Vessel-Controlled Chemodivergence Meets Vinylcyclopropane–Cyclopentene Rearrangement
Published in Chemistry
Gem-difluorocyclopropanes have attracted sustained interest from our research group. Through long-term and focused investigations, we have uncovered reactivity patterns that distinguish these fluorinated cyclopropanes from their non-fluorinated counterparts. We attribute this distinctive behavior to the presence of the gem-difluoromethylene group, which exerts profound electronic and steric effects that modulate reaction pathways.
Given the unique properties of gem-difluorocyclopropanes, we became intrigued by the reactivity of gem-difluorinated vinylcyclopropanes. Vinylcyclopropanes (VCPs) represent a class of small-ring molecules characterized by high ring strain and versatile reactivity, making them valuable building blocks in organic synthesis. Their ability to undergo various ring-opening and rearrangement processes has been extensively exploited for the construction of complex molecular architectures.
We hypothesized that, under transition-metal catalysis and in the presence of the gem-difluoro effect, these substrates might generate unusual alkenyl allylic intermediates with enhanced electrophilicity. Such intermediates could open new avenues for reaction development by engaging in diverse transformations, including rearrangement, functionalization, and cycloaddition. The enantioconvergent VCP–cyclopentene rearrangement we ultimately discovered was both anticipated—given our understanding of the underlying reactivity—and unexpectedly surprising in its mechanistic details and outcome.
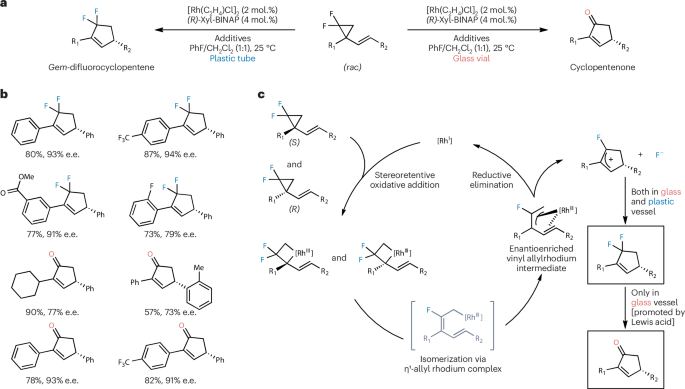
To initiate our investigation, we prepared a model substrate from commercially available chalcones. This type of substrates, bearing two phenyl substituents, was designed to generate a cationic alkenyl allylic intermediate that is both relatively stable and highly reactive. We sought to explore whether this intermediate would undergo rearrangement in the absence of an external coupling partner, or participate in functionalization or cycloaddition when such a partner was present.
In our initial experiments, we did not observe the expected VCP–cyclopentene rearrangement product. Instead, cyclopentenone derivatives were formed as the major products. Remarkably, the reaction proceeded in an enantioconvergent manner, converting racemic starting materials into enantioenriched products—a transformation that is notoriously challenging to achieve with strained cyclopropanes.
We hypothesized that the gem-difluoromethylene group plays a pivotal role in this process. Notably, both fluorine atoms are transformed in the presence of oxygen donors. Elucidating the source and mode of action of these oxygen donors became essential for a deeper mechanistic understanding. Through systematic mechanistic investigations, we discovered that oxides present in common glass materials serve as highly effective oxygen donors and are well tolerated under the reaction conditions. Water, while also capable of acting as an oxygen donor, exhibited significantly lower reaction efficiency. This observation echoes the “vessel effect” previously reported by Nielsen and others, who demonstrated that the material of the reaction vessel can influence reactivity by sequestering fluoride to generate Lewis acids that participate in C–F bond activation.
In our system, the vessel material dictates the reaction outcome in a striking manner, and we called it as an unusual vessel-controlled chemodivergence. Glass vessels, which contain abundant oxygen donors, favor the formation of cyclopentenones; in contrast, plastic vessels, which lack such donors, lead to the selective formation of gem-difluorocyclopentenes. With encouragement from the editor and valuable input from several reviewers, we have developed a deeper mechanistic understanding. The gem-difluoro effect not only promotes the enantioconvergent rearrangement but also enhances the electrophilicity of the key intermediates. Furthermore, the fluorine atoms participate in a fluorine recycling process or interact with oxygen donors to release nucleophilic oxygen species, thereby governing the chemoselectivity of the transformation.
These intriguing findings motivated us to further explore the synthetic utility and potential applications of this strategy. We first evaluated the biological activities of readily accessible cyclopentenones and gem-difluorocyclopentenes. Given that cyclopentenones constitute the core structure of prostaglandins—a class of bioactive lipids with diverse physiological functions—we initiated a collaboration with the group of Prof. Xin Wei in the Department of Urology at West China Hospital, Sichuan University, to investigate the anti-bladder cancer activity of these compounds.
Interestingly, several enantioenriched products exhibited promising anticancer activity, while racemic products showed even greater potency. This counterintuitive observation highlights the potential for further exploration of these molecules. The fact that racemic mixtures can outperform their enantioenriched counterparts in biological assays suggests that the stereochemical complexity of these compounds may engage multiple biological targets in a synergistic manner.
Beyond biological evaluation, we demonstrated that both cyclopentenones and gem-difluorocyclopentenes can be efficiently converted into complex five-membered ring compounds bearing multiple contiguous stereocenters. This transformation provides a powerful platform for the synthesis of diverse prostaglandin analogues and derivatives, which are of significant interest in medicinal chemistry. The ability to rapidly access such structurally complex scaffolds from simple, readily available starting materials represents a notable advance in synthetic methodology.
Currently, additional transformations based on this enantioconvergent rearrangement strategy are under active investigation in our laboratory. These include explorations of new reaction partners, catalyst modifications, and extensions to other classes of strained-ring systems. The resulting compound libraries will be systematically evaluated for anti-bladder cancer activity in collaboration with Prof. Wei’s group. We anticipate that this integrated approach—combining mechanistic insight, synthetic innovation, and biological evaluation—will not only deepen our exploration on gem-difluorocyclopropane chemistry but also pave the way for the discovery of new therapeutic leads.
Follow the Topic
-
Nature Chemistry
A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in