Another twist in the saga of forever chemicals
Published in Bioengineering & Biotechnology, Ecology & Evolution, and Microbiology
The PFAS conundrum
Synthetic per-and polyfluoroalkyl substances (PFASs) have highly desirable surface-active properties, making them attractive for a broad range of consumer, industrial, and military products and processes 1. Many routines in our daily lives would be different without PFASs - no smart phones, no electric cars, inferior stain-resistant fabrics, less durable cosmetics, to give a few examples. Unfortunately, PFASs have structural elements (i.e., perfluorinated carbon atoms) that do not occur naturally, and emerged as recalcitrant (i.e., not biodegradable), global contaminants nicknamed ‘forever chemicals’. Hundreds to thousands of different types of PFASs have entered the environment, and at least some of them are of potential concern for human and environmental health. To strike a chord with everyone reading this, PFASs are found in the blood of nearly every person on planet Earth, very likely including yours.
Unexpected microbe – PFAS interactions
Finding natural processes that destroy PFASs is a high priority goal. We explored if bacteria could transform n:3 fluorotelomer carboxylic acids (FTCAs), a subgroup of polyfluoroalkyl substances commonly found in the environment. When we analyzed cultures of the soil bacterium Pseudomonas sp. strain 273 exposed to 8:3 FTCA using a mass spectrometry-based lipidomics approach developed in a prior study 2, we made a puzzling observation: the analysis of the mass spectra suggested the presence of glycerophospholipids with one or both acyl tails replaced by intact 8:3 FTCA molecules (Fig. 1).
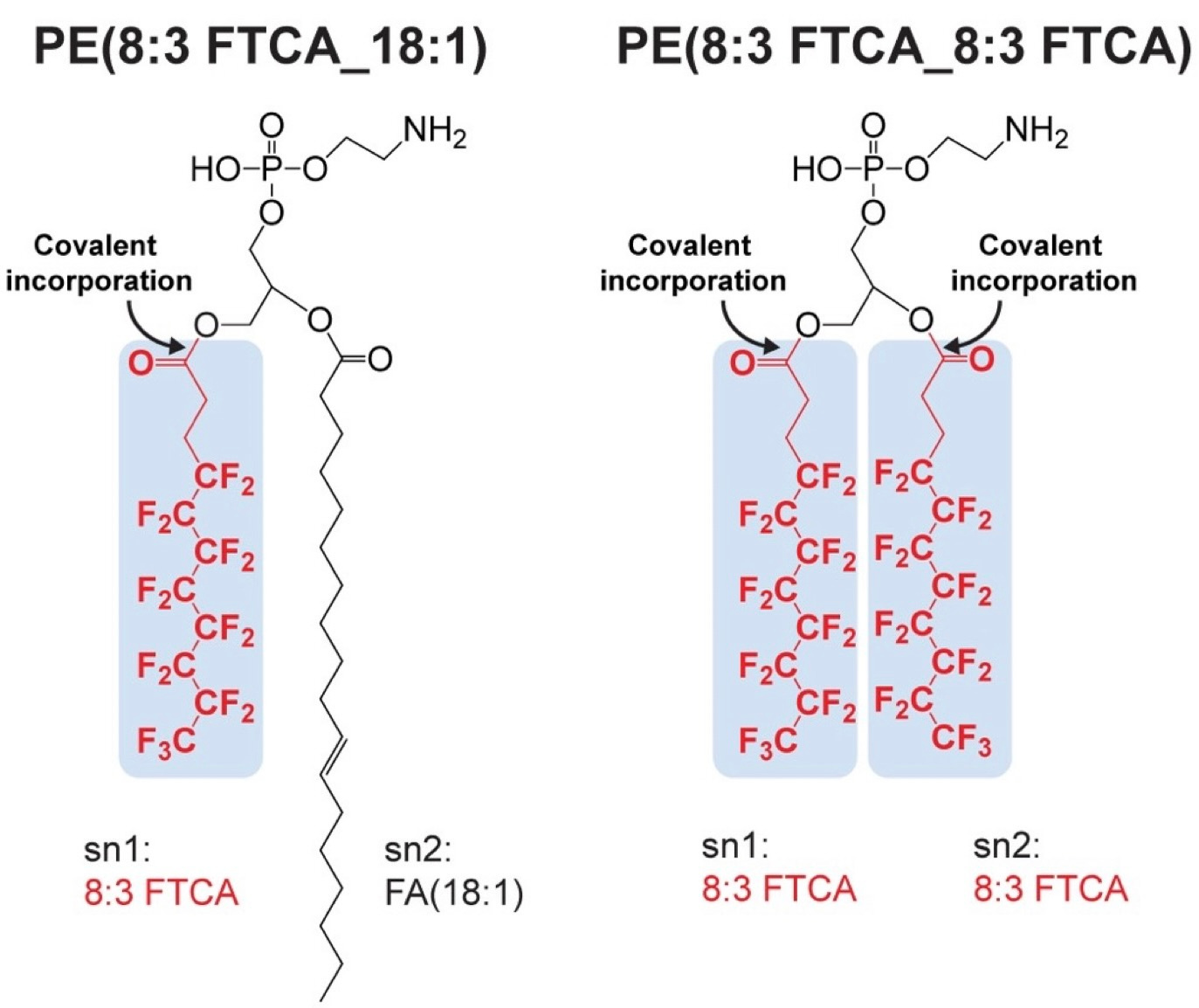
Figure 1. Representative PFASylated phosphatidylethanolamine (PE) species detected in cultures of strain 273 grown in the presence of 8:3 FTCA. Mass spectrometry analysis identified the molecular structures of PE(8:3 FTCA_18:1) (left panel) and PE(8:3 FTCA_8:3 FTCA) (right panel).
Extensive validation in two independent analytical laboratories confirmed that this bacterium covalently incorporates 8:3 FTCA into glycerophospholipids and produces ‘fluoromembranes’. Pseudomonas sp. strain 273 covalently incorporates C8 to C12 n:3 FTCAs, with the highest incorporation percentage observed for C10 (7:3 FTCA), which is a PFAS commonly observed in the environment. Experiments with other bacteria, including members of the human gut microbiome, demonstrated that the covalent incorporation of n:3 FTCAs into lipids is a shared feature, suggesting fluoromembrane formation is a common phenomenon. Remarkably, the formation of fluoromembranes had no apparent toxic effects on the bacterium, and strain 273 grew equally well in medium with and without 8:3 FTCA.
