Detection of COVID-19 mRNA vaccines in recently vaccinated patients and the factors associated with persistence in the heart
Published in Biomedical Research
You might have heard of mRNA vaccines as a breakthrough technology that helped fight the COVID-19 pandemic. But do you know how they work and what happens to them after you get vaccinated?
mRNA vaccines are like tiny instruction manuals for your immune system. mRNA (messenger RNA) are molecules that carry genetic information from DNA to the protein-making machinery of the cell. SARS-CoV-2 mRNA vaccines use synthetic mRNA that encodes the spike protein, the part of the virus that attaches to human cells. By injecting this mRNA into your muscle, the vaccine teaches your immune system to recognize and fight the spike protein, without exposing you to the actual virus.
mRNA vaccines did not just happen overnight
Years of mRNA vaccine development culminated to an immediate response to the COVID-19 pandemic. Scientists had to overcome many challenges, such as modifying coronavirus spike protein antigen sequence to make it more stable and effective (1); changing the nucleotides themselves (i.e., 1-methyl-3’-pseudouryldyl) to enhance protein production and cause less immune reactions in cells (2); designing lipid nanoparticles (LNPs) to protect and deliver mRNA into the cells (3-6); and using a special enzyme, T7 polymerase, to produce large amounts of mRNA quickly (7-8). Thanks to years of research and innovation, two SARS-CoV-2 mRNA vaccines were developed expediently: BNT162b2 (BioNTech-Pfizer) and mRNA-1273 (Moderna). Both vaccines produce the full-length SARS-CoV-2 spike protein and have been shown to reduce hospital burden and severity of COVID-19 considerably.
Assays to screen and detect SARS-CoV-2 mRNA vaccines in autopsy tissues
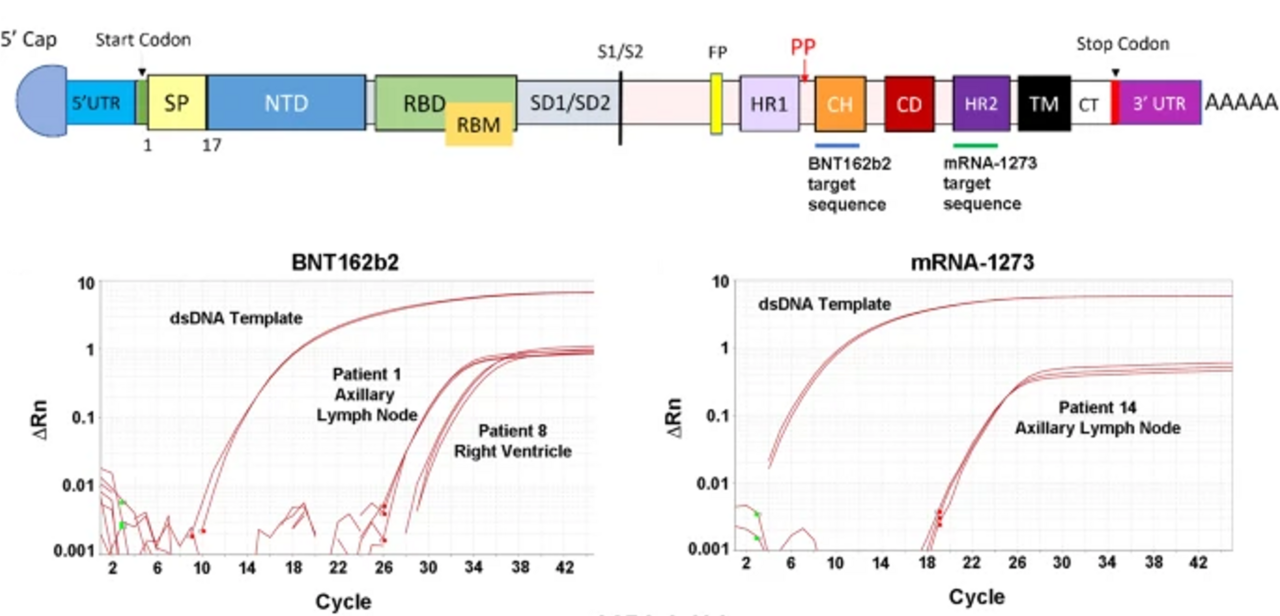
But how long do these vaccines stay in your body and where do they go? This is a question that many people are curious about, but not much is known about it. To find out, our lab developed RT-qPCR assays to detect the presence and amount of BNT162b2 and mRNA-1273 in human tissues after death. We collected samples from different organs, such as lymph nodes, heart, liver, and spleen, from 20 people who had received the vaccine, and 5 people who had not been vaccinated. In none of the vaccinated patients was the cause of death linked to the vaccine.
