A de novo gene originating from plant mitochondria
Published in Ecology & Evolution

De novo genes are new genes derived from non-coding sequences following the accumulation of mutations during evolution. Random mutations are likely to generate a small ORF, but not a long ORF. Consequently, de novo genes tend to be smaller than other ancient genes. Although such de novo genes may play a lineage-specific function, these small genes have generally not been annotated even for model species. Interestingly, mitochondrial genome in plants possesses 10- to 50- times larger size than those in animals. Due to a high rearrangement rate of plant mitochondrial genomes, plants tend to create de novo genes with 2- to 10- times higher proportion in mitochondrial genomes than that of nucleus genomes (Fig. 1). Furthermore, in multiple plant lineages, the mitochondrial genome has frequently been inserted into the nuclear genome via intergenomic gene transfer (IGT) events. That is, many de novo genes that emerged in plant mitochondrial genomes have been transferred into nuclear genomes. However, the functions of de novo genes derived from the mitochondrial genome are unclear.
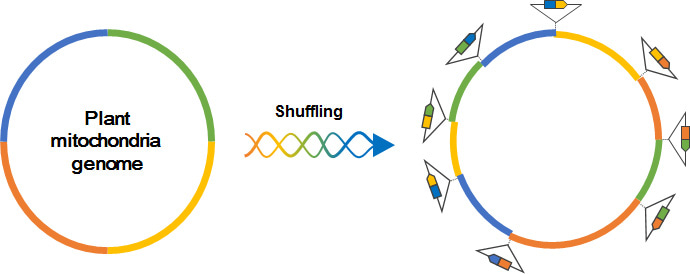
In this study, we focused on a small gene sequence (sORF2146) encoding 66 amino acids, which emerged in the mitochondrial genome of the Brassica lineage. In A. thaliana, sORF2146 was transferred into the nuclear genome during a large-scale IGT event. Consequently, there are identical sORF2146 sequences in the nuclear and mitochondrial genomes (Fig. 2).
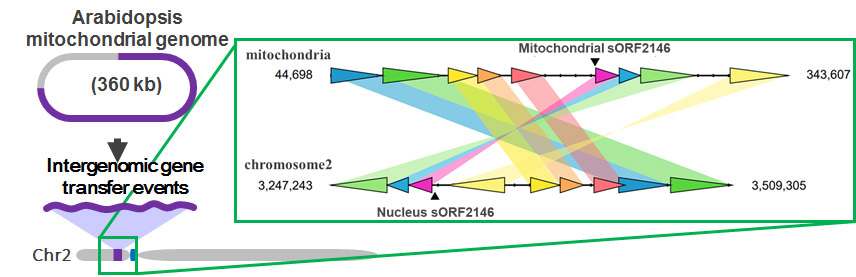
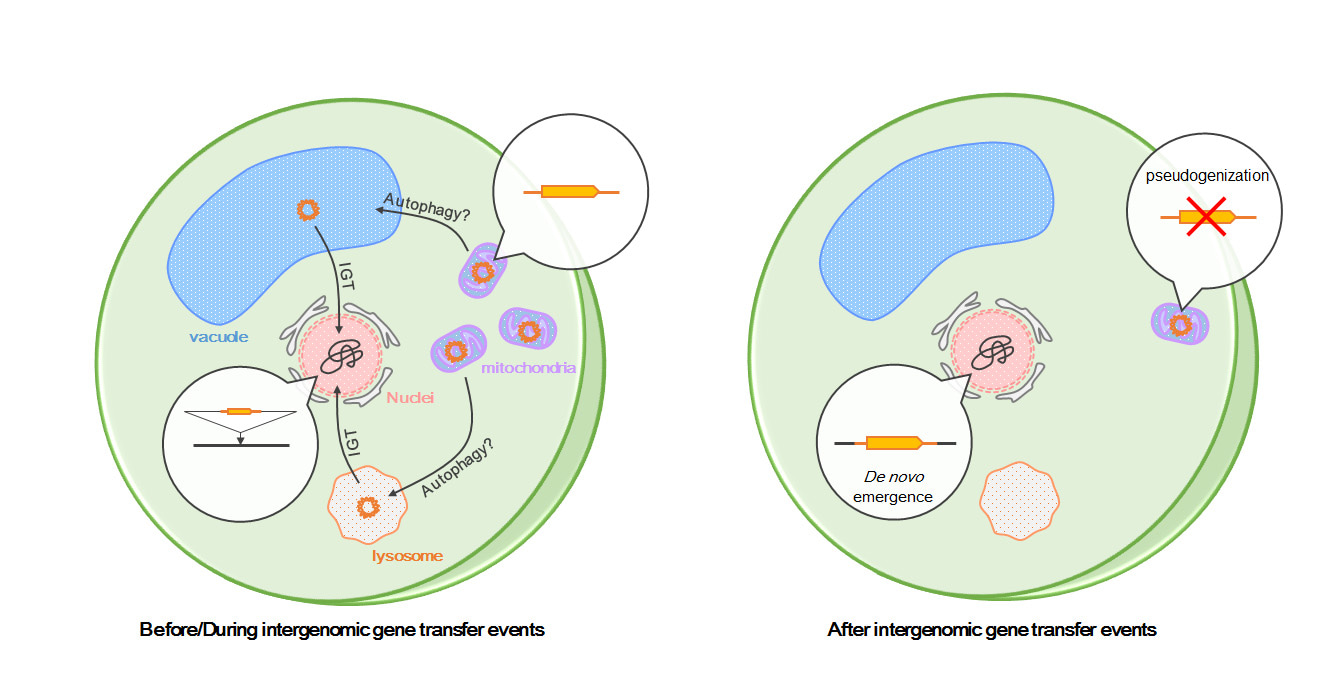
We identified the full-length sORF2146 transcript and mapped it to a region on chromosome 2, but it was not mapped to a sequence in the mitochondrial genome because of a large deletion. This implies that the nuclear sORF2146 is transcribed into mRNA. Furthermore, we revealed that the amplified sORF2146 transcript was derived exclusively from the nuclear genome. We also found that the subcellular localization of nuclear sORF2146 was mitochondria by sORF2146 products fused wit green fluorescent