The finest medical research often begins with a question at the bedside and ends with an answer in the laboratory
Published in Biomedical Research
Every paper has its origin; every scientific paper has a starting point. Our story did not begin at the lab bench, but in a hospital ward. Many years ago, among my students was a young couple. The young man was a resident in pancreatobiliary surgery. After witnessing the joys and sorrows of life in the ward, and the unbearable suffering of patients ravaged by disease, he came home every day and asked his girlfriend the same question: “Why do pancreatic cancer patients suffer such terrible pain?” For patients with pancreatic cancer, pain can be more frightening than death itself; sometimes, it can even drive people to suicide.
One of the most striking features of pancreatic ductal adenocarcinoma (PDAC) is perineural invasion, in which cancer cells spread along nerves. This process not only contributes to excruciating pain, but also provides a “highway” for tumor metastasis. Yet the mechanism behind this process remained a mystery at that time. Back then, I was only a listener to the story, not yet an investigator of this question. But those stories stayed in my heart. The faces of those patients stayed with me.
Years passed, the young man began graduate training in hepatopancreatobiliary surgery under the supervision of my senior colleague. Around the same time, the young woman joined my laboratory as a doctoral student. As their supervisor, I worked side by side with them. Together, we set out to understand what really happens when pancreatic cancer meets the peripheral nervous system.
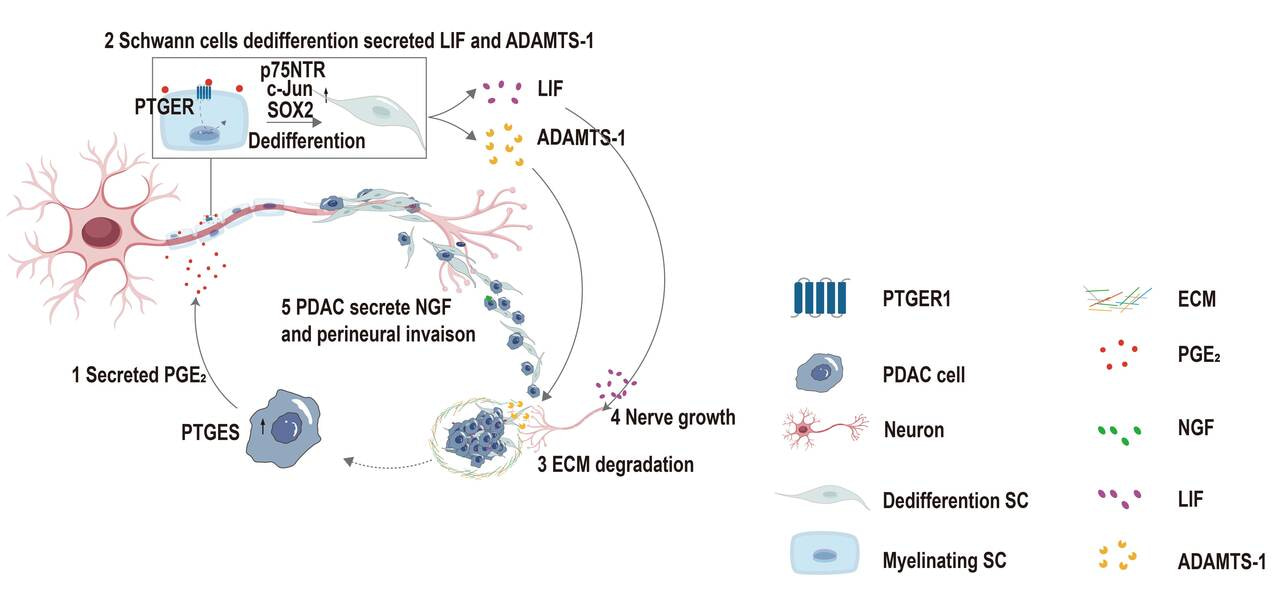
We immersed ourselves in the literature. One cell type kept appearing: Schwann cells (SCs), the glial cells that ensheath and protect peripheral nerves. Previous studies had suggested that SCs could be hijacked and reprogrammed by pancreatic cancer cells to facilitate neural invasion, but the mechanism was unclear. Gradually, a hypothesis took shape: what factor secreted by cancer cells turns SCs from guardians of nerves into accomplices of tumor progression?
