Birth and blood loss: an epigenetic perspective on oxytocin use during labor
Published in Healthcare & Nursing
Oxytocin and childbirth
Until the early 1900s, midwives and obstetricians relied upon herbs1 known to cause uterine contractions when it was necessary to stimulate labor or treat postpartum bleeding. However, when conducting an experiment on a cat in 1906, Sir Henry Dale, an English physiologist, discovered that a substance produced by the pituitary gland caused uterine contraction.2 Work in the 1950s by American biochemist, Vincent du Vigneaud, led to discovery of the amino acid sequence and structure for a substance in the secretion.3,4 This Nobel Prize winning revelation quickly led to standardization, preparation and widespread clinical use of the medication, oxytocin. Nearly 70 years later, the majority of individuals giving birth in a US hospital will receive oxytocin (Pitocin®, Syntocinon®) during labor either for labor induction or accelerating natural labor.5 Additional oxytocin is given after delivery of the newborn to contract the empty uterus, thus constricting the blood vessels that supplied the placenta, and minimize maternal blood loss.6,7
“Medicalization” of the birth process has contributed, in part, to increased survival of newborns in the setting of prolonged labor or other obstetric conditions (e.g., high blood pressure) and can be lifesaving for the mother, particularly during a postpartum hemorrhage (at least 1 liter of blood loss). However, overuse of oxytocin has been raised as a concern as well due to potential risks, particularly when delivery does not need to be expedited.8–10 Because oxytocin can act quickly on the oxytocin receptors (OXTR) on uterine muscle tissue, it can cause excessively frequent or lengthy contractions leading to decreased blood flow to the placenta and decelerations in the fetal heart rate.11 Conversely, many studies have linked longer oxytocin exposure with reduced contractility due to the tissue losing responsiveness over time, which is associated with Cesarean birth and/ or increased rates of postpartum hemorrhage.12–14
Oxytocin does not work equally well in every body
Over the years, we have learned that there is a wide variety in individual responses to pharmacologic oxytocin, for example the gestational age of the pregnancy or the person’s body mass index is associated with differences in sensitivity.15,16 What has not been examined closely is the role of innate individual differences in OXTR / oxytocin function. Put another way, if a person comes into their pregnancy and birth process with less responsive OXTR, it might help us understand why some people have weaker uterine contractions, longer labors and why these people also experience hemorrhage more often.