protein. Thus, we can speculate that sORF2146 is functional in mitochondria. Originally, there was functional redundancy between the mitochondrial sORF2146 and the nuclear sORF2146 immediately after IGT event (Fig. 3). Currently, the mitochondrial sORF2146 may have been modulated by a pseudogenization because of redundant functions (Fig. 3). In future, we speculate that the mitochondrial sORF2146 may disappear by accumulated mutations.
We performed a transcriptomic analysis of transgenic plants overexpressing the nuclear sORF2146. The transcriptome analysis showed that sORF2146 suppresses floral transition by up-regulating the expression of several genes encoding flowering regulators. Furthermore, overexpression and suppression of sORF2146 in transgenic plants represented delayed and early flowering, respectively (Fig. 4). On the basis of these findings, we conclude that a mitochondrion-originating de novo gene (sORF2146) influences A. thaliana floral transition.

It is well known that the regulation of floral transition is important for biotic/abiotic stress tolerance. Indeed, in our previous study examining 33 developmental stages and environmental conditions, abiotic stresses were observed to substantially up-regulate sORF2146 expression. Additionally, plant mitochondria regulate several developmental processes, including the floral transition under abiotic stress conditions. Thus, sORF2146 derived from mitochondria might contribute to abiotic stress tolerance of plants.
To our knowledge, this is the first report to reveal the functional role of mitochondria-originating de novo gene. We previously identified more than 100 candidates of de novo genes derived from mitochondrial genome, which may have resulted in an incomplete functional characterization of the de novo genes. Considered together, our results indicate that many de novo genes in the nuclear genome were derived from the mitochondrial genome.
Follow the Topic
-
Plant Molecular Biology
An international journal dedicated to rapid publication of original research articles in all areas of plant biology.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in