A New Strategy to Alleviate Oxidative Stress
Published in Chemistry
Note: This blog entry was co-authored by Ivana Ivanović-Burmazović
The overproduction of reactive oxygen species has been linked to a huge array of terrifying health conditions, including cardiovascular, inflammatory, and neurological disorders. The body employs numerous antioxidants to control the concentrations of reactive oxygen species and prevent oxidative stress from occurring. Among these antioxidants are superoxide dismutases (SODs), which are enzymes that use redox-active metal ions (manganese, iron, nickel, copper) to catalyze the degradation of superoxide into dioxygen and hydrogen peroxide.
Given the possible benefits to human health, there’s been great interest in developing small molecule functional mimics of SODs. The vast majority of these mimics contain manganese. Manganese has the advantage of being less toxic than other redox-active metal ions that have the required catalytic activity, but the use of this metal has several disadvantages as well. Manganese is kinetically labile, and its ability to rapidly exchange even polydentate ligands destabilizes the mimic and limits how long catalysis can persist. Free manganese can lead to an adverse neurological condition called, logically enough, manganism. Last, cellularly abundant phosphate anions have been found to inhibit most of the manganese-containing catalysts.
In our Nature Chemistry article, we use a substantially different approach to achieve SOD mimicry. Instead of a redox-active transition metal ion, we use a zinc complex with a redox-active ligand. The redox-active portion of the ligand is a quinol, which can react with superoxide to attain one of three oxidation states: quinol, quinoxyl radical, and para-quinone. The zinc, conversely, remains in the +2 oxidation state throughout catalysis. The zinc is still essential to the catalysis in that it draws superoxide anions towards the complex, brings reactive moieties into close proximity to each other, and modulates the acidity of the ligand and consequently its redox potential. The zinc binds more tightly to the ligand than manganese, and even if it were to be released, the zinc dications are much less toxic. The few known instances of zinc poisoning have resulted from people eating metallic objects.
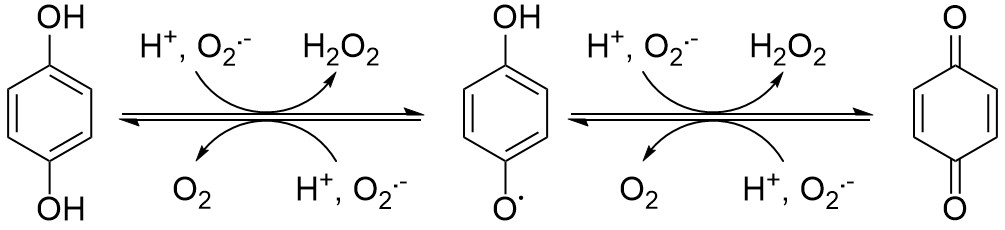
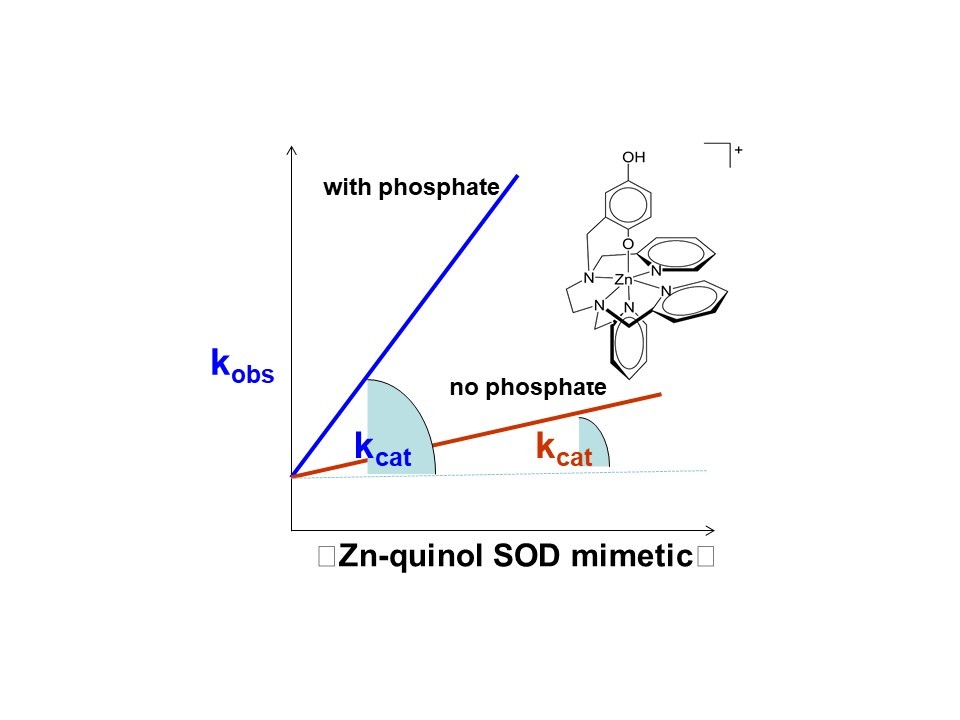
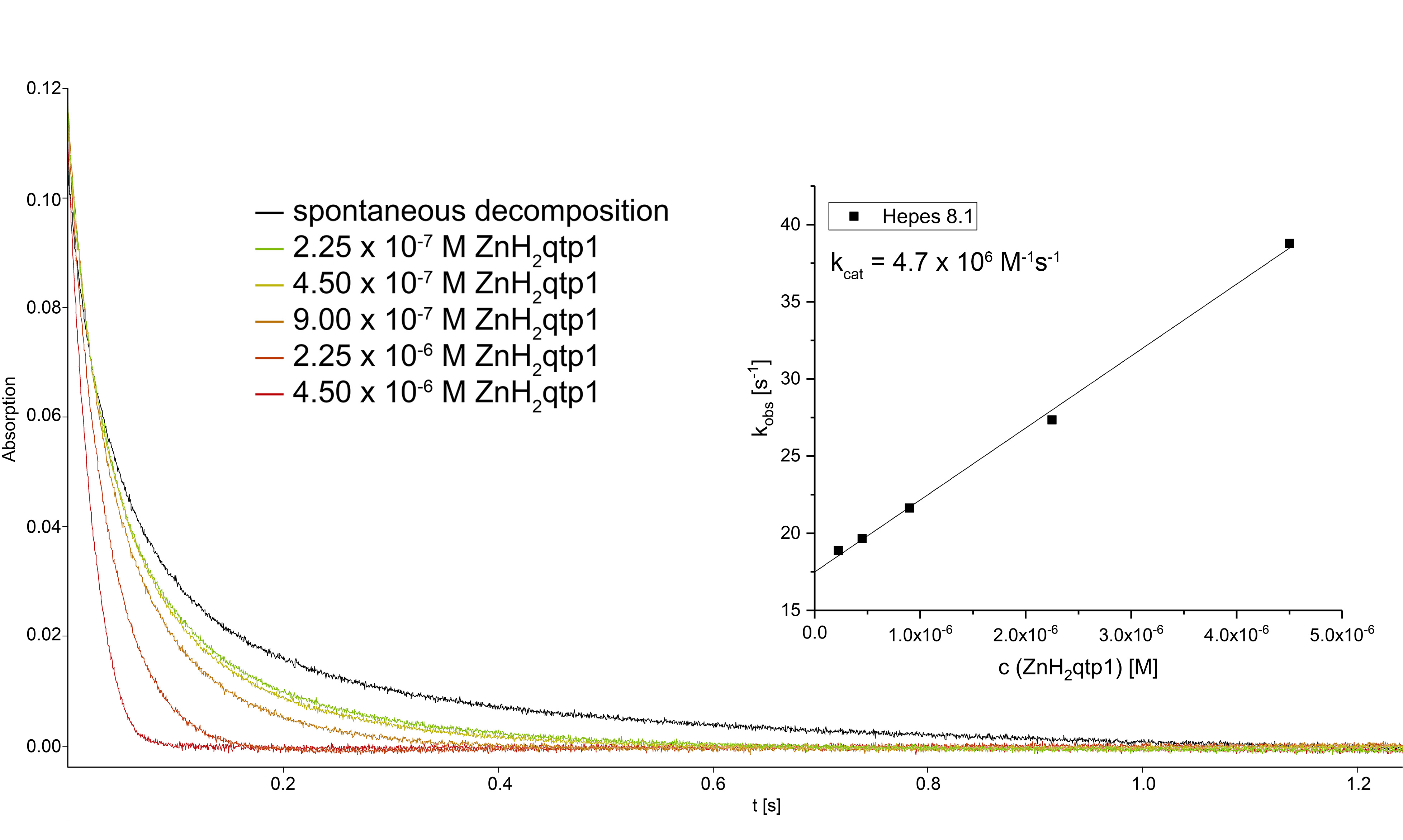
We found that without zinc, the organic ligand is not functional, whereas its zinc complex behaves like a SOD enzyme despite the lack of a redox-active transition metal. Contrary to what has been found for manganese-containing SOD mimics, the activity of the catalyst is actually enhanced by phosphate – a phenomenon that we hypothesize results from more efficient proton transfer between the two negatively charged buffer components and the positively charged zinc species in the catalytic cycles. Efficient proton transfer facilitates the electron transfer required for catalytic superoxide degradation. We were able to generate a transient paramagnetic zinc complex by reacting the starting material with a one-electron oxidant and a base. We believe that this corresponds to the zinc-quinoxyl radical complex that would result from the initial one-electron oxidation by superoxide.
This new strategy could lead to the development of new inorganic pharmaceuticals. Biochemists seeking SODs have traditionally focused on enzymes that contain redox-active transition metals; our work demonstrates that having such a metal is not a strict requirement for SOD activity.
Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in