The hypothesis for our study was stimulated by a large body of research examining OXTR/ oxytocin variation relative to brain function, behavior and broader physiologic regulation.17–19 One kind of variation is an epigenetic modification of the OXTR gene, namely DNA methylation. Methylation is a chemical modification on a gene but not a change in the DNA code itself—typically methylation is retained during normal cell replication. As a whole, prior studies indicate that greater OXTR methylation reduces gene expression and subsequent oxytocin responses or oxytocin-linked behaviors.20 These lines of study also report how early life adversity or protective features of the social environment may be a way by which methylation occurs and the epigenome is shaped.21–23
.png)
Epigenetic modification of OXTR underpins uterine response to oxytocin
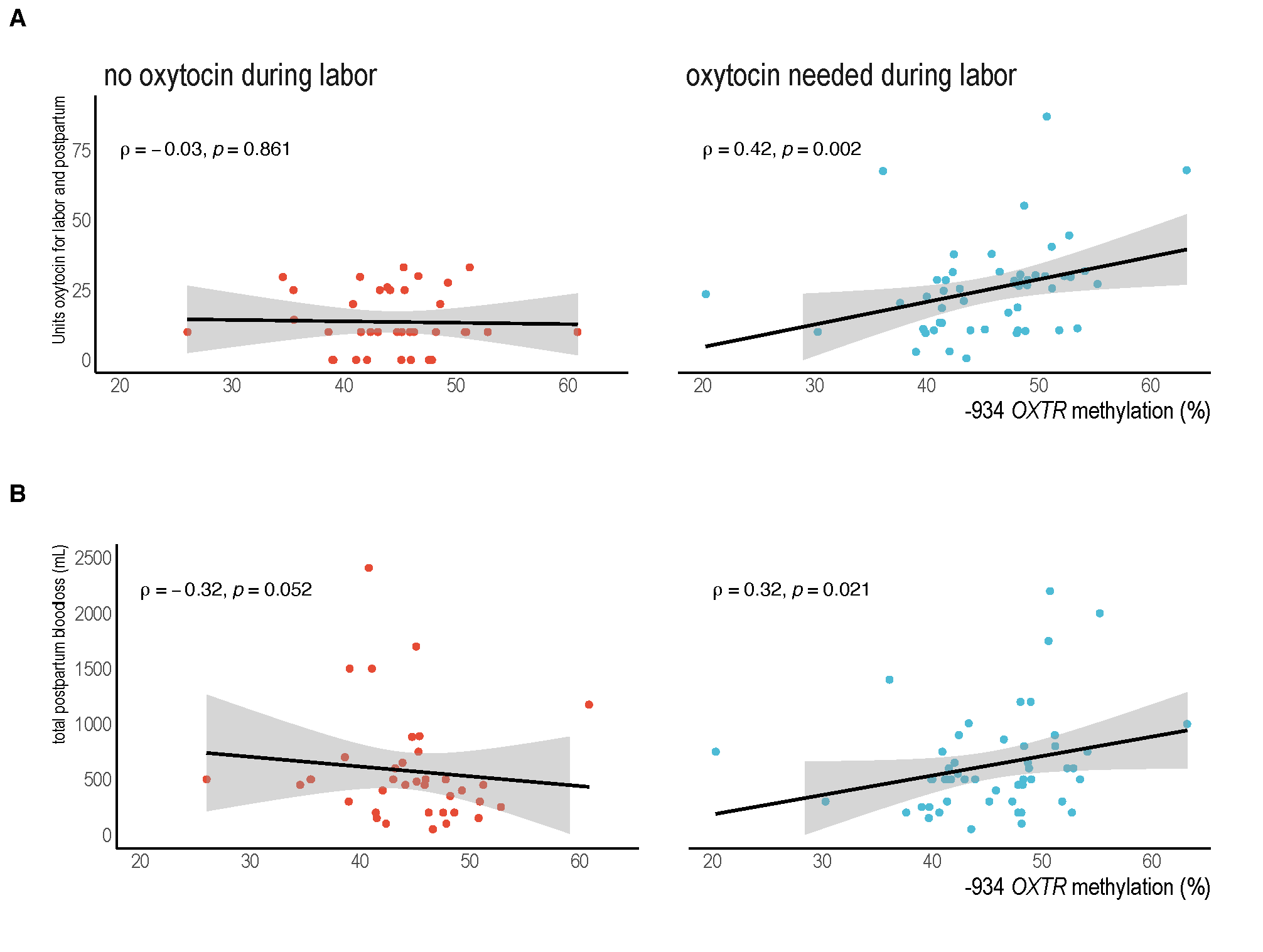
Using a case-control study design, we sought to understand if OXTR methylation might be linked to how well oxytocin stimulates uterine contractions and controls postpartum bleeding. To do this work, we enrolled individuals who had a term vaginal birth and either experienced heavy postpartum bleeding linked to weak uterine contraction after birth (n=69) or those with typical blood loss (n=50). DNA methylation from maternal blood samples was measured at a specific site of the OXTR gene, shown in human tissue to be linked to changes in gene expression.20
We show that higher OXTR methylation is associated with greater oxytocin needs as well as the severity of maternal blood loss. Specifically, the participants with oxytocin-stimulated labor lost nearly 200mL more blood per increment of higher DNA methylation. Those with higher methylation and oxytocin stimulation also have nearly three times the relative risk for postpartum hemorrhage as those who did not have oxytocin during in labor. Data also demonstrate that higher dosage of oxytocin is linked to giving birth for the first time or having a higher body mass index, but these characteristics are unrelated to the level of methylation itself. These key findings fit with our understanding from the literature: that greater DNA methylation is generally linked to reduced gene expression and fewer receptors. Therefore, with greater methylation, more medication is needed to achieve effective contractions and excessive bleeding is more likely.