Covalent incorporation of n:3 FTCA – mechanisms are beginning to unravel
n:3 FTCAs are polyfluorinated fatty acids, and the enzymes bacteria utilize to synthesize glycerophospholipids apparently do not discriminate between polyfluorinated and non-fluorinated fatty acids. Using Pseudomonas sp. strain 273 genome information and protein expression data, we identified putative genes and proteins involved in the covalent incorporation of n:3 FTCAs into the glycerophospholipid pool, which could potentially serve as biomarkers for this incorporation process. Notably, covalent incorporation of n:3 FTCAs is fundamentally distinct from partitioning of PFASs into lipid bilayers 3. The physicochemical properties allow PFASs to reversibly associate with lipid bilayers, a process based on hydrophobic interactions and van der Waals forces that does not involve covalent bond and fluoromembrane formation.
Unrecognized PFAS pools
The experimental efforts with strain 273 demonstrated that non-fluorinated a- and b-carbons and a carboxylic acid functional group are preferred structural elements for covalent incorporation into glycerophospholipids, and n:3 FTCA chain length and organismal features (e.g., enzyme specificity) affect incorporation efficiency. A diversity of polyfluoroalkyl substances commonly encountered in the environment are susceptible to biotransformation leading to the formation of n:3 FTCAs, which we show are subject to incorporation into the glycerophospholipid pool. Bacteria are abundant in soils and groundwater aquifers, and a substantial PFAS fraction could potentially reside covalently incorporated in biomass.
Implications for PFAS monitoring
Many countries have set Maximum Contaminant Levels (MCLs) for select PFASs in the parts per trillion (ng per liter) range, and mandate PFAS monitoring of aqueous matrices (e.g., drinking water, groundwater, wastewater), solid matrices (soil, sediment, biosolids), air emissions, foods, and consumer products. The recovery of PFASs from environmental matrices includes extraction steps; however, PFASs covalently incorporated into biomass are generally not captured, suggesting the current methodology may underestimate the true PFAS burden.
