathy in the bone marrow of patients with acute myeloid leukemia
Published in Cancer and Anatomy & Physiology

Acute myeloid leukemia (AML) is an aggressive hematologic malignancy and the most common acute leukemia in adults, with its occurrence increasing with age. Although AML is relatively rare among all cancers (~1.1 % of cancer diagnoses), the 5-year overall survival rate is only 21% [1]. This rate positions AML similarly to highly lethal solid malignancies like liver, lung, and brain cancers.
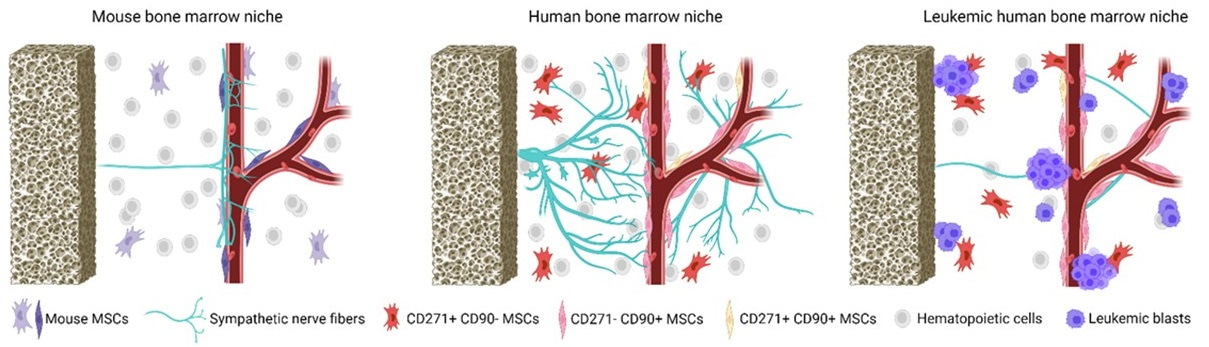
The expansion of leukemic cells is associated with the impairment of normal hematopoiesis that disturbs balanced blood cell production. In addition, recent studies suggested that leukemic cells alter the bone marrow (BM) microenvironment, known as the BM niche, a complex three-dimensional (3D) structure that provides factors necessary for the maintenance, survival, and differentiation of healthy hematopoietic cells [2]. Detailed characterizations of healthy and leukemic BM niches have been primarily conducted in mouse models. Different cell types, including mesenchymal stromal cells (MSCs), endothelial cells, macrophages, lymphocytes, megakaryocytes, and osteoblasts, play an important role in the regulation of hematopoiesis [3]. In addition, sensory and sympathetic nerve fibers (SNF) penetrating the BM were described for the first time in animals more than 50 years ago [4]. The recent development of 3D imaging suggests that the sympathetic nervous system regulates balanced blood cell production [5], cell migration [6], and regeneration of hematopoiesis in response to genotoxic stresses [7] in murine models.
Despite a detailed characterization of the murine BM environment, the human BM niche remains poorly investigated. Whereas all bones support hematopoiesis in mice, the axial skeleton is the major hematopoietic site in humans. The hematopoietic (red) marrow of long human bones is progressively replaced in adolescence by adipocytes, which reduces the hematopoietic activity of BM, except for the proximal regions of the long bones. Therefore, it is still being determined if all HSC niche components in mouse BM are also relevant for the human BM niche. Most studies of human stromal BM cells have been performed after ex-vivo expansion; however, this strategy poorly reflects the in vivo cell interaction.
The loss of information about human 3D architecture is explained by the difficulty of obtaining BM biopsies, as many clinics have shifted from performing trephine biopsies for diagnosis to primarily using BM aspirates. This lack of important information about the intact human BM niche inspired our work. For the first time in human biopsy material, we aimed to take a closer look at the 3D architecture of the human BM niche, including MSC distribution and sympathetic innervation in intact BM and BM of AML patients at diagnosis, during, and after cytotoxic therapy (CT).