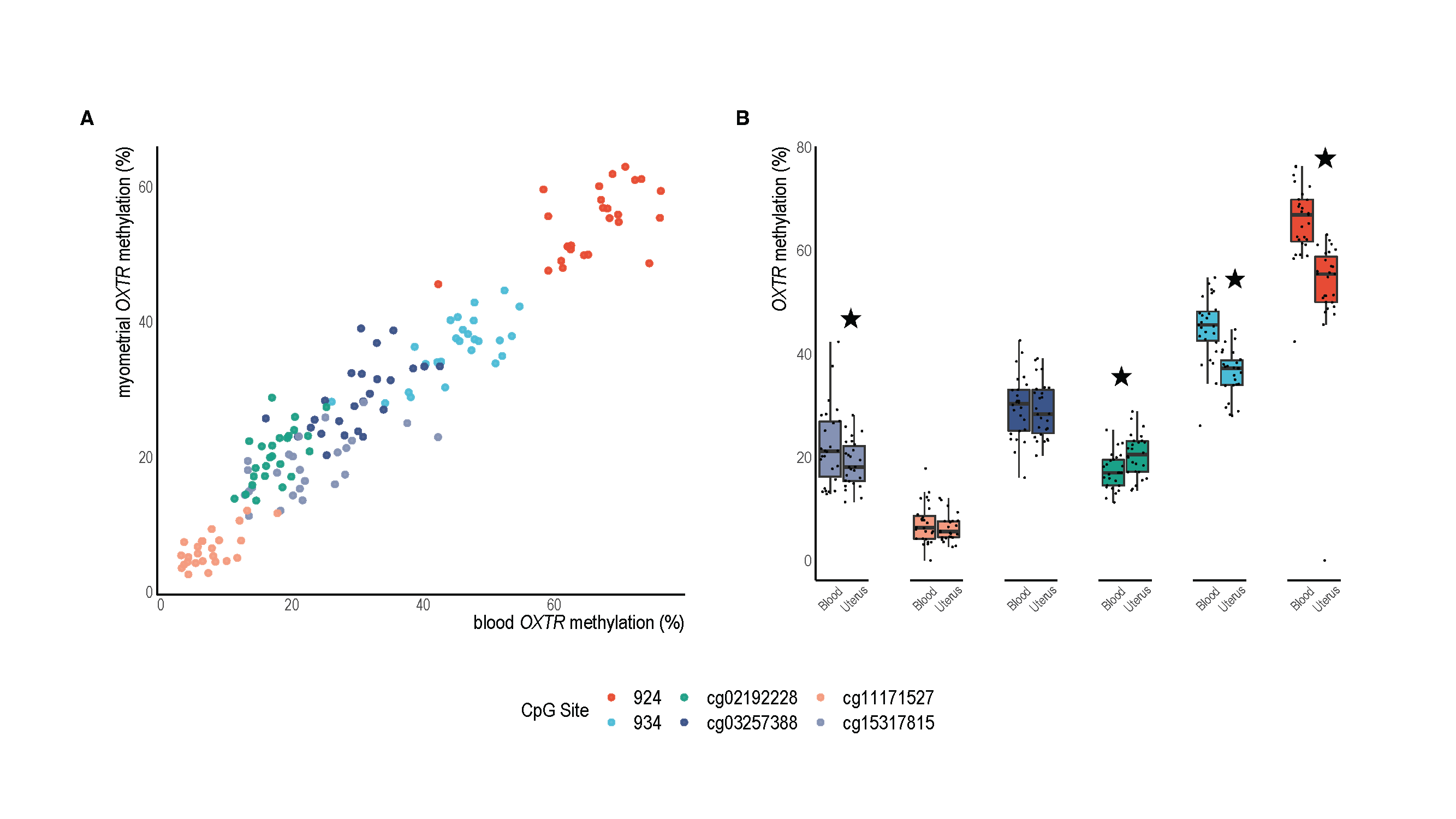
In our paper, we also report on another experiment where we correlate DNA methylation from matched samples of blood and uterine tissue from individuals undergoing planned Cesarean birth to determine if our blood-based biomarker generally reflects the methylation status of the uterine muscle (which it does at several methylation sites).
