Unmasking the Invisible Impact of Binge Drinking: A Journey Into the Brain's Immune Response
Published in Neuroscience

In the bustling world of neuroscience research, we are often driven by a compelling quest to unravel the mysteries of the human brain. Our recent journey, embarked upon by a team of dedicated scientists lead by Dr. Ansel Hillmer, was inspired by a notable gap in the literature: the acute effects of alcohol on the brain's neuroimmune system. While the long-term effects of alcohol use disorder are fairly well-documented at this point[1–3], the immediate aftermath of a binge drinking episode remained largely unexplored.
Before we delve deeper into our research, let's lay some groundwork and shine a light on the complex world of the neuroimmune system, focusing particularly on a star player: the microglial cell. The neuroimmune system serves as the brain's dedicated defense mechanism, a network of intricate processes that monitor and respond to potential threats. At the heart of this system are the microglia, unique immune cells that reside in the brain and spinal cord. These vigilant cells are the brain's first line of defense, springing into action at the first sign of trouble. They work tirelessly, patrolling the brain, looking out for signs of infection, injury, or other disruptions. When they detect such a threat, microglia transform from a surveillance state into an active state, releasing inflammatory substances to protect brain tissue.
Our research journey sought to illuminate this unexplored domain, employing positron emission tomography (PET) with a radiotracer, [11C]PBR28. This tracer is known to target the 18-kDa translocator protein, a marker of microglia number that is responsive to immune challenges[4-5]. While our endeavor was meticulously structured, it was not exempt from obstacles. Each participant embarked on a day-long data acquisition marathon in the research scanner. The day started with a baseline scan, followed by a lunch break. Then came the binge drinking session, featuring four to five cocktails not exactly in vogue in 2023: 80-proof vodka mixed with a decarbonated, non-caffeinated, and non-caloric drink. This was followed by another scan and a seemingly endless stream of questionnaires.
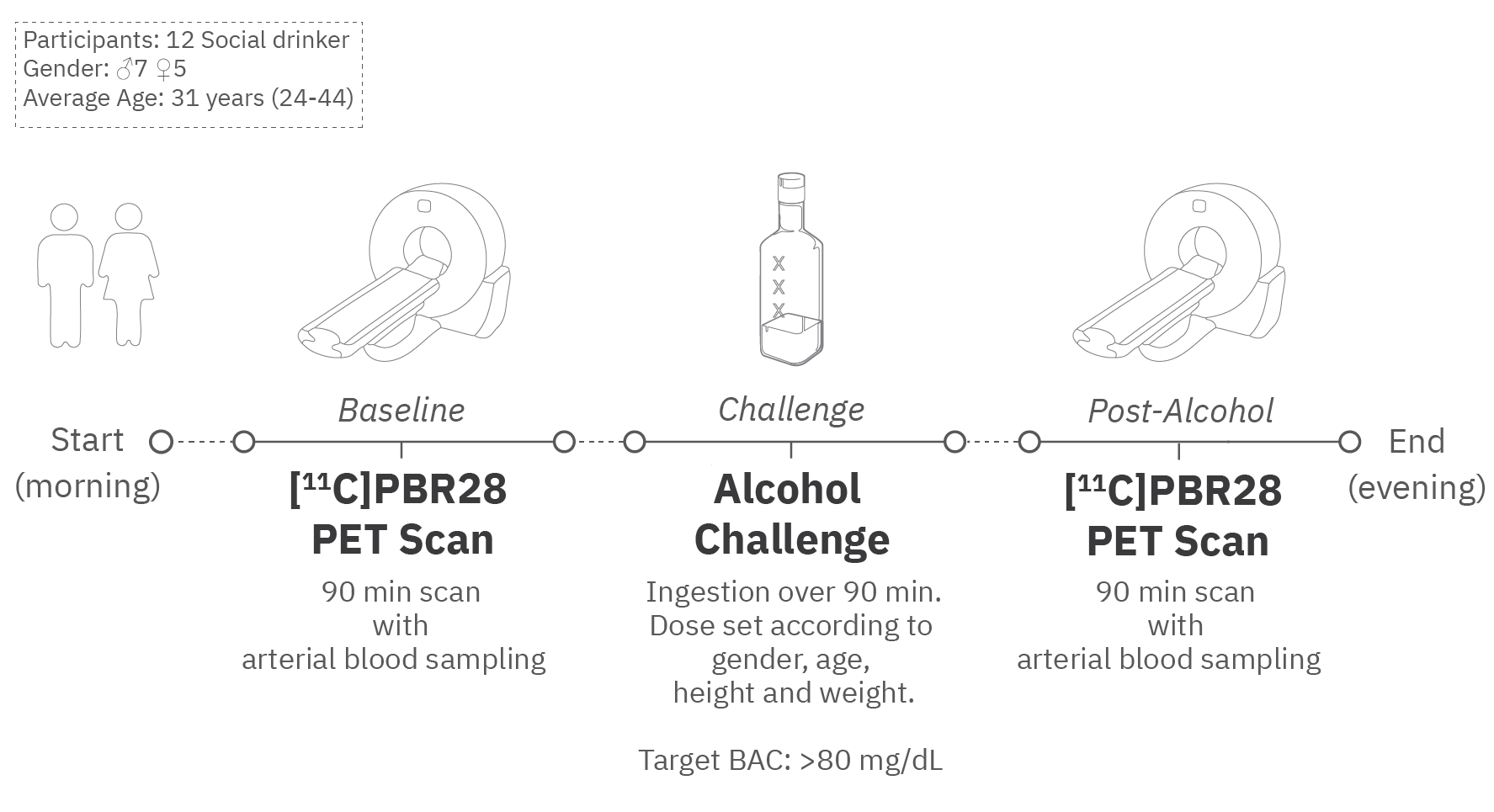
The melody of this intricate ballet was further complicated by the ongoing COVID-19 pandemic, which introduced additional logistical challenges and made participant recruitment more difficult. At times, technical glitches forced us to discard some scans, adding yet another layer of complexity to our already intricate symphony. Despite these obstacles, our team's perseverance remained unwavering. We are deeply grateful to our