Circulating tumour cells and PD-L1-positive small extracellular vesicles: THE liquid biopsy combination for prognostic information in patients with metastatic non-small cell lung cancer
Published in Cancer
The term of “liquid biopsy” was proposed for the first time by Catherine Alix-Panabières and Klaus Pantel in 2010 [1]. Since then, this approach has received tremendous attention in oncology because it offers the opportunity to monitor cancer in a non-invasive manner using a blood sample. Circulating tumour cells (CTCs), circulating tumour DNA (ctDNA), and most recently, extracellular vesicles (EVs), immune cells and circulating microbiota are fascinating complementary analytes for liquid biopsy [2, 3]. These circulating biomarkers are detected mainly in blood, but also in all other physiological fluids (e.g., urine, bone marrow, cerebrospinal fluids) and can be used concomitantly in various cancer care approaches. These circulating biomarkers are currently employed in many clinical trials, but their clinical utility is still investigated with promising initial results. Besides their predictive function, liquid biopsy biomarkers have many potential clinical applications, from early cancer detection to cancer staging improvement, minimal residual disease monitoring, real-time treatment response monitoring and identification of therapeutic targets and resistance mechanisms [2].
The inspiration for this new research came from our idea [4] that it is important to incorporate all possible liquid biopsy indicators if we want to fully understand each patient’s clinical picture in the context of personalized medicine and targeted therapy. Indeed, due to cancer complexity and heterogeneity, it is not reasonable to rely on a single biomarker. Based on when it is detected as well as the cancer type and stage, each biomarker can provide distinct and complementary information [4]. This knowledge can be of great use in cancer management.
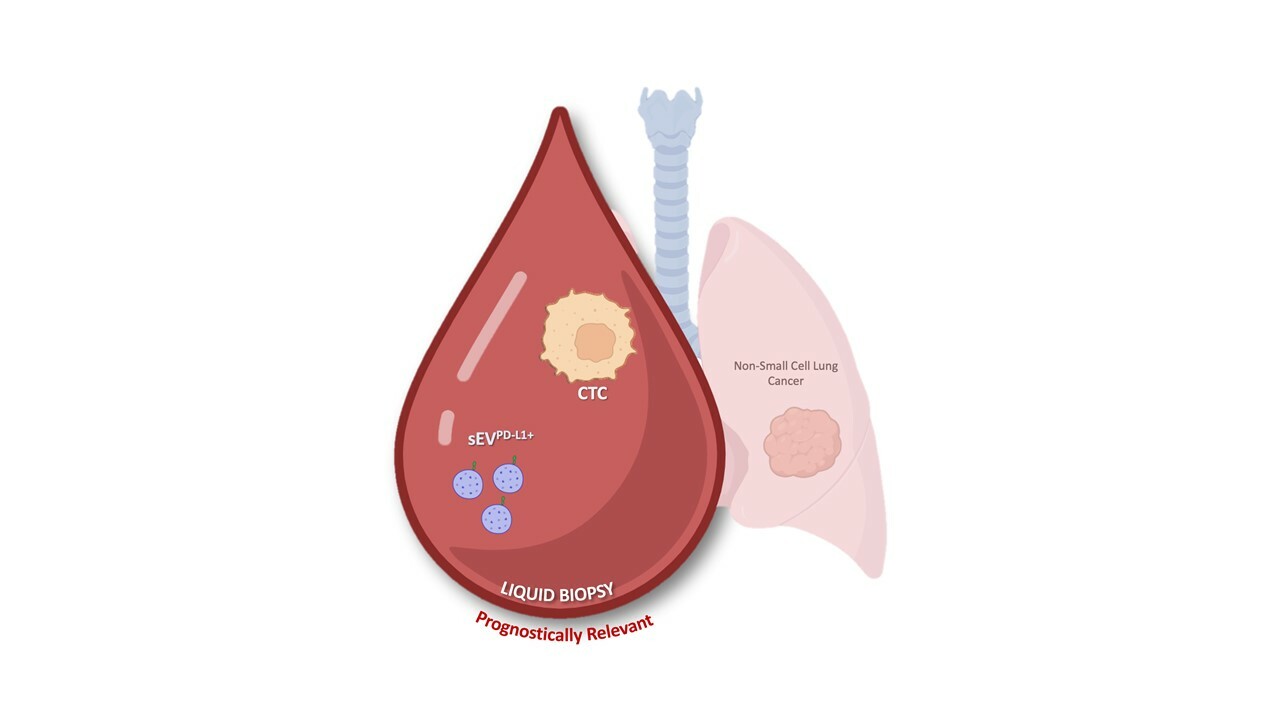
Therefore, we investigated the prognostic value of different liquid biopsy biomarkers (CTCs, PD-L1+ CTCs, small EVs (sEVs), PD-L1+ sEVs and ctDNA) alone and in combination in a cohort of patients with non-small cell lung cancer (NSCLC), regardless of their cancer treatment, subtype or stage, to determine whether their combination provides more precise prognostic information. Our findings indicate that CTCs deliver robust prognostic information, independently of NSCLC molecular subtype and treatment [5]. PD-L1+ sEV, but not total sEV, concentration also offered prognostic information, especially when combined with the CTC status (Figure 1). On the other hand, ctDNA mutation detection had no predictive value because the study was performed in the whole cohort, and not after stratification in function of the treatment type. Notably, patients without CTCs and high PD-L1+ sEV concentration exhibited shorter progression-free survival and patients with high PD-L1+ sEV concentration and CTCs had the worst overall survival .
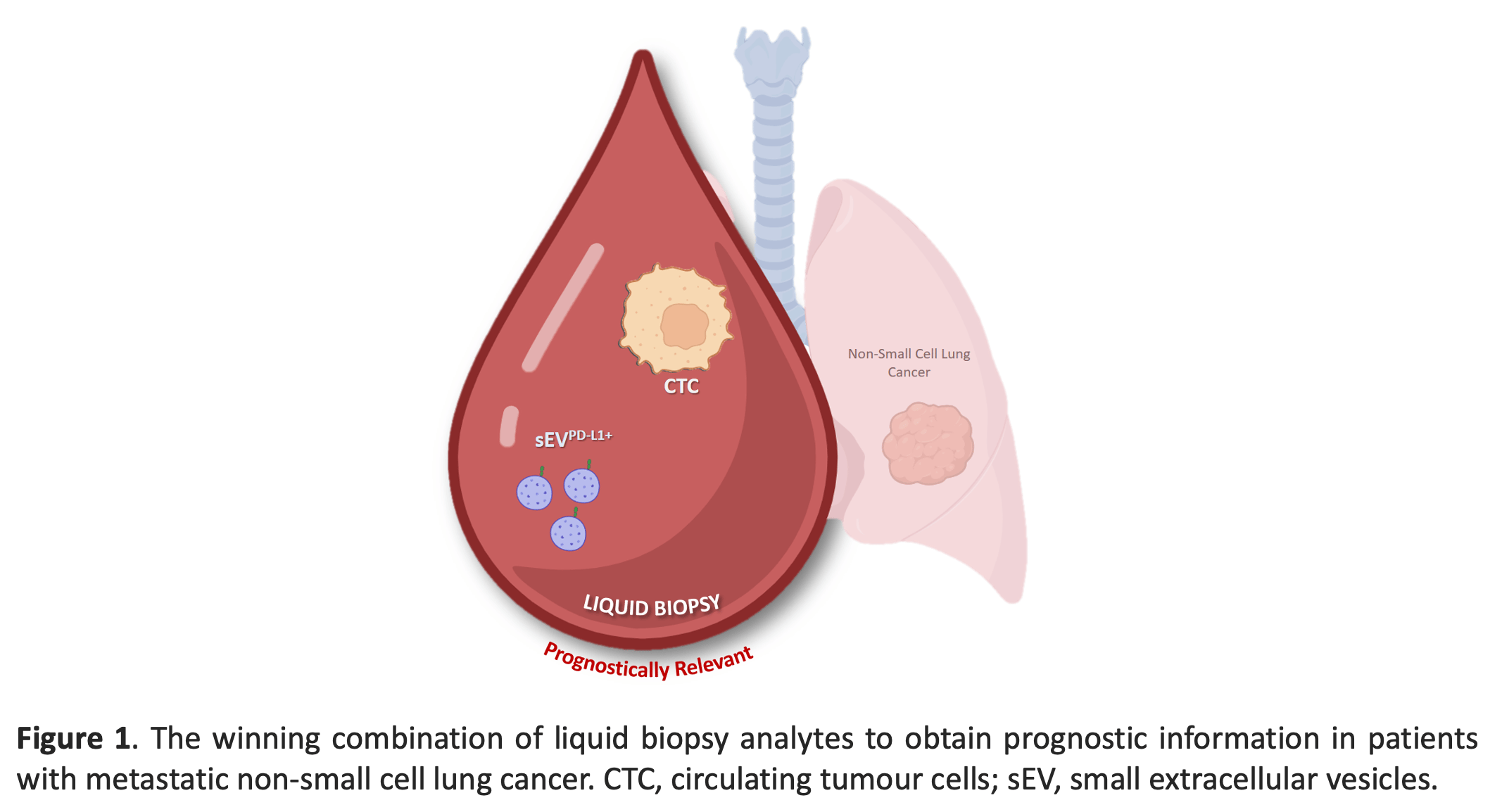
This proof-of-concept study indicates that all circulating biomarkers must be viewed as complementary for personalized medicine and targeted therapy approaches. More interventional clinical trials with a larger number of patients are needed to confirm our findings and assess whether this combination of liquid biopsy analytes helps to predict prognosis in clinical practice. Therefore, future clinical studies should prioritize the simultaneous analysis of multiple liquid biopsy biomarkers to personalize treatment in each patient.
For this purpose, the international consortium European Liquid Biopsy Society (ELBS; www.elbs.eu), which includes 84 partners from academia and industry (in 2024), has been established to standardize and organize large multicentre clinical trials in order to promote liquid biopsy implementation in clinical practice. Finally, this new multi-omics liquid biopsy approach can lead to the development of an algorithm that combines data from different liquid biopsy analytes to obtain precise tumour information for guiding therapeutic decision-making.
References
- Pantel, K. and C. Alix-Panabières, Circulating tumour cells in cancer patients: challenges and perspectives. Trends Mol Med, 2010. 16(9): p. 398-406.
- Alix-Panabières, C. and K. Pantel, Liquid Biopsy: From Discovery to Clinical Application. Cancer Discovery, 2021. 11(4): p. 858-873.
- Edsjö, A., et al., Precision cancer medicine: Concepts, current practice, and future developments. J Intern Med, 2023. 294(4): p. 455-481.
- Alix-Panabières, C., The future of liquid biopsy. Nature, 2020. 579(7800): p. S9.
- Sinoquet, L., et al., Programmed Cell Death Ligand 1-Expressing Circulating Tumor Cells: A New Prognostic Biomarker in Non-Small Cell Lung Cancer. Clin Chem, 2021. 67(11): p. 1503-1512.
Follow the Topic
-
British Journal of Cancer
This journal is devoted to publishing cutting edge discovery, translational and clinical cancer research across the broad spectrum of oncology.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in