Next steps in our investigation
The findings from these studies highlight a path toward improving our use of oxytocin during childbirth and addressing hemorrhage from a pharmaco-epigenetic perspective. The interest in precision methods for addressing perinatal conditions is growing,24 though the attention is typically fetal/newborn health focused. In an effort to improve maternal-specific outcomes, we will continue to study OXTR methylation and other variations25 in the gene as prospective biomarkers for oxytocin sensitivity during childbirth and hemorrhage risk. Someday we may be able to predict how well a person will respond to this lifesaving medication before labor starts and develop new methods for prevention or intervention in obstetric practice.
Equally compelling are the numerous opportunities to study the precipitating factors underpinning more or less DNA methylation. We can use this biomarker to study the connection between early life experience, social adversity and maternal health. The persistent inequity in maternal health outcomes in the US has gained the attention of public advocates and policy makers in the last few years.26 The inequity in maternal survival and health through pregnancy and birth is associated with social adversity and other social determinants of health.27–29 Showing that OXTR methylation is a biologically plausible contributor to maternal morbidity (postpartum hemorrhage) underscores a need to understand how epigenetic programming could be influencing this effect and also exacerbating some disparities.
References
- Gruber CW, O’Brien M. Uterotonic Plants and their Bioactive Constituents. Planta Med. 2011;77(3):207-220. doi:10.1055/s-0030-1250317
- Dale HH. On some physiological actions of ergot. J Physiol. 1906;34(3):163-206.
- du Vigneaud V. Trail of Sulfur Research: from Insulin to Oxytocin. Science. 1956;123(3205):967-974. doi:10.1126/science.123.3205.967
- Ottenhausen M, Bodhinayake I, Banu MA, Stieg PE, Schwartz TH. Vincent du Vigneaud: following the sulfur trail to the discovery of the hormones of the posterior pituitary gland at Cornell Medical College. Journal of neurosurgery. Published online October 2015:1-5.
- Page K, McCool WF, Guidera M. Examination of the Pharmacology of Oxytocin and Clinical Guidelines for Use in Labor. Journal of Midwifery and Women’s Health. 2017;200(1):9. doi:10.1111/jmwh.12610
- Salati JA, Leathersich SJ, Williams MJ, Cuthbert A, Tolosa JE. Prophylactic oxytocin for the third stage of labour to prevent postpartum haemorrhage. Cochrane Database of Systematic Reviews. 2019;(4). doi:10.1002/14651858.CD001808.pub3
- Begley CM, Gyte GM, Devane D, McGuire W, Weeks A, Biesty LM. Active versus expectant management for women in the third stage of labour. Cochrane Database of Systematic Reviews. 2019;(2). doi:10.1002/14651858.CD007412.pub5
- Simpson KR, Knox GE. Oxytocin as a high-alert medication: Implications for perinatal patient safety. MCN The American Journal of Maternal/Child Nursing. 2009;34(1):8-15. doi:10.1097/01.NMC.0000343859.62828.ee
- Häggsgård C, Persson EK. Management of oxytocin for labour augmentation in relation to mode of birth in Robson group 1. Midwifery. 2020;90:102822. doi:10.1016/j.midw.2020.102822
- Monks DT, Palanisamy A. Oxytocin: at birth and beyond. A systematic review of the long-term effects of peripartum oxytocin. Anaesthesia. 2021;76(11):1526-1537. doi:10.1111/anae.15553
- Heuser CC, Knight S, Esplin MS, et al. Tachysystole in term labor: incidence, risk factors, outcomes, and effect on fetal heart tracings. Am J Obstet Gynecol. 2013;209(1):32.e1-6. doi:10.1016/j.ajog.2013.04.004
- Balki M, Ramachandran N, Lee S, Talati C. The recovery time of myometrial responsiveness after oxytocin-induced desensitization in human myometrium in vitro. Anesthesia and Analgesia. 2016;122(5):1508-1515. doi:10.1213/ANE.0000000000001268
- Phaneuf S, Asboth G, Carrasco M, et al. Desensitization of oxytocin receptors in human myometrium. Hum Reprod Update. 1998;4(5):625-633.
