Sonoluminescence and Crystallic Fusion – A brief note on converting sound into light.
Published in Bioengineering & Biotechnology

Having maintained a record of articles here for a while now, I am adding yet another brief opinion to this ongoing writing collection; this time on sonoluminescence and crystallic fusion in nature and in life sciences. The phenomenon of sonoluminescence [Hilgenfeldt, 1999] is incidentally known as the unrecognized secret behind nitroglycerine explosions, which spelled major success in 1865 at Dynamit Nobel A.G.; a chemical company funded by Alfred Nobel himself. Based on this observation, in recent years physicist Kirk McDonald wryly remarked that all Nobel prizes are therefore due to sonoluminescence [McDonald 1998]. In its mechanism-of-action, the process can be described relative to the explosion of nitroglycerine, where a pressure wave is shaped in the compound by dropping it, leading to the formation of cavitation bubbles that collapsed upon interaction with the reflected pressure wave, to emit a flash of light lasting picoseconds – in a process that converts sound into light (Movie 1).
![Having maintained a record of articles here for awhile now, I am adding yet another brief opinion to this ongoing writing collection; this time on sonoluminescence and crystallic fusion in nature and in life sciences. The phenomenon of sonoluminescence [Hilgenfeldt, 1999] is incidentally known as the unrecognized secret behind nitroglycerine explosions, which spelled major success in 1865 at Dynamit Nobel A.G.; a chemical company funded by Alfred Nobel himself. Based on this observation, in recent years physicist Kirk McDonald wryly remarked that all Nobel prizes are therefore due to sonoluminescence [McDonald 1998]. In its mechanism-of-action, the process can be described relative to the explosion of nitroglycerine, where a pressure wave is shaped in the compound by dropping it, leading to the formation of a cavitation bubble that collapsed upon interaction with the reflected pressure wave, to emit a flash of light lasting picoseconds – in a process that converts sound into light.](https://images.zapnito.com/cdn-cgi/image/metadata=copyright,fit=scale-down,format=auto,quality=95/https://images.zapnito.com/uploads/aJe9wKbsRaeqficxMY1x_movie_1.gif)
Crystallic fusion also emits light, although in this instance it occurs when a freezing pyroelectric crystal is heated to generate a high voltage and produce ‘nuclear fusion’ on a tabletop at room temperature (Movie 2). The focused field can accelerate a beam of deuterium ions to about 1 percent the speed of light for sustained fusion [Peplow 2005]. Such crystals can also give off X-rays as it accelerates electrons to create a tiny radioactive source with interesting potential applications in the life sciences. This post briefly combines the interdisciplinary concepts of materials chemistry and applied physics to explore applications in the life sciences.

Movie 2: When a lithium niobate crystal is heated it accelerates electrons to energies in excess of 40 keV, to make the patterns seen in the movie. Image courtesy of: Acoustics Research, Physics, UCLA, Link: https://acoustics-research.physics.ucla.edu/crystallic-fusion/
Sonoluminescence in the lab and in nature
In 1934, Hermann Frenzel and colleagues first technically observed sonoluminescence in the lab at the University of Cologne, when they added an ultrasound transducer in a tank of photographic developer fluid [Frenzel 1934]. The process serendipitously led to the formation of tiny dot-like bubbles in the film that emitted light in the presence of active ultrasound. This event is known as multi-bubble sonoluminescence. The remarkable properties of the phenomenon include the transduction of acoustic energy into light energy with a brief light emission lifetime. Applied physicist T.J. Matula demonstrated the initial cavitation and single-bubble sonoluminescence to elaborate on its mechanism-of-action, which occurred between the threshold of luminescence and extinction (Figure 1) [Matula 1999].
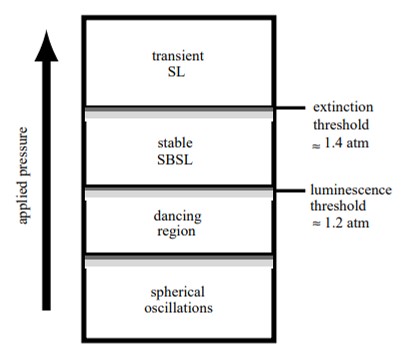
The conversion of sound to light via sonoluminescence also occurs in nature, among snapping shrimp. These organisms can produce a loud cracking noise that violently disturbs underwater communication. The phenomenon arises due to the collapse of a large cavitation bubble, originally formed as the snapper claw of shrimps shut to generate tensile forces of a high-velocity water jet. The process of bubble collapse underwater is accompanied by a short intense flash of light – like sonoluminescence, therefore termed ‘shrimpoluminescence’ [Versluis 2001]. This biological shock wave is undetected with the naked eye and is used to stun or even kill prey animals. The phenomenon of light emission highlights the extreme heat conditions of the bubble interior at collapse, to generate a biologically viable event.