Implications for environmental fate and transport of PFASs
A comprehensive understanding of the processes that control migration of PFASs following their release into the environment is needed for faithfully predicting their distribution and fate. The covalent incorporation of n:3 FTCAs into glycerophospholipids would reduce their respective aqueous phase concentrations and mobility, the latter now being governed by the transport of bacterial cells rather than solutes. Bacteria with fluoromembranes could represent a PFAS pool that is currently unaccounted for, and measuring this pool, as well as determining biomass turnover and the stability of fluoromembranes, should be accomplished. Notably, efforts to close PFAS mass balances are often unsuccessful, possibly due to unidentified pools of organofluorine that escape detection with contemporary analytical approaches.
Fundamental discovery with practical and human health implications
Fire extinguishing training exercises using PFAS-laden aqueous film-forming foam (AFFF) has resulted in environmental contamination, and many military installations are impacted. To address this issue, the Strategic Environmental Research and Development Program (SERDP), one of two U.S. Department of Defense's installation innovation programs, supports research that leads to a better understanding of the behavior of PFASs in the environment. We are collaborating with practitioners and environmental firms to bring our findings into context of environmental monitoring and cleanup, and to advance technologies that protect environmental health, meet regulatory requirements, and can be cost-efficiently implemented.
The discovery will directly impact human health-related research. Oral ingestion is a major route of human exposure to PFASs, and our findings suggest that bacteria of the human gut microbiome sequester certain PFASs into their membranes. Fecal excretion of ‘fluorinated’ bacterial biomass (i.e., bacteria with fluoromembranes) would expel PFASs and thus protect the host from PFAS exposure.
These examples illustrate why exploring PFAS – microbiome interactions, in the human gut and in the environment, is relevant for environmental cleanup and the protection of human health.
Looking ahead
Bacterial biomass with fluoromembranes represents a heretofore unrecognized PFAS pool that demands quantitative assessment. Unfortunately, analytical standards of PFASylated glycerophospholipid species are currently unavailable, and their synthesis must be achieved for monitoring fluoromembrane pool sizes and dynamics. Refined analytical protocols and monitoring programs that account for all PFAS pools will reveal pathways and entry points of PFASs into the environment and/or supply chains and allow more effective measures for preventing environmental and human exposure to PFASs.
Glycerophospholipids are the building blocks of the lipid bilayer (i.e., the membrane), and the incorporation of PFASylated glycerophospholipid species will impact membrane functionality. Detailed studies will reveal adaptive mechanisms allowing bacteria to cope with this disruption. When we presented this work at professional conferences, a frequently asked question has been “Do human cells have PFASylated lipids?”. The enzymatic machineries for glycerophospholipid biosynthesis in bacteria and eukaryotes differ; however, this is an intriguing possibility, and future work will explore if promiscuous incorporation of FTCAs occurs in higher organisms.
A student’s curiosity – essential for making unexpected discoveries
Crucial for the discovery of bacterial fluoromembrane synthesis detailed on our Nature Microbiology paper (doi.10.1038/s41564-026-02301-x) was the curiosity, motivation, and perseverance of then graduate student Yongchao Xie (Fig. 2).

Dr. Xie is now an Associate Professor in the State Key Laboratory of Pollution Control and Resource Reuse, School of Environment, at Nanjing University in China, where he continues the exploration of microbe - PFAS interactions.
References
- Evich, M.G. et al. Per- and polyfluoroalkyl substances in the environment. Science 375, eabg9065 (2022). DOI: 10.1126/science.abg9065
- Xie, Y. et al. Pseudomonas sp. strain 273 incorporates organofluorine into the lipid bilayer during growth with fluorinated alkanes. Environ. Sci. Technol. 56, 8155-8166 (2022). DOI: 10.1021/acs.est.2c01454
- Fitzgerald, N.J. et al. Partitioning and accumulation of perfluoroalkyl substances in model lipid bilayers and bacteria. Environ. Sci. Technol. 52, 10433-10440 (2018). DOI: 10.1021/acs.est.8b02912
Follow the Topic
-
Nature Microbiology
An online-only monthly journal interested in all aspects of microorganisms, be it their evolution, physiology and cell biology; their interactions with each other, with a host or with an environment; or their societal significance.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in