The Molecular Aegis: Defending the Gut Against IBD
Published in Cell & Molecular Biology and General & Internal Medicine
The intestinal epithelium is one of the most dynamic tissues in the human body, constantly regenerating to maintain a barrier against a world of microbes and toxins. At the heart of this renewal are the Lgr5⁺ intestinal stem cells (ISCs), tucked safely within the base of the crypts. But in Inflammatory Bowel Disease (IBD), this “home” is under siege. Our recent study in Nature Communications reveals a previously unknown molecular shield, a molecular aegis, that protects these vital cells during the storm of inflammation.
A Legacy of Two Decades: From Molecules to Mice and Men
This discovery culminates a 20-year journey rooted in a 2007 landmark study (Mol. Cell 2007; 26:75-87), which identified a biochemical "master switch": CBP phosphorylation at Ser1382/1386. This switch acts as a critical decision-maker for cell fate, shifting CBP’s binding preference from p53 (limiting growth) to NF-κB (promoting survival).
To explore this in a living organism, our laboratory generated the CBPAA mouse model using a high-precision conventional knock-in strategy shortly after. By replacing endogenous Serine residues with Alanines (S1383/1387A; corresponding to human S1382/1386), we created a systemic, “always-off” version of this switch. For years, several researchers sought to find the physiological significance of this mutation, but the clear phenotype remained elusive.
The Turning Point: A Shift from T Cells to the Colon
When I took over this project in 2014, we initially hypothesized that the CBP switch would be critical for T cell development. However, years of rigorous testing yielded no expected phenotype in the immune system.
Refusing to give up, I decided to step back and perform a systematic “whole-body” examination of the CBPAA mice. To our surprise, while most organs appeared normal despite the systemic mutation, we found a striking and specific defect: the colon was significantly shorter. This prompted us to look closer at human samples, where we observed a similar reduction in CBP phosphorylation in patients with Ulcerative Colitis (UC). Initially, this suggested a classic IBD scenario driven by immune dysregulation. However, our findings defied conventional wisdom. Through a series of bone marrow chimera experiments, we demonstrated that the colonic abnormalities remained even when the CBPAA mice were given a healthy immune system. This was the defining moment of the study: we realized the defect was independent of the immune system. The immune cells were like “caretakers” guarding our gut’s homeostasis; however, these suspected “caretakers” had not failed in their duty. Our attention then shifted from the guards to the “home” itself, the intestinal epithelium.
The Mystery of the Shrinking Organoids
Focusing on the intestinal epithelium, a BrdU pulse-chase assay provided our first breakthrough: CBPAA mice exhibited slower epithelial turnover compared to wild-type. This suggested that the regenerative engine of the gut, the intestinal stem cells, might be impaired.
To test this, we turned to ex vivo organoid cultures, the gold standard for assessing stem cell function. The results were striking: organoids derived from CBPAA mice were significantly smaller and struggled to grow. Based on the 2007 discovery that CBP phosphorylation directs binding preference, we initially hypothesized that in CBPAA cells, CBP would favor the p53 pathway, potentially driving the stem cells toward cell cycle arrest or apoptosis. However, when we used Lgr5-eGFP reporter mice to precisely visualize and count the cells, the data told a different story: the number of Lgr5⁺ stem cells had not decreased. The Lgr5⁺ stem cells, our research’s “infants”, were still there, and they weren't simply dying off. This led to a crucial realization: the defect wasn't an intrinsic failure of the cell themselves, but a failure of their “cradle”. We realized that these vital stem cells were like babies without a bed, exposed and unsupported because their microenvironment (the niche) was no longer providing the protective embrace they needed to thrive.
The Hunt: Finding the Unsung Hero
To find what was wrong with the stem cell “home”, we turned to RNA sequencing. However, whole-tissue data proved too noisy, and even our ex vivo organoids failed to yield clear candidates—likely because the potent growth factors in the culture media (Wnt, R-spondin, etc.) were masking the native phenotype by artificially rescuing the CBPAA defect. To capture the true state of the stem cell niche, we sharpened our focus and directly isolated only the crypts from the mouse colon. This was the turning point: after cross-referencing our datasets, the Venn diagram revealed a striking and undeniable result—only one single target remained. That target was Vcan (encoding the proteoglycan versican). We discovered that CBP phosphorylation acts as the master weaver, triggering the production of versican. This proteoglycan doesn't just sit there; it weaves itself into a soft, resilient molecular cradle (the Aegis) within the extracellular matrix. This cradle acts as a sanctuary, shielding the vulnerable stem cells from the harsh, inflammatory storms of the colon, allowing them to focus on the essential work of regeneration.
