W/V Dual-Atom Doping MoS2-Mediated Phase Transition for Efficient Polysulfide Adsorption/Conversion Kinetics in Lithium–Sulfur Battery
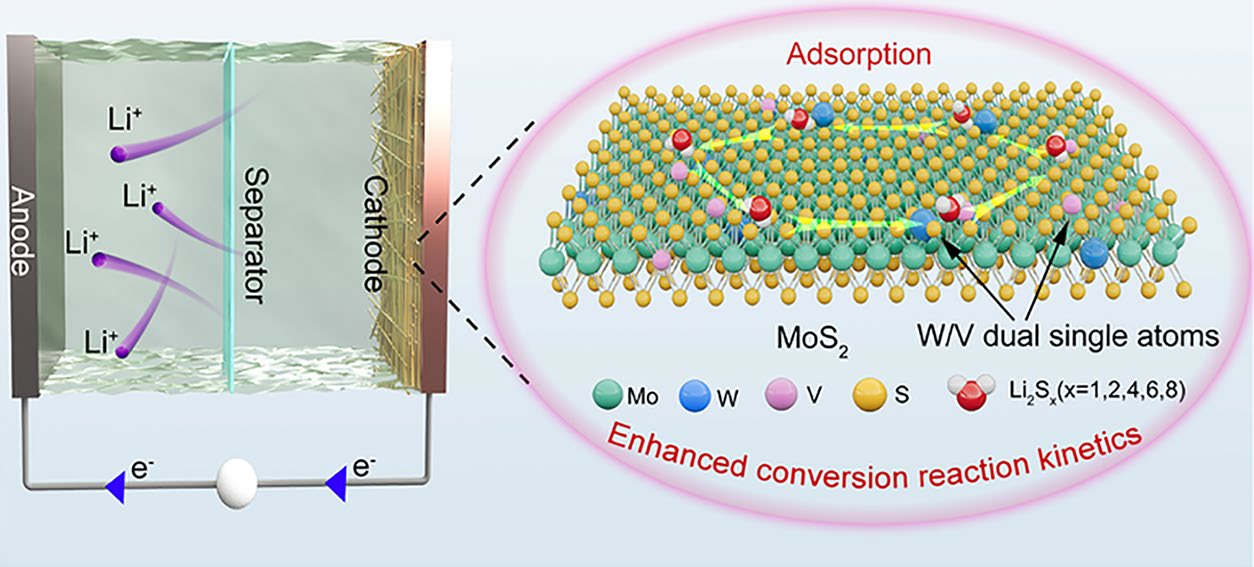
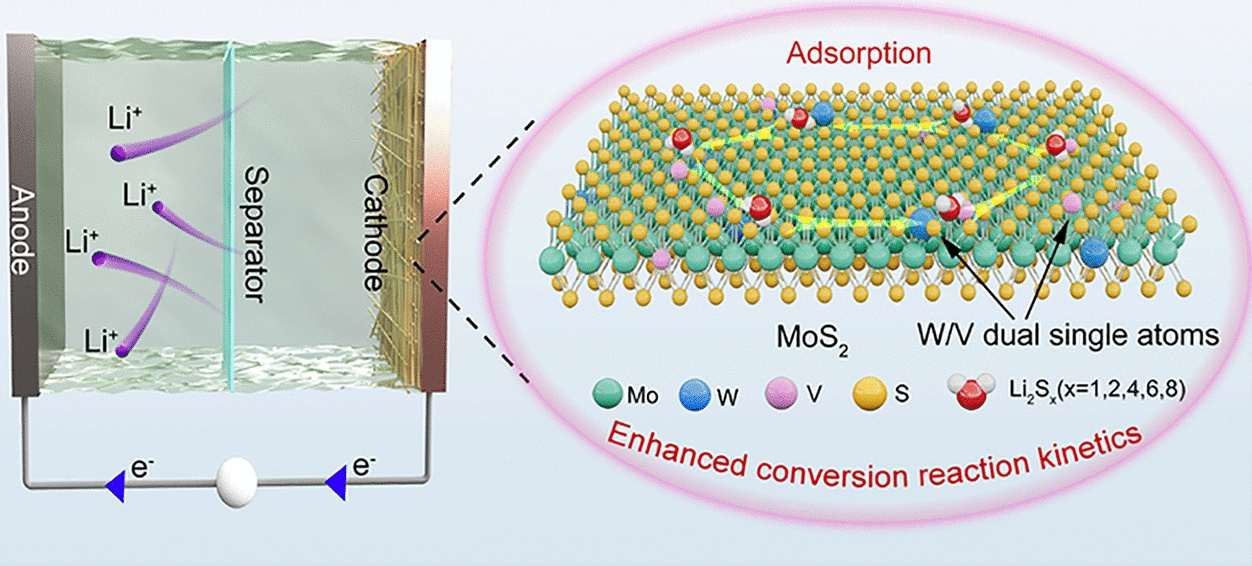
The dissolvable polysulfides and sluggish Li2S conversion kinetics are acknowledged as two significant challenges in the application lithium–sulfur (Li–S) batteries. Herein, we introduce a dual-doping strategy to modulate the electronic structure of MoS2, thereby obtaining a multifunctional catalyst that serves as an efficient sulfur host. The W/V dual single-atom-doped MoS2 grown on carbon nanofibers (CMWVS) demonstrates a strong adsorption ability for lithium polysulfides, suppressing the shuttle effects. Additionally, the doping process also results in the phase transition from 2H-MoS2 to 1T-MoS2 and generates sufficient edge sulfur atoms, promoting the charge/electron transfer and enriching the reaction sites. All these merits contribute to the superior conversion reaction kinetics, leading to the outstanding Li–S battery performance. When fabricated as cathodes by compositing with sulfur, the CMWVS/S cathode delivers a high capacity of 1481.7 mAh g−1 at 0.1 C (1 C = 1672 mAh g−1) and maintains 816.3 mAh g−1 after 1000 cycles at 1.0 C, indicating outstanding cycling stability. Even under a high sulfur loading of 7.9 mg cm−2 and lean electrolyte conditions (E/S ratio of 9.0 μL mg−1), the cathode achieves a high areal capacity of 8.2 mAh cm−2, showing great promise for practical Li–S battery applications. This work broadens the scope of doping strategies in transition-metal dichalcogenides by tailoring their electronic structures, providing insightful direction for the rational development of high-efficiency electrocatalysts for advanced Li–S battery applications.
Follow the Topic
-
Nano-Micro Letters
Nano-Micro Letters is a peer-reviewed, international, interdisciplinary and open-access journal that focus on science, experiments, engineering, technologies and applications of nano- or microscale structure and system in physics, chemistry, biology, material science, and pharmacy.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in