- Erickson EN, Carlson NS. Predicting Postpartum Hemorrhage After Low-Risk Vaginal Birth by Labor Characteristics and Oxytocin Administration. Journal of Obstetric, Gynecologic & Neonatal Nursing. 2020;49(6):549-563. doi:10.1016/j.jogn.2020.08.005
- Carlson NS, Corwin EJ, Lowe NK. Oxytocin Augmentation in Spontaneously Laboring, Nulliparous Women: Multilevel Assessment of Maternal BMI and Oxytocin Dose. Biological Research for Nursing. 2017;19(4):382-392. doi:10.1177/1099800417701831
- Kim SC, Lee JE, Kang SS, Yang HS, Kim SS, An BS. The regulation of oxytocin and oxytocin receptor in human placenta according to gestational age. J Mol Endocrinol. 2017;59(3):235-243. doi:10.1530/JME-16-0223
- Feldman R, Monakhov M, Pratt M, Ebstein RP. Oxytocin Pathway Genes: Evolutionary Ancient System Impacting on Human Affiliation, Sociality, and Psychopathology. Biological Psychiatry. 2016;79(3):174-184. doi:10.1016/j.biopsych.2015.08.008
- Carter CS, Kenkel WM, MacLean EL, et al. Is Oxytocin “Nature’s Medicine”? Pharmacol Rev. 2020;72(4):829-861. doi:10.1124/pr.120.019398
- Quintana DS, Guastella AJ. An Allostatic Theory of Oxytocin. Trends in Cognitive Sciences. Published online 2020. doi:10.1016/j.tics.2020.03.008
- Danoff JS, Wroblewski KL, Graves AJ, et al. Genetic, epigenetic, and environmental factors controlling oxytocin receptor gene expression. Clin Epigenetics. 2021;13(1):23. doi:10.1186/s13148-021-01017-5
- Krol KM, Moulder RG, Lillard TS, Grossmann T, Connelly JJ. Epigenetic dynamics in infancy and the impact of maternal engagement. Sci Adv. 2019;5(10):eaay0680. doi:10.1126/sciadv.aay0680
- Danoff JS, Connelly JJ, Morris JP, Perkeybile AM. An epigenetic rheostat of experience: DNA methylation of OXTR as a mechanism of early life allostasis. Compr Psychoneuroendocrinol. 2021;8:100098. doi:10.1016/j.cpnec.2021.100098
- Perkeybile AM, Carter CS, Wroblewski KL, et al. Early nurture epigenetically tunes the oxytocin receptor. Psychoneuroendocrinology. 2019;99:128-136. doi:10.1016/J.PSYNEUEN.2018.08.037
- Stevenson DK, Wong RJ, Aghaeepour N, et al. Towards personalized medicine in maternal and child health: integrating biologic and social determinants. Pediatr Res. 2021;89(2):252-258. doi:10.1038/s41390-020-0981-8
- Erickson EN, Krol KM, Perkeybile AM, Connelly JJ, Myatt L. Oxytocin receptor single nucleotide polymorphism predicts atony-related postpartum hemorrhage. BMC Pregnancy and Childbirth. 2022;22(1):884. doi:10.1186/s12884-022-05205-w
- U. S. Government Accountability Office. Maternal Health: Outcomes Worsened and Disparities Persisted During the Pandemic. Accessed January 18, 2023. https://www.gao.gov/products/gao-23-105871
- Erickson EN, Carlson NS. Maternal Morbidity Predicted by an Intersectional Social Determinants of Health Phenotype: A Secondary Analysis of the NuMoM2b Dataset. Reprod Sci. 2022;29(7):2013-2029. doi:10.1007/s43032-022-00913-2
- Gillispie-Bell V. The Contrast of Color: Why the Black Community Continues to Suffer Health Disparities. Obstet Gynecol. 2021;137(2):220-224. doi:10.1097/AOG.0000000000004226
- Crear-Perry J, Correa-de-Araujo R, Lewis Johnson T, McLemore MR, Neilson E, Wallace M. Social and Structural Determinants of Health Inequities in Maternal Health. Journal of Women’s Health. 2021;30(2):230-235. doi:10.1089/jwh.2020.8882
Follow the Topic
-
Communications Medicine
A selective open access journal from Nature Portfolio publishing high-quality research, reviews and commentary across all clinical, translational, and public health research fields.
Related Collections
With Collections, you can get published faster and increase your visibility.
Public health and health governance in China
Publishing Model: Open Access
Deadline: Jul 31, 2026
Life Course Epidemiology
Publishing Model: Open Access
Deadline: Jun 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in