Dual-Functional Aerogel Assists in the Portable Biosensing
Published in Materials
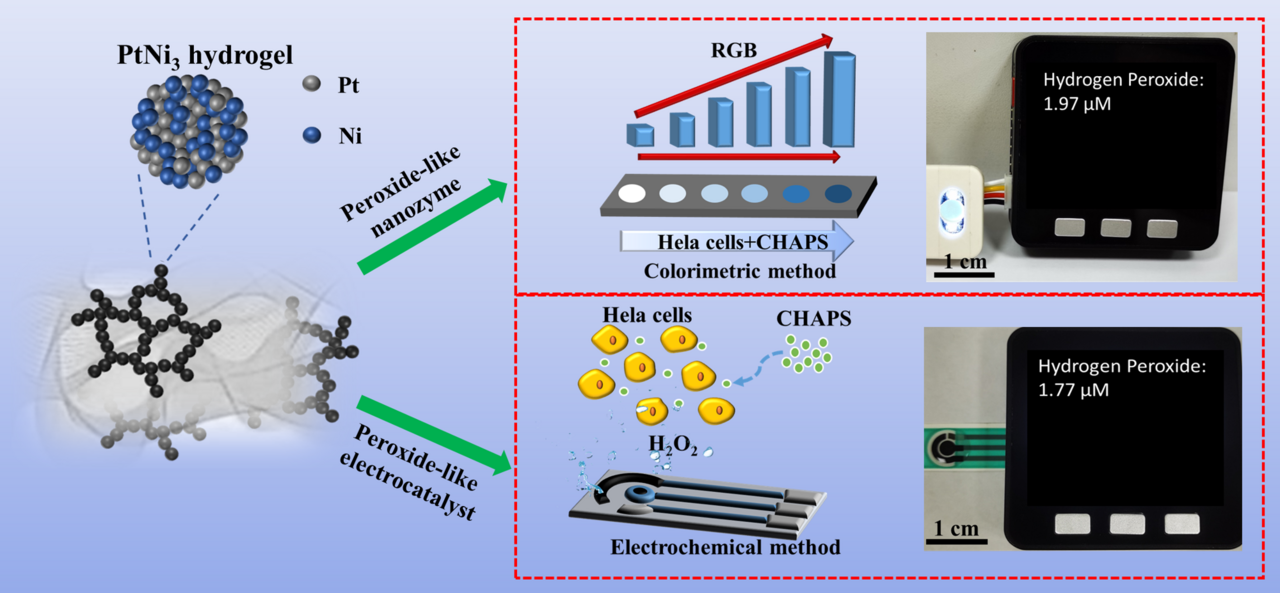
In this study, we developed the Pt-Ni hydrogels composed of the alloyed nanowires and Ni(OH)2 nanosheets, which had been demonstrated excellent peroxidase-like and electrocatalytic properties toward hydrogen peroxide (H2O2). On the basis of these characteristics, a portable dual-mode H2O2 sensor was successfully constructed (Figure 1).