participants, whose contributions have created an invaluable data set. This data will not only advance our understanding of alcoholism but also shed new light on the neuroimmune system, paving the way for future breakthroughs.
As we navigated these difficulties, we were rewarded with moments of revelation that sparked a sense of wonder. Our data unveiled a profound and global response in the brain's neuroimmune system across all participants following a binge drinking episode. This was a surprising outcome. While we anticipated a response based on rodent[6] non-human primate data[7], the extent of the response was more dramatic than we had expected. These novel findings present an exciting leap forward in the field of neuroscience. The neuroimmune system, a somewhat nebulous entity, came into sharper focus with our discovery. We found that a significant increase in neuroimmune activity could occur after just one session of binge drinking - a fact that many people had not considered. This revelation could have profound implications for future research, suggesting that blood alcohol levels might be an important factor to consider when scanning individuals for neuroimmune markers.
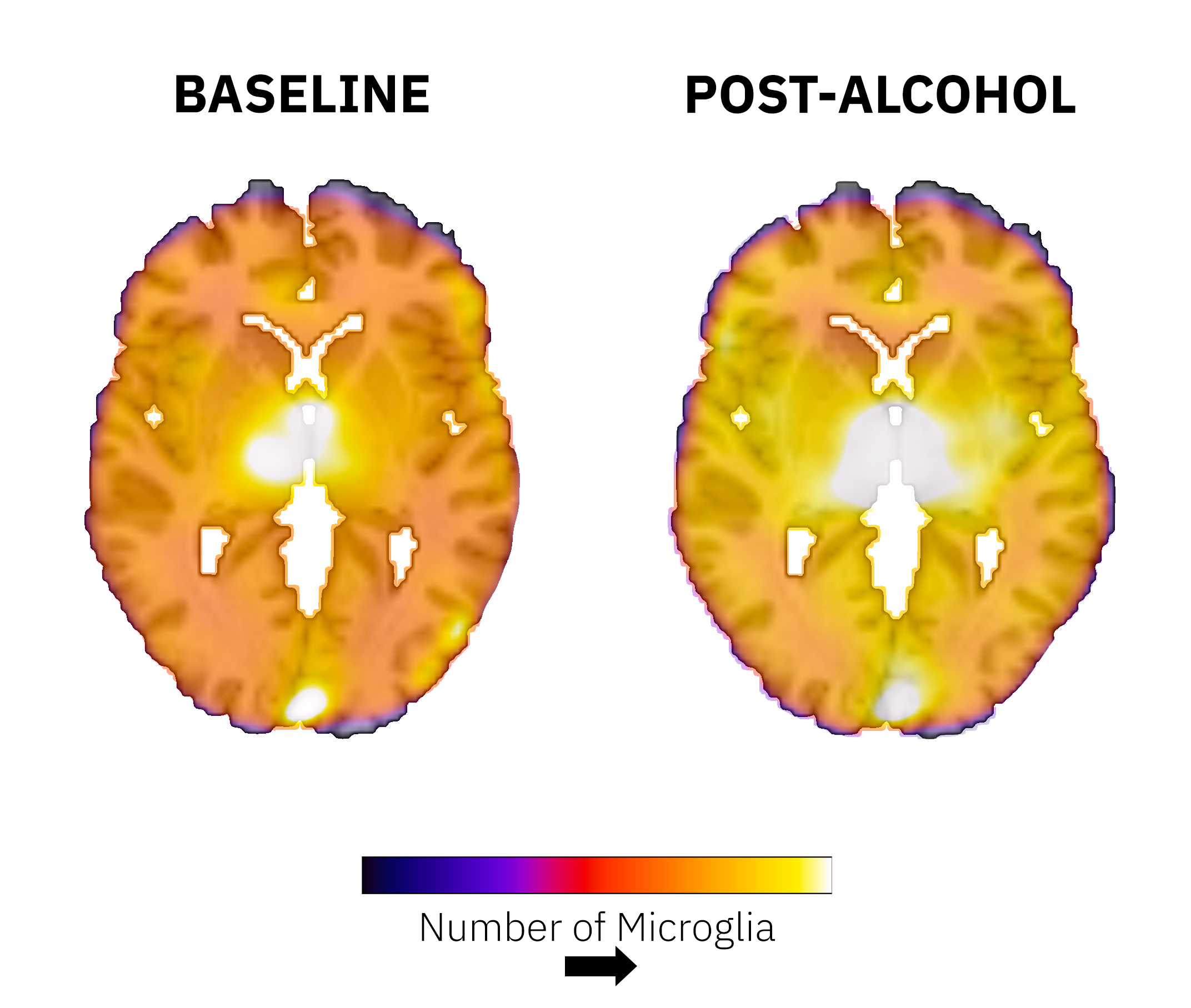
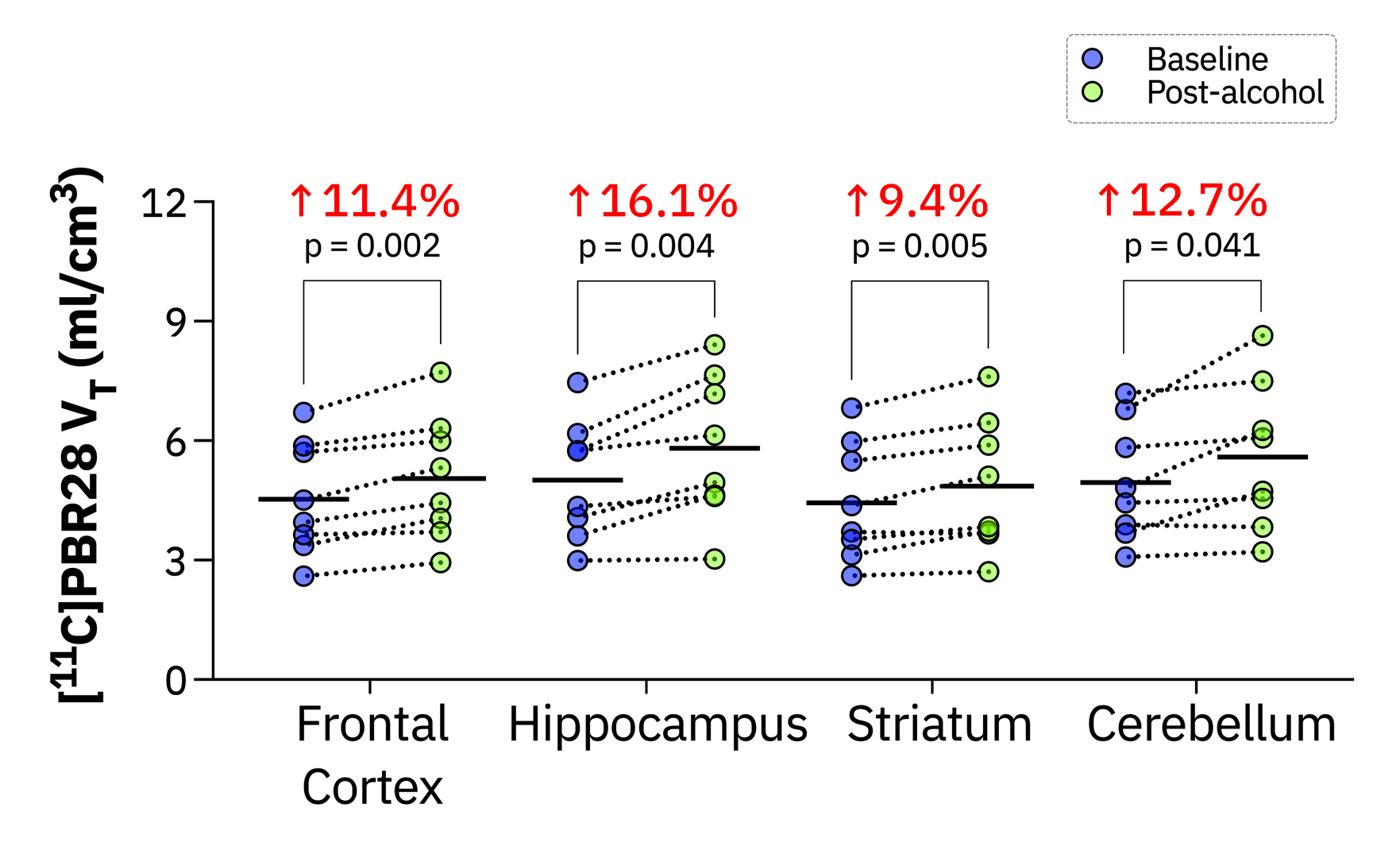