3D imaging and analysis
To visualize 3D niche architecture, we used 2-photon multicolor confocal microscopy that allowed us to penetrate into samples up to 300 µm deep. Initially, we examined MSC localization in control BM samples from patients without diseases affecting the bone marrow. Notably, no consensus exists on unique and definitive cellular markers for MSC identification. While some researchers consider CD271 a pan-MSC marker [8], others propose that most MSCs express CD90 [9]. Our combined staining with anti-CD90 and CD271 antibodies showed that CD271+ CD90+ MSCs localize along the long cylindrical structures typical for BM vessels. At the same time, most CD271+ CD90- cells are irregularly distributed in cellular parenchyma (Fig.1). Therefore, we propose using CD271 as a pan-MSC marker, while CD90 most probably defines perivascular cells.
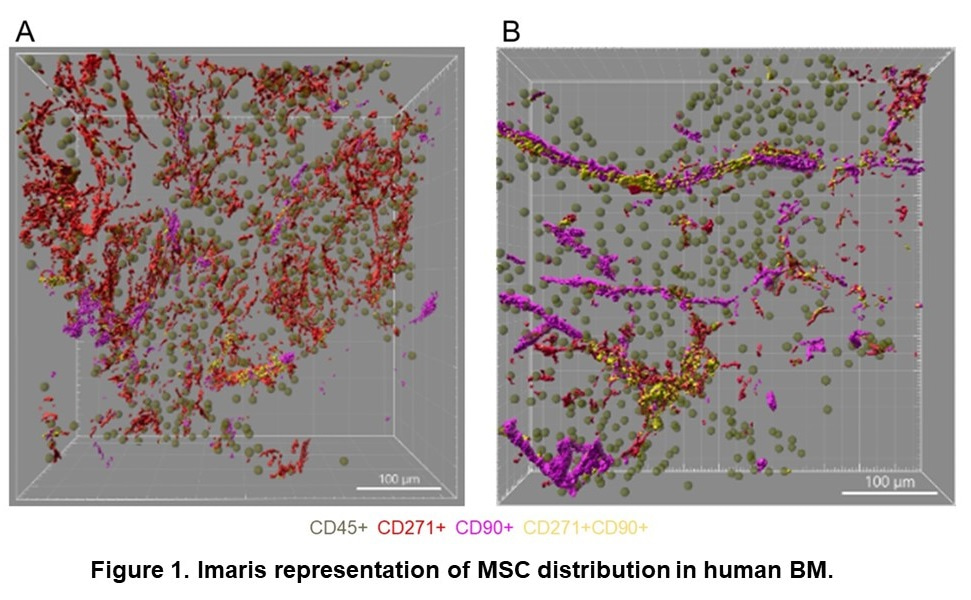
Next, we investigated the distribution of functional sympathetic nerve fibers (SNF) using staining against tyrosine hydroxylase: the rate-limiting enzyme in the metabolic pathway leading to the synthesis of dopamine and noradrenaline by sympathetic neurons. In contrast to the corkscrew-shaped representation observed in murine BM [5,7,10], TH+ SNFs disperse irregularly within the parenchyma without wrapping around blood vessels (Fig. 2A).