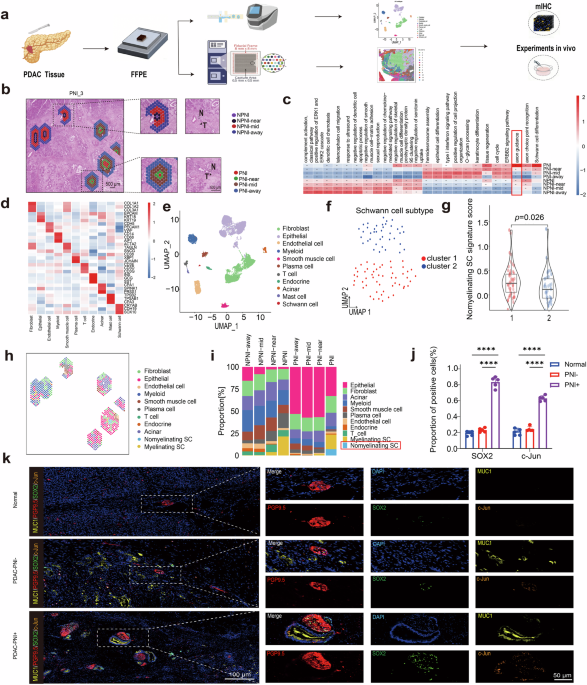
The early stages of the project were full of setbacks. Dorsal root ganglia are tiny and delicate, and our co-culture system suffered from unstable growth and inconsistent positioning. Eventually, we tried something different. Using a microinjector, we embedded dorsal root ganglia in a more controlled way, and then co-cultured them with tumor cells in time-lapse imaging dishes and an incubator system more commonly used in reproductive medicine. This allowed us to observe the interactions in real time without repeatedly disturbing the cultures.
Multi-omics analyses pointed us to one candidate pathway in pancreatic cancer cells: PTGES/PGE2. We then treated Schwann cells with PGE2 in vitro. At first, nothing changed. We adjusted the concentration, extended the treatment time, and repeated the experiments again and again, but the results remained ambiguous. Then one day, the young man called out excitedly to the young woman from the microscope: “Come quickly — the cells have changed!”Under the microscope, we saw that the Schwann cells had shifted from their original morphology into elongated, spindle-like cells. Molecular assays confirmed the upregulation of dedifferentiation markers. It was one of those moments that reminds you why scientific discovery is so compelling: we had found direct evidence that PGE2 induces Schwann cell dedifferentiation.
Once dedifferentiated, these Schwann cells began secreting higher levels of neurotrophic factors and chemokines, which in turn attracted cancer cells toward nerves and appeared to help them open paths along nerve fibers. The picture finally came into focus: PGE2 drives Schwann cell dedifferentiation, and this opens the door for perineural invasion by pancreatic cancer cells.
That discovery brought us back to the clinical question that started it all. The severe pain experienced by patients with pancreatic cancer may arise, at least in part, from this “open door”: nerves left vulnerable to direct tumor invasion and persistent inflammatory stimulation. It also suggested a possible therapeutic strategy. If we can target the PTGES/PGE2 axis, we may be able not only to relieve pain, but also to block an important route of tumor spread.
Today, the young man is both a clinician and a researcher in hepatopancreatobiliary surgery. When he walks into the ward and sees patients suffering from pancreatic cancer pain, he sees not only agony to be relieved, but a scientific problem we are striving to solve. The young woman, together with our team, continues to explore the finer details of this pathway in the laboratory.
Behind this paper, there was no dramatic coincidence—only a clinician’s compassion for patients, a basic researcher’s curiosity about mechanisms, and shared perseverance from everyone involved. We believe that the finest medical research often begins with a question at the bedside and ends with an answer in the laboratory.
We are deeply grateful to our mentors, colleagues, and families for their support, and to the patients who participated in this study. They remind us that the ultimate purpose of scientific research is to return to the clinic and help more people.
Follow the Topic
-
Signal Transduction and Targeted Therapy

This is an international, peer-reviewed, open-access journal publishing articles related to signal transduction in physiological and pathological processes, alongside signal transduction-targeted therapeutics in the form of biological agents and small molecular drugs used to treat human diseases.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in