The societal implications of our research are equally compelling. Binge drinking, a pervasive practice in many cultures, has now been linked to immediate impacts on the brain's immune system. This finding dispels common misconceptions and brings to light an unseen consequence of alcohol consumption. It forms part of a broader narrative that promotes responsible drinking habits and underscores the intricate ways in which alcohol affects the brain.
What does it mean to have a neuroimmune response in the brain, especially in response to alcohol? At the most basic level, the neuroimmune response involves an increase in microglia activity in the brain. When alcohol enters the brain, it triggers an immune response, causing potential microglia proliferation and causing them to release inflammatory substances- cytokines. This increased cytokine and neuroimmune activity, as we've found in our study, can happen even after a single binge drinking episode. But is this good or bad for your brain?
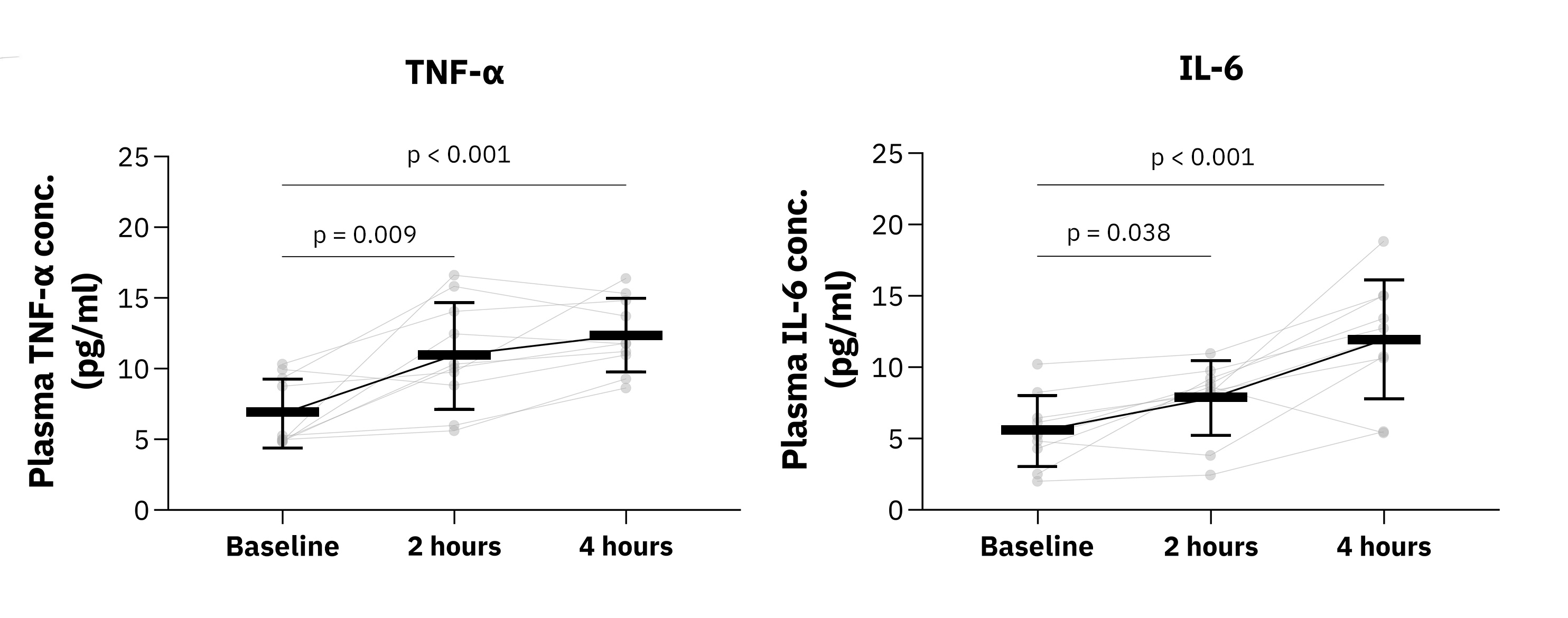
The answer isn't straightforward. On one hand, an active immune response can help protect the brain from potential damage. On the other, if this response is too robust or prolonged, it can cause inflammation and damage to brain cells. With alcohol use disorder (AUD), studies have shown a reduction in the number of microglia[1], potentially due to a kind of 'burnout' from the repeated immune activation caused by regular heavy drinking.