But how do these concepts in applied physics and materials chemistry translate to the life sciences?
Thus far, the light emanating from sonoluminescence has a significant spectral ultraviolet component, and these bubble oscillations can be triggered by ultrasound waves. The process has scope in photopharmacology to photoactivate smart drugs with molecular photoswitches to regulate the bioactivity of light, further optimized with magnetic, fluorescent and plasmonic nanoparticles integrated for therapeutic applications [Maksymov 2022], [Sheng 2017]. For instance, sonodynamic therapy is a process in which sonoluminescence in the presence of ultrasound and ground-state molecular oxygen facilitates violent bubble collapse alongside reactive oxygen species production. The outcomes can activate classes of drugs with localized cytotoxic oxygen production, for promising applications to treat aggressive and resistant tumor cells, at locations that are hard to reach (Figure 2) [Beguin 2019], [Chen 2013].
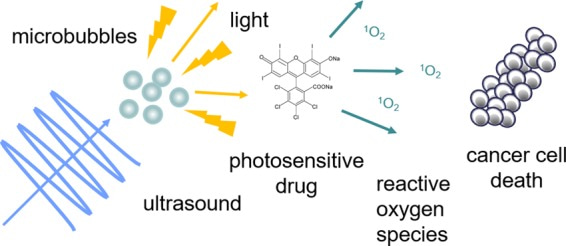
Figure 2: The activation of a photodynamic therapy agent using microbubbles and ultrasound. Ultrasound can accurately focus on larger tissue depths, opening potential for chemotherapeutic drug delivery to reach solid tissues [Beguin 2019].
From Sonoluminescence to Crystallic Fusion - Radiation on tap and a moonshot.
In 2005, inventor and materials scientist B. Naranjo et al. showed that gently heating a pyroelectric crystal of lithium tantalate in a deuterated atmosphere can generate fusion on a desktop (Figure 3)[Naranjo 2005]. While this toaster-sized device cannot function as a nuclear fusion machine at room temperature (since it does not support a self-sustaining thermonuclear reaction), the team saw its scope to treat cancer and power spacecrafts.
During its mechanism-of-action, the electrostatic field of the crystal in a deuterated environment can generate and accelerate a deuteron beam, that strikes a deuterated target to produce large-scale neutron flux for applications in a simple, palm-sized neutron generator. Since the commonly available forms of radiation therapy tend to blast healthy cells alongside cancerous cells, even at present, the applications in life sciences are relatively direct. This invention from the archives offers a mechanism for precision radiation oncology of cancer cells with potential for interventional radiotherapy at a targeted tumor area, in a potentially biocompatible and piezo-compatible contraption that can be turned on and off at will.
While the invention remains to be fully explored in medicine, it lays the groundwork for several technological spinoffs. The concept of crystallic fusion is also applicable beyond life sciences in rocket propulsion as ion engines that eject a stream of charged xenon gas to propel forward as seen during the European Space Agency’s SMART-1 mission to the moon that successfully demonstrated advanced ion propulsion for navigation [Foing 2006].
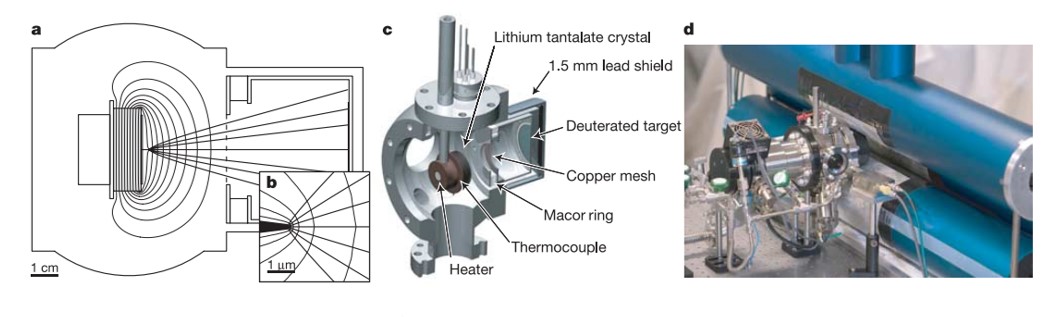
Figure 3: Experimental geometry. a) Calculated equipotentials and trajectories for a crystal charged to 100 kV., b) Same trajectories shown near the tip., c) Vacuum chamber cut-away view., d) Arrangement of neutron and X-ray detectors [Naranjo 2005].