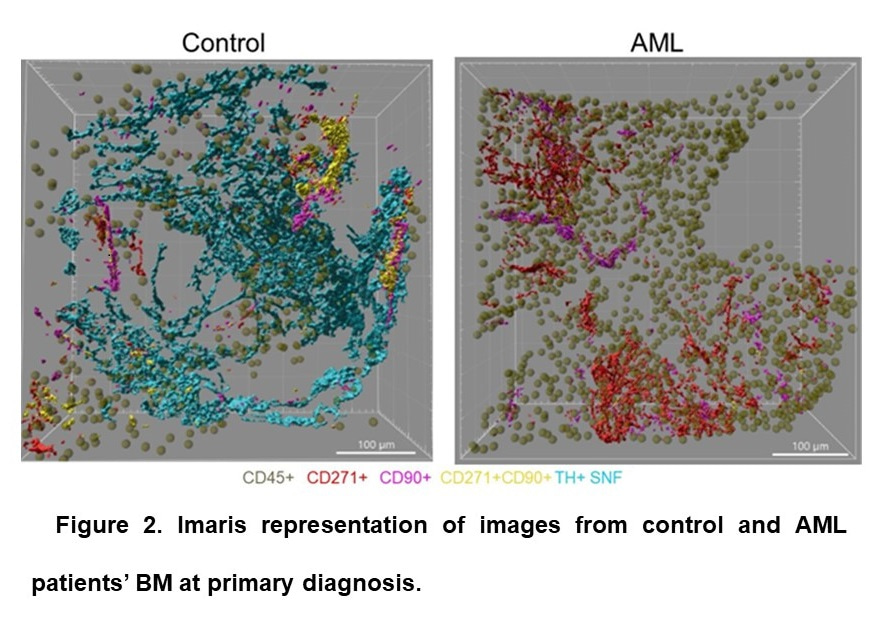
Subsequently, we compared control and leukemic BM from 25 AML patients to assess changes in MSC and SNF density in AML patient BM at diagnosis, during, and after cytotoxic therapy. Although the density and composition of MSC populations were not changed in the BM of AML patients, the density of functional TH+ SNFs was dramatically reduced in patients already at the primary AML diagnosis compared to controls, further reduced after CT, and it did not restore even seven months after CT.
Take-home message
We visualized MSCs and functional TH+ SNFs in human BM for the first time in human biopsy material. Our findings revealed a dramatic loss of sympathetic nerves at the primary diagnosis of AML, with their further destruction due to cytotoxic therapy. Moreover, sympathetic neuropathy persisted for more than seven months after induction therapy, which could lead to prolonged hematological dysfunction. Therefore, protecting or restoring sympathetic nerve fibers in human BM during or after cytotoxic treatment could be a therapeutic goal for acute myeloid leukemia patients.
Moreover, exploring whether these observed changes are exclusive to AML or common across other hematological malignancies (e.g., acute lymphoblastic leukemia, chronic myelogenous and myelomonocytic leukemias, etc) is intriguing. Discoveries on other blood disorders will help us to understand the potential shared or unique impacts of various leukemias on human BM niche architecture and will facilitate the development of new therapy strategies.
- Canadian Cancer Statistics Advisory Commitee Canadian Cancer Statistics. 2019. Available online: ca/Canadian-Cancer-Statistics-2019-EN
- Méndez-Ferrer S, Bonnet D, Steensma DP, et al. Bone marrow niches in haematological malignancies. Nat. Rev. Cancer. 2020; 20:285–298
- Mendelson A, Frenette P. Hematopoietic stem cell niche maintenance during homeostasis and regeneration. Nat Med. 2014;20:833–846.
- Calvo W. The Innervation of the Bone Marrow in Laboratory Animals. American Journal of Anatomy. 1968; 123(2):315-28.
- Maryanovich M, Zahalka AH, Pierce H, et al. Adrenergic nerve degeneration in bone marrow drives aging of the hematopoietic stem cell niche. Med. 2018;24(6):782–791.
- Katayama Y, Battista M, Kao WM, et al. Signals from the sympathetic nervous system regulate hematopoietic stem cell egress from bone marrow. Cell. 2006;124(2):407–421.
- Lucas D, Scheiermann C, Chow A, et al. Chemotherapy-induced bone marrow nerve injury impairs hematopoietic regeneration. Med. 2013;19(6):695–703.
- Quirici N, Soligo D, Bossolasco P, Servida F, Lumini C, Deliliers GL. Isolation of bone marrow mesenchymal stem cells by anti-nerve growth factor receptor antibodies. Exp Hematol. 2002; 30(7):783-91.
- Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006; 8(4):315-7.
- Hanoun M, Zhang D, Mizoguchi T, et al. Acute myelogenous leukemia-induced sympathetic neuropathy promotes malignancy in an altered hematopoietic stem cell niche. Cell Stem Cell. 2014;15(3):365–375.
Follow the Topic
-
Leukemia
This journal publishes high quality, peer reviewed research that covers all aspects of the research and treatment of leukemia and allied diseases. Topics of interest include oncogenes, growth factors, stem cells, leukemia genomics, cell cycle, signal transduction and molecular targets for therapy.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in