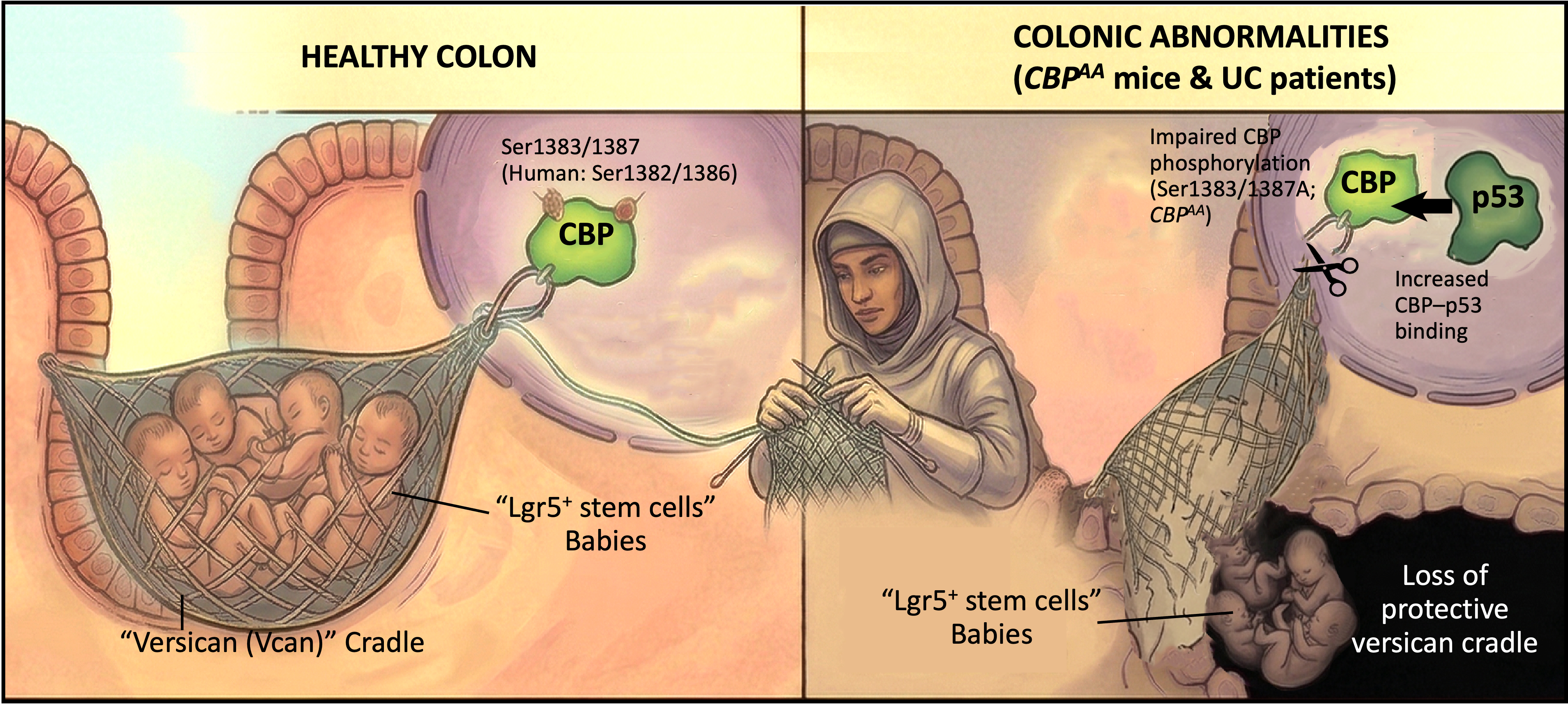
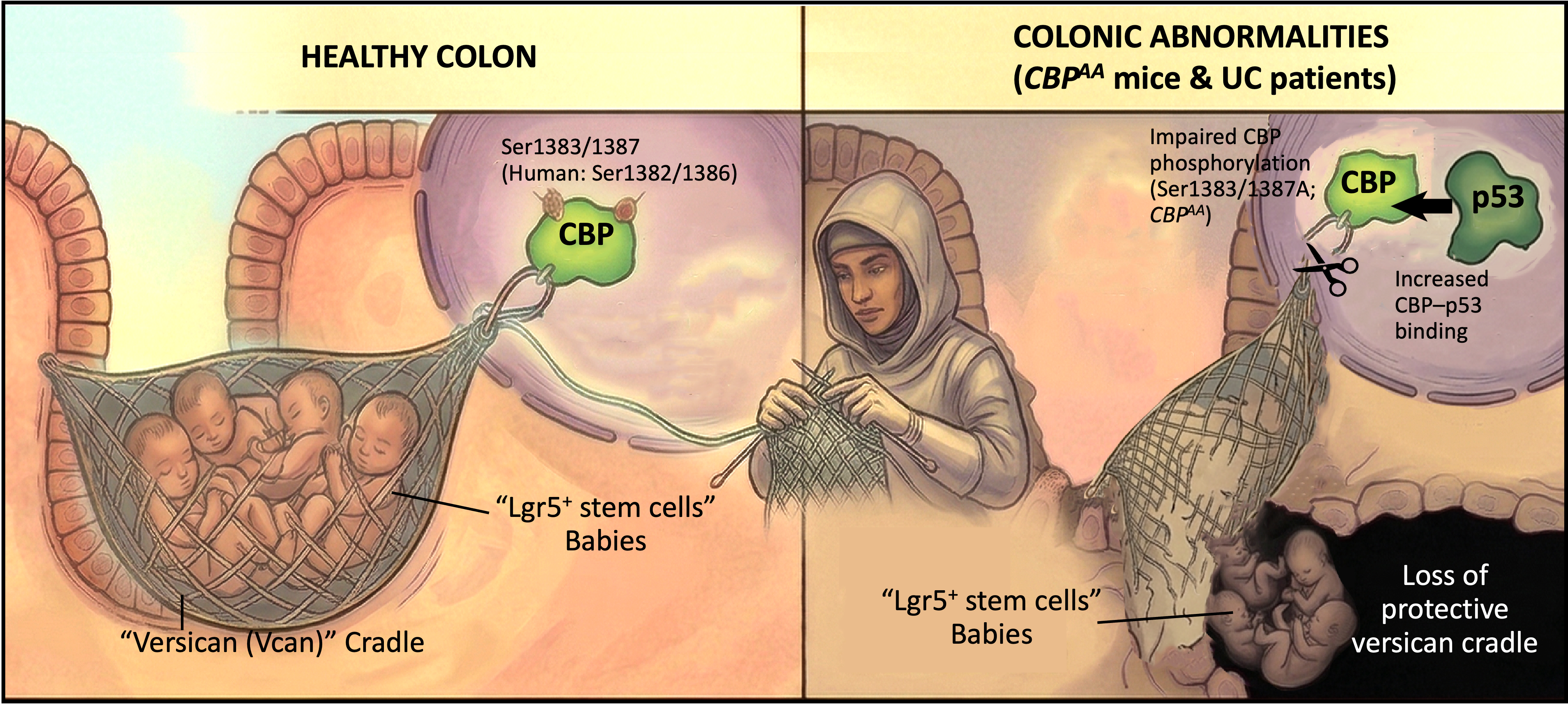
The Molecular Aegis: Safeguarding the Intestinal Stem Cell Nursery through CBP-Versican Axis. This conceptual illustration summarizes our decade-long journey to uncover the "Unsung Hero" of intestinal homeostasis. Left (Healthy Colon): CBP phosphorylation at Ser1382/1386 acts as a "Master Weaver," triggering the production of versican (Vcan). This proteoglycan forms a protective molecular cradle (aegis) that shields vulnerable Lgr5⁺ stem cells (babies) from environmental stress. Right (Colonic Abnormalities): In CBPAA mice and UC patients, impaired CBP phosphorylation leads to increased CBP–p53 binding, hijacking the switch. This causes the cradle to collapse (loss of versican), leaving the stem cells exposed and defenseless against inflammatory damage, despite their quantity remaining unchanged. (Image concept by Y.T. Lin; visualized with AI assistance)
Final Reflections: A Two-Decade Quest By connecting a 2007 landmark molecular discovery to this comprehensive study, we have completed a two-decade odyssey spanning from the molecular level to translational medicine, and from cells to mice and men. We’ve demonstrated that gut defense transcends immune cells; it relies on a "molecular aegis" where CBP phosphorylation safeguards intestinal homeostasis by maintaining the stem cell niche through versican. By identifying versican as a critical protective shield, our work offers new hope for IBD patients, shifting the therapeutic focus from merely suppressing inflammation to fortifying the niche and restoring the gut’s natural resilience. Our work reveals that sometimes, the best defense is a well-woven cradle.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in