Then vs. Now – The outlook
The concepts of pyroelectricity and pyroelectric crystals are not new, having lasted for centuries [Lang 2013], with modern applications of pyroelectric imaging relevant across diverse fields [Kulwicki 1992]. In 1898, W. Voigt noted that the electrification of certain crystals such as tourmaline by heating (pyroelectricity) or their electrification by deformation (piezoelectricity) are relatively similar phenomena [Franklin 1899]. The Curie brothers historically discovered piezoelectricity in March 1880. Fast-forward to the present timeline, the concept is suited for a variety of medical applications such as implants, sensors, and bioinspired constructs compatible with piezoelectric biological structures in vivo [Zaszczynska 2020].
Biological tissue with piezoelectric properties includes actin and myosin structures of muscle, keratin of hair, collagen in tendons, skin, and bones, as well as elastin in living tissue (Table 1). The capacity to develop piezocompatible/biocompatible devices will suit better integration of devices, as already seen with bone and cartilage regeneration in vivo [Rodriguez 2016]. Despite the varying nature of piezoelectricity in the human body, many research efforts aim to understand the underlying biomechanics to bioengineer possible interventional solutions to address disease conditions and accelerate the improvement of human health [Chen-Glasser 2018].
Table 1: Tissues with piezoelectric properties and the driving source of piezoelectricity [Chen-Glasser 2018]
|
Organ |
Piezoelectric molecule |
|
Muscle |
Actin and myosin |
|
Hair |
Keratin |
|
Bone |
Collagen |
|
Tendon |
Collagen |
|
Lung tissue |
Elastin |
|
Skin (dermis) |
Collagen |
|
Skin (epidermis) |
Keratin |
|
Breast tissue |
Collagen |
|
Outer hair cell |
Prestin |
Afterword
Discoveries and inventions of the past can in this way continue to influence scientific advances at present and optimize research outcomes in the future. This article simply explores the existing possibilities to advance medicine relative to the broader concepts of crystallic fusion, piezoelectricity, and sonoluminescence that converts sound to light, to highlight the interdisciplinarity of life sciences.
Header Image: Sonoluminescence - Cavitation hots up, Nature, doi: https://doi.org/10.1038/434033a
References
- Hilgenfeldt S. et al. A simple explanation of light emission in sonoluminescence, Nature, doi: https://doi.org/10.1038/18842
- McDonald K. T., Nobel sonoluminescence, Joseph Henry Laboratories, Princeton University, Princeton. url: http://kirkmcd.princeton.edu/examples/popsci.pdf
- Peplow M., Crystal creates table-top fusion, Nature, doi: 10.1038/news050425-3
- Frenzel H. et al., Luminescenz im ultraschallbeschickten Wasser, Zeitschrift für Physikalische Chemie, doi: 10.1515/zpch-1934-2737
- Matula, T.J. Inertial cavitation and single–bubble sonoluminescence, Philosophical Transactions of the Royal Society A., doi: https://doi.org/10.1098/rsta.1999.0325
- Versluis M. et al. Shrimpoluminescence, Bibcode: 2001APS..DFD.EA009V
- Maksymov I et al. Gas Bubble Photonics: Manipulating Sonoluminescence Light with Fluorescent and Plasmonic Nanoparticles, MDPI., doi:
- Sheng Y. et al. Magnetically responsive microbubbles as delivery vehicles for targeted sonodynamic and antimetabolite therapy of pancreatic cancer, Journal of Controlled Release doi: 10.1016/j.jconrel.2017.07.040
- Chen H. et al. Ultrasound-targeted microbubble destruction for chemotherapeutic drug delivery to solid tumors, Journal of Therapeutic Ultrasound, doi: 10.1186/2050-5736-1-10
- Beguin E. et al. Direct Evidence of Multibubble Sonoluminescence Using Therapeutic Ultrasound and Microbubbles, ACS Applied Materials, and Interfaces, doi: 10.1021/acsami.9b07084
- Naranjo B. et al. Observation of nuclear fusion driven by a pyroelectric crystal, Nature, doi: 10.1038/nature03575
- Lang S. B. A 2400 year history of pyroelectricity: from Ancient Greece to exploration of the solar system, Taylor & Francis Online, doi: https://doi.org/10.1179/096797804225012765
- Foing B. H. et al. SMART-1 mission to the Moon: Status, first results and goals, Advances in Space Research., doi: https://doi.org/10.1016/j.asr.2005.12.016
- Kulwicki B. et al. Pyroelectric imaging, IEEE., 10.1109/ISAF.1992.300607
- Franklin W.S., Pyroelectricity and Piezoelectricity, Science, 10.1126/science.9.221.457.a
- Zaszczynska A et al. Progress in the Applications of Smart Piezoelectric Materials for Medical Devices, MDPI, 10.3390/polym12112754
- Rodriguez R. et al. Piezoelectric properties of synthetic hydroxyapatite-based organic-inorganic hydrated materials, Results in Physics, doi: 10.1016/j.rinp.2016.11.005
- Chen-Glasser M. et al. Piezoelectric Materials for Medical Applications, Intech Open, doi:10.5772/intechopen.76963



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in