So, could this spike in neuroimmune activity during an acute exposure to alcohol be a precursor to the eventual decrease in microglia seen in chronic AUD? It's a compelling question, and one that warrants further research. If this is the case, it could mean that each heavy drinking episode contributes to a long-term reduction in the brain's ability to protect itself against damage, potentially leading to the cognitive and behavioral impairments often seen in chronic AUD.
As researchers, we found fulfillment in our shared pursuit of discovery and in the profound insights we gained into the complexities of the human brain. This exploration, while marked by challenges, was ultimately a testament to the power of scientific curiosity and resilience. Our findings serve as a beacon of knowledge, illuminating a path for future research and offering a deeper understanding of the unseen effects of alcohol on the brain's immune system.
As we unravel the complexities of the neuroimmune response to alcohol, we are gaining critical insights into the inner workings of the brain. These discoveries could have significant implications for understanding, preventing, and treating alcohol-related disorders. As we venture further into this unexplored territory, we remain committed to our quest to unravel the mysteries of the neuroimmune system. We hope that our findings will serve as a foundation for future studies, fostering a deeper understanding of the brain's response to alcohol, and aiding in the development of interventions to mitigate its effects. Through our research, we hope to contribute to a broader narrative that encourages responsible drinking habits, promoting the health and wellbeing of individuals and communities alike.
References:
- Hillmer AT, Sandiego CM, Hannestad J, Angarita GA, Kumar A, McGovern EM, et al. In vivo imaging of translocator protein, a marker of activated microglia, in alcohol dependence. Mol Psychiatry. 2017;22:1759–1766.
- Kalk NJ, Guo Q, Owen D, Cherian R, Erritzoe D, Gilmour A, et al. Decreased hippocampal translocator protein (18 kDa) expression in alcohol dependence: a [11C]PBR28 PET study. Transl Psychiatry. 2017;7:e996.
- Kim SW, Wiers CE, Tyler R, Shokri-Kojori E, Jang YJ, Zehra A, et al. Influence of alcoholism and cholesterol on TSPO binding in brain: PET [11C]PBR28 studies in humans and rodents. Neuropsychopharmacology. 2018;43:1832–1839.
- Sandiego CM, Gallezot J-D, Pittman B, Nabulsi N, Lim K, Lin S-F, et al. Imaging robust microglial activation after lipopolysaccharide administration in humans with PET. Proc Natl Acad Sci U S A. 2015;112:12468–12473.
- Tournier BB, Tsartsalis S, Ceyzériat K, Medina Z, Fraser BH, Grégoire M-C, et al. Fluorescence-activated cell sorting to reveal the cell origin of radioligand binding. J Cereb Blood Flow Metab. 2020;40:1242–1255.
- Marshall SA, McClain JA, Kelso ML, Hopkins DM, Pauly JR, Nixon K. Microglial activation is not equivalent to neuroinflammation in alcohol-induced neurodegeneration: The importance of microglia phenotype. Neurobiol Dis. 2013;54:239–251.
- Saba W, Goutal S, Auvity S, Kuhnast B, Coulon C, Kouyoumdjian V, et al. Imaging the neuroimmune response to alcohol exposure in adolescent baboons: a TSPO PET study using 18 F-DPA-714. Addict Biol. 2018;23:1000–1009.
*Note: Cover image was created using Adobe Firefly.
Follow the Topic
-
Molecular Psychiatry
This journal publishes work aimed at elucidating biological mechanisms underlying psychiatric disorders and their treatment, with emphasis on studies at the interface of pre-clinical and clinical research.
Your space to connect: The Psychedelics Hub
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcement



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in