SARS-CoV-2 mRNA vaccines persist in axillary lymph nodes within 30 days of vaccination
The tissue where we detected the vaccines most was in the axillary lymph nodes, located near the armpit where the injections are given. These lymph nodes are part of the immune system and are key in generating antibody responses. We found that both vaccines were present in most (73%) lymph node samples from people who died within 30 days of vaccination, but not in any of the samples from people who died after 30 days. This is consistent with another study that used a different technique called in-situ hybridization to detect the vaccine in lymph nodes within 30 days of vaccination (9).
mRNA vaccines were also detected in a few heart autopsy samples
Interestingly, we also detected vaccine mRNA in the heart muscle of three people, all of whom had healing injury that occurred before or around the time of vaccination. This type of injury causes inflammation and attracts immune cells called monocytes to the heart where they become macrophages. Using immunohistochemistry, we counted the number of macrophages in the heart samples and found that those who had vaccine mRNA in their hearts had significantly more macrophages than those who did not. This suggests that the vaccine might have been carried by monocytes/macrophages to the heart as part of their normal function. Another possibility is that the heart damage was accompanied by changes in small blood vessels that made it easier for the vaccine to get into the heart muscle. The liver samples from these people did not show signs of similar healing injury. It is also possible that the vaccine particles went through or between the cells that line the blood vessels in areas of healthy heart tissue.
mRNA vaccine persistence
What does this mean for you? Our study provides a map of where and how long the SARS-CoV-2 mRNA vaccines stay in the body after vaccination. Given that the SARS-CoV-2 mRNA vaccine was detected in heart muscle with healing injury and that the effects are unclear at the present time, you might want to talk to your doctor before getting an LNP-based vaccine if you have recently had a heart attack. As many other LNP-based mRNA vaccines are developed and widely used (10-11), understanding how they behave in the body will help us improve their safety and effectiveness.
The study is entitled “Duration of SARS-CoV-2 mRNA vaccine persistence and factors associated with cardiac involvement in recently vaccinated patients” published in npj Vaccines, 8, 141, (2023). Link is below:
https://www.nature.com/articles/s41541-023-00742-7
REFERENCES:
- Pallesen J, Wang N, Corbett KS, et al. Immunogenicity and structures of a rationally designed prefusion MERS-CoV spike antigen. Proc Natl Acad Sci U S A. 2017;114(35):E7348-E7357. doi:10.1073/pnas.1707304114
- Andries O, Mc Cafferty S, De Smedt SC, Weiss R, Sanders NN, Kitada T. N(1)-methylpseudouridine-incorporated mRNA outperforms pseudouridine-incorporated mRNA by providing enhanced protein expression and reduced immunogenicity in mammalian cell lines and mice. J Control Release. 2015;217:337-344. doi:10.1016/j.jconrel.2015.08.051
- Meyer O, Kirpotin D, Hong K, et al. Cationic liposomes coated with polyethylene glycol as carriers for oligonucleotides. J Biol Chem. 1998;273(25):15621-15627. doi:10.1074/jbc.273.25.15621
- Hafez IM, Maurer N, Cullis PR. On the mechanism whereby cationic lipids promote intracellular delivery of polynucleic acids. Gene Ther. 2001;8(15):1188-1196. doi:10.1038/sj.gt.3301506
- Chen D, Love KT, Chen Y, et al. Rapid discovery of potent siRNA-containing lipid nanoparticles enabled by controlled microfluidic formulation. J Am Chem Soc. 2012;134(16):6948-6951. doi:10.1021/ja301621z
- Cullis PR, Hope MJ. Lipid Nanoparticle Systems for Enabling Gene Therapies. Mol Ther. 2017;25(7):1467-1475. doi:10.1016/j.ymthe.2017.03.013
- Tabor S, Richardson CC. A bacteriophage T7 RNA polymerase/promoter system for controlled exclusive expression of specific genes. Proc Natl Acad Sci U S A. 1985;82(4):1074-1078. doi:10.1073/pnas.82.4.1074
- Sample PJ, Wang B, Reid DW, Presnyak V, McFadyen IJ, Morris DR, Seelig G. Human 5' UTR design and variant effect prediction from a massively parallel translation assay. Nat Biotechnol. 2019 Jul;37(7):803-809. doi: 10.1038/s41587-019-0164-5. Epub 2019 Jul 1. PMID: 31267113; PMCID: PMC7100133.
- Röltgen K, Nielsen SCA, Silva O, et al. Immune imprinting, breadth of variant recognition, and germinal center response in human SARS-CoV-2 infection and vaccination. Cell. 2022;185(6):1025-1040.e14. doi:10.1016/j.cell.2022.01.018
- Miao L, Zhang Y, Huang L. mRNA vaccine for cancer immunotherapy. Mol Cancer. 2021;20(1):41. Published 2021 Feb 25. doi:10.1186/s12943-021-01335-5
- Xu X, Xia T. Recent Advances in Site-Specific Lipid Nanoparticles for mRNA Delivery. ACS Nanosci Au. 2023;3(3):192-203. Published 2023 Mar 30. doi:10.1021/acsnanoscienceau.2c0006
Follow the Topic
-
npj Vaccines
A multidisciplinary journal that is dedicated to publishing the finest and high-quality research and development on human and veterinary vaccines.
Related Collections
With Collections, you can get published faster and increase your visibility.
Therapeutic HPV vaccines
Publishing Model: Open Access
Deadline: Jun 30, 2026
Lipid nanoparticle (LNP)-adjuvanted vaccines
Publishing Model: Open Access
Deadline: May 19, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in