Decoding the Physical Language of Myelination
Published in Bioengineering & Biotechnology, Materials, and Neuroscience
Why mechanics matters in the brain
In our mechanobiology lab, we study how cells "feel" their surroundings, and how physical cues shape their behaviour. The brain makes this especially intriguing. It is one of the softest tissues in the body, yet it is packed with billions of intricate microscopic wires called axons. These axons are wrapped in insulating myelin produced by specialised cells known as oligodendrocytes.
As a mechanical engineer interested in the mechanics of life, I have always been fascinated by how oligodendrocytes wrap around such soft structures. In earlier work, I showed that cells themselves behave like soft, porous and elastic materials (Moeendarbary et al., 2013). This made me particularly curious about the role of axonal physical properties, such as stiffness and size, in dictating the dynamic wrapping behaviour of oligodendrocytes.
The challenge: untangling coupled signals
Previous studies have shown that oligodendrocytes are highly sensitive to physical cues, such as axon stiffness and diameter. However, in the brain, these signals are coupled. A cell experiences many signals simultaneously, and we cannot easily change the stiffness of an axon without also altering its chemistry or size.
At the same time, recent research has suggested that age-related changes in the oligodendrocyte microenvironment may influence their behaviour more strongly than changes in the oligodendrocytes themselves (Segel et al., 2019). This matters because ageing and many neurological diseases are accompanied by changes in brain mechanics (Hall et al., 2023), which can alter how oligodendrocytes behave.
This raised a key question for us: how do oligodendrocytes interpret individual physical signals when those signals are always intertwined in the brain?
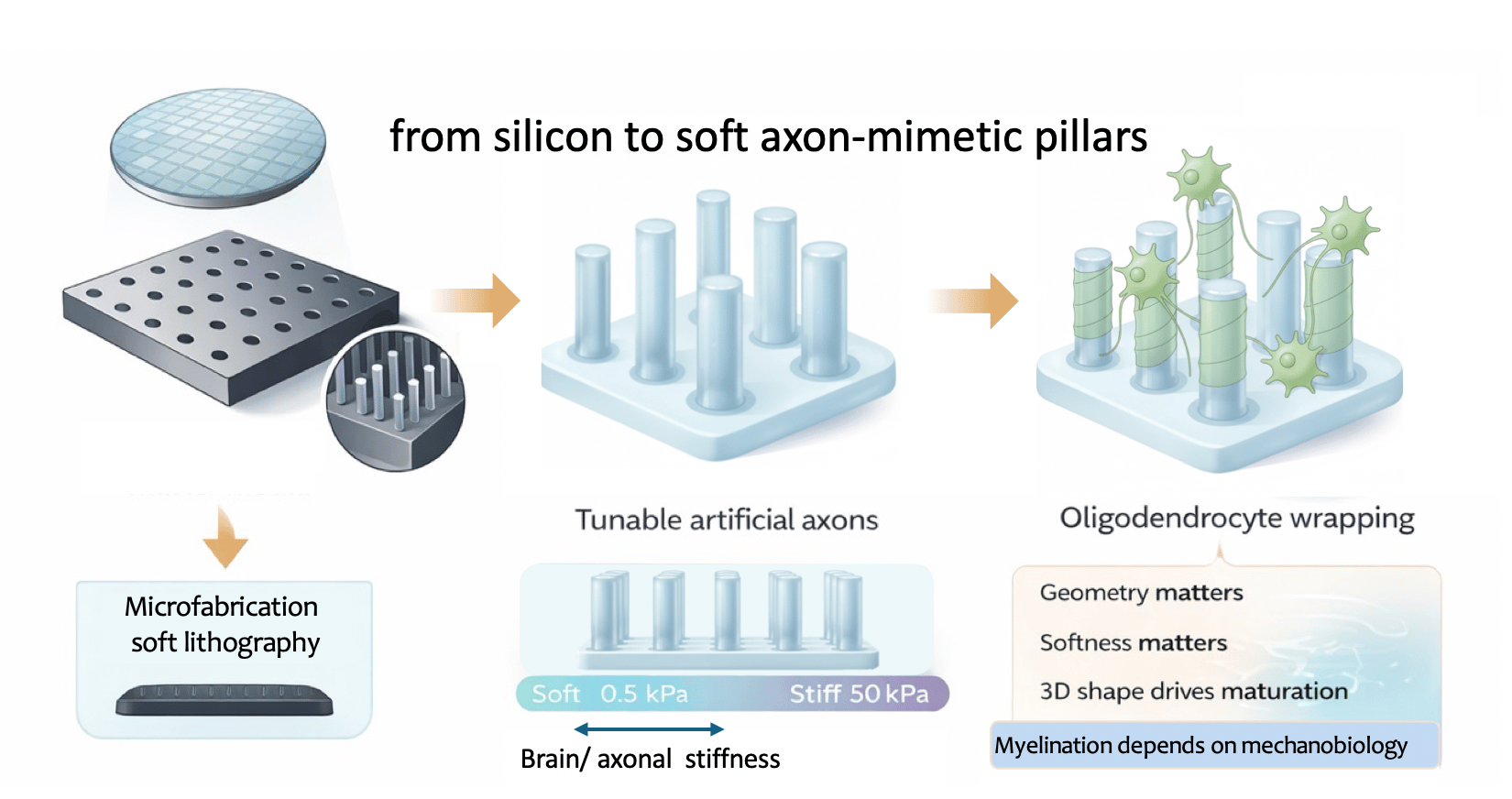
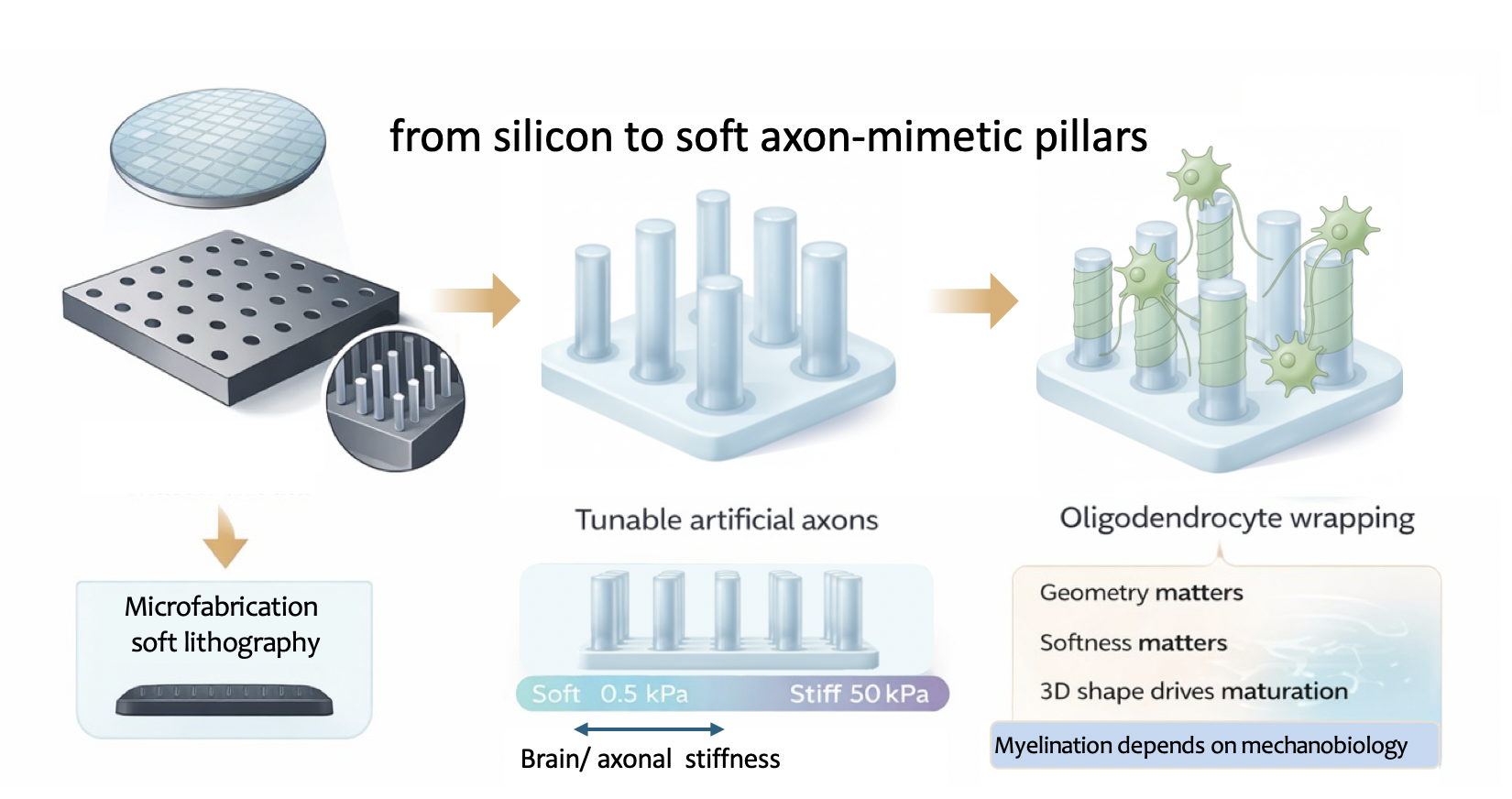
Building axon-mimetic pillars
To answer that, we needed a way to decouple them. We were motivated to create an assay in which axon-like stiffness and geometry could be finely tuned to reproduce features of ageing or neurological disease. Such a system could help us study disease mechanisms in a much more controlled and physiologically relevant way.
To mimic the brain’s architecture with high precision, we borrowed Deep Reactive Ion Etching (DRIE) from the semiconductor industry, using it to etch vertical micro-wells into silicon that served as moulds for our micropillars. Using high-precision lithography, we created arrays in which we could vary the diameter of these wells (from 2 to 10 µm) and their spacing, and then used the silicon masters to cast soft polyacrylamide hydrogel pillars.
What really made the system work was the material transition from the rigid world of silicon to soft, tunable cylindrical hydrogels that mimic axons.
The moment it worked and the surprises
One of the most exciting moments came when we first looked through the confocal microscope and saw oligodendrocytes extending and wrapping around our hydrogel pillars. After so much fabrication work in the cleanroom (collapsing structures, peeling gels, etc.), seeing the biology and engineering finally come together was transformative for the entire team. This work was driven by the persistence and creativity of my PhD student, Soufian Lasli, whose dedication over several years made this platform possible.
For the first time, we could create "axon-mimetic pillars" that did not just look like axons; they felt like them. Once we began seeding oligodendrocytes onto these arrays, it became clear that geometry and stiffness were not redundant cues but independent regulators of myelination. Oligodendrocytes preferentially wrapped thicker pillars, mimicking what happens in the brain with larger-calibre axons. But this preference was significantly modulated by the substrate stiffness. Even more strikingly, our transcriptomic analysis showed that simply moving a cell from a flat surface to a 3D micropillar environment (even at the same stiffness) triggered a massive shift in gene expression toward a mature, myelinating phenotype. In other words, the shape of the environment alone was enough to push cells toward maturation.
A key lesson for drug discovery
One result that really stayed with us came from the drug experiments. We found that the effects of well-known pro-myelinating compounds were significantly boosted on stiffer, non-physiological substrates but muted on soft, brain-mimicking pillars. For us, this was a reminder of a core mechanobiology principle: the physical context of an assay can change the pharmacological outcome. It suggests that some candidate drugs may appear more promising than they really are simply because they were tested in environments that are too rigid.
Encouragingly, human iPSC-derived oligodendrocytes responded to these mechanical cues just as robustly as rodent cells, forming compact, multi-layered myelin around our hydrogel axons.
Bridging engineering and biology
In many ways, this paper came from treating a biological question like an engineering problem. By borrowing tools from semiconductor and hydrogel fabrication, we built a tunable microenvironment that allowed us to begin decoding the physical language oligodendrocytes use to build the brain’s insulation. We hope this platform can serve as a bridge between mechanobiology and clinical neurology, helping identify therapies that work not just in a dish, but in the soft reality of the human brain.
Looking ahead
Looking ahead, we hope to further develop this system into a scalable screening platform for drug discovery. By more faithfully recapitulating the physical properties of human axons, this assay could help identify therapies that promote myelination in environments that closely resemble the diseased brain.
In particular, we see strong potential for applications in multiple sclerosis and other neurological disorders where myelin repair fails. Ultimately, our goal is to reduce the gap between in vitro success and clinical translation by providing a more predictive and physiologically relevant platform for testing therapeutic candidates.
References
Moeendarbary, E., Valon, L., Fritzsche, M., Harris, A.R., Moulding, D.A., Thrasher, A.J., Stride, E., Mahadevan, L. and Charras, G.T., 2013. The cytoplasm of living cells behaves as a poroelastic material. Nature Materials, 12(3), pp.253-261. https://doi.org/10.1038/nmat3517
Segel, M., Neumann, B., Hill, M.F., Weber, I.P., Viscomi, C., Zhao, C., Young, A., Agley, C.C., Thompson, A.J., Gonzalez, G.A. and Sharma, A., 2019. Niche stiffness underlies the ageing of central nervous system progenitor cells. Nature, 573(7772), pp.130-134. https://doi.org/10.1038/s41586-019-1484-9
Hall, C.M., Lasli, S., Serwinski, B., Djordjevic, B., Sheridan, G.K. and Moeendarbary, E., 2023. Hippocampus of the APPNL–G–F mouse model of Alzheimer’s disease exhibits region-specific tissue softening concomitant with elevated astrogliosis. Frontiers in Aging Neuroscience, 15, p.1212212. https://doi.org/10.3389/fnagi.2023.1212212
Follow the Topic
-
Nature Methods
This journal is a forum for the publication of novel methods and significant improvements to tried-and-tested basic research techniques in the life sciences.
Your space to connect: The Psychedelics Hub
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
Methods development in Cryo-ET and in situ structural determination
Publishing Model: Hybrid
Deadline: Jul 28, 2026







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in