Acidic CO2RR Employing Bimetallic Catalysts Increases the Carbon Utilization Efficiency in CO2 Electrolysis
Published in Chemistry
The electrochemical CO2 reduction reaction (CO2RR) to multi-carbon products, such as ethanol and ethylene, offers a means to produce fuels and chemicals, all powered using renewable (a.k.a. green) electricity. The most selective and active CO2RR electrolyzers to date have relied on locally alkaline conditions such as in alkaline flow cells and anion exchange membrane (AEM) electrolyzers. Even the most carbon-efficient of these, however, suffer low carbon utilization, as well as severe crossover of liquid products. Both CO2 crossover and product crossover lead to the need for more costly product separation.
CO2RR in acidic media is known to eliminate carbonate formation; relying on acidic electrolytes enables the use of proton exchange membranes that block liquid crossover. However, the competing hydrogen evolution reaction in acidic solution militates against both selectivity and activity of CO2RR, especially for CO2-to-multi-carbon products.
We therefore sought routes to suppress the competing hydrogen evolution reaction (HER). Metals with a strong binding affinity toward CO* weaken H* binding affinity due to adsorbate-adsorbate interactions. Using DFT calculations, we screened different bimetallic X-Cu (X = Cr, Mo, W, Mn, Re, Fe, Ru, Co, Rh, Ir, Ni, Pt, and Pd) catalysts and found that Pd-Cu offers the lowest ∆GOCCOH* and ∆GOCCOH* - ∆GCHO*. The adsorption energy of H* is 0.2 eV weaker on Pd–Cu compared to on Cu, pointing toward the suppression of HER.
Experimentally, we synthesized Pd–Cu bimetallic catalysts for implementation in an acidic CO2RR electrolyzer employing an acidic bulk environment and operating under conditions that produce a mildly alkaline local environment at the catalyst surface. We found that electrolytes with pH 2 led to a locally mildly alkaline environment under practical operating current densities: this enabled both high CO2RR activity and yet sufficient acidity in the bulk liquid in order to convert carbonate back into CO2.
We report as a result liquid product crossover < 0.05% with a CO2-to-C2+ Faradaic efficiency (FE) of (89 ± 4)% and single-pass carbon efficiency of (60 ± 2)% to C2+ (total CO2 utilization of (68 ± 4)% when considering the sum of C1 and C2+ products) at 500 mA cm-2 in acidic media.
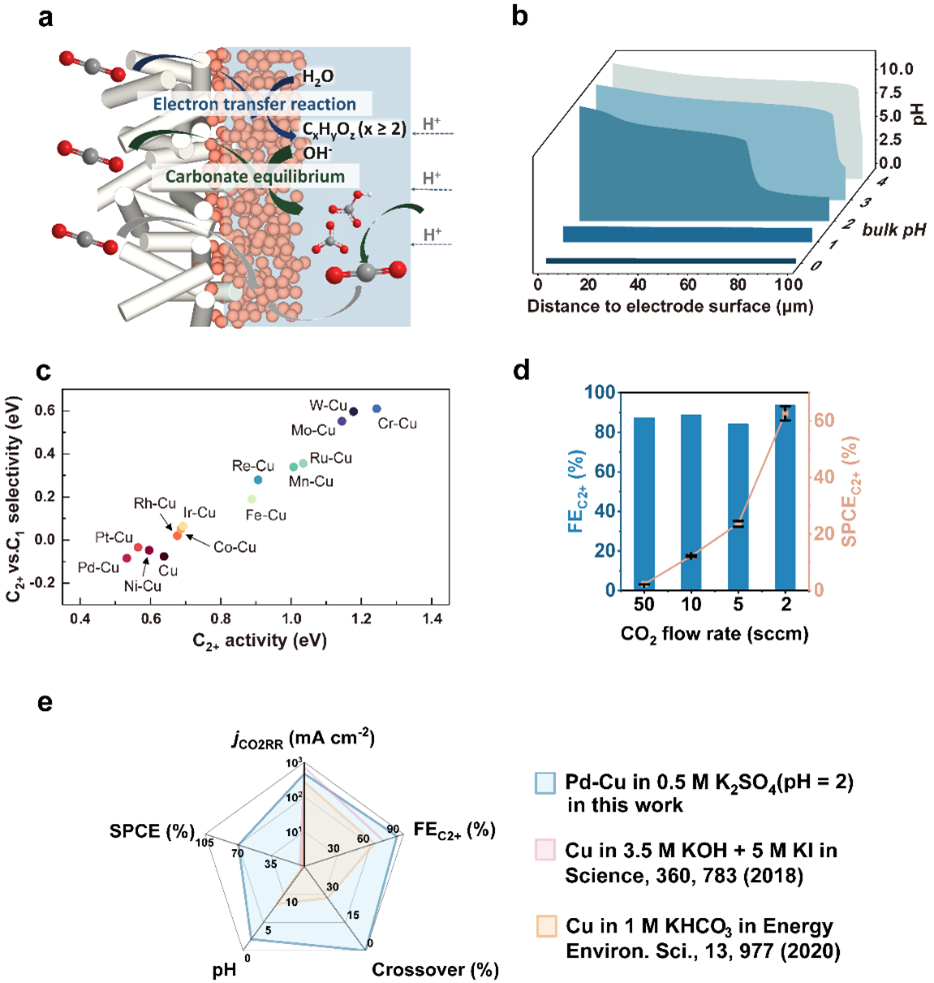
Fig. 1 | (a) Schematic of acidic CO2RR electrolyser. (b) Modeled pH changes along the catalyst’s surface at solution with various pH under applied current density of 500 mA cm-2 . (c) Two-dimensional C2+ activity (∆GOCCOH*) and C2+ vs. C1 selectivity (∆GOCCOH* − ∆GCHO*) plot of CO2RR to C1 and C2+ products. (d) FEs and single-pass carbon efficiency (SPCEC2+) of CO2-to-C2+ on 6.2% Pd–Cu with different CO2 flow rates (applied current density = 500 mA cm-2). (e) Comparison of CO2RR partial current density, C2+ product FE, liquid product crossover fraction, pH of bulk electrolyte, and single-pass carbon efficiency (SPCE) of the 6.2%-Pd-Cu with state-of-art CO2RR catalysts.
Follow the Topic
-
Nature Catalysis
This journal brings together researchers from across all chemistry and related fields, publishing work on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, incorporating both fundamental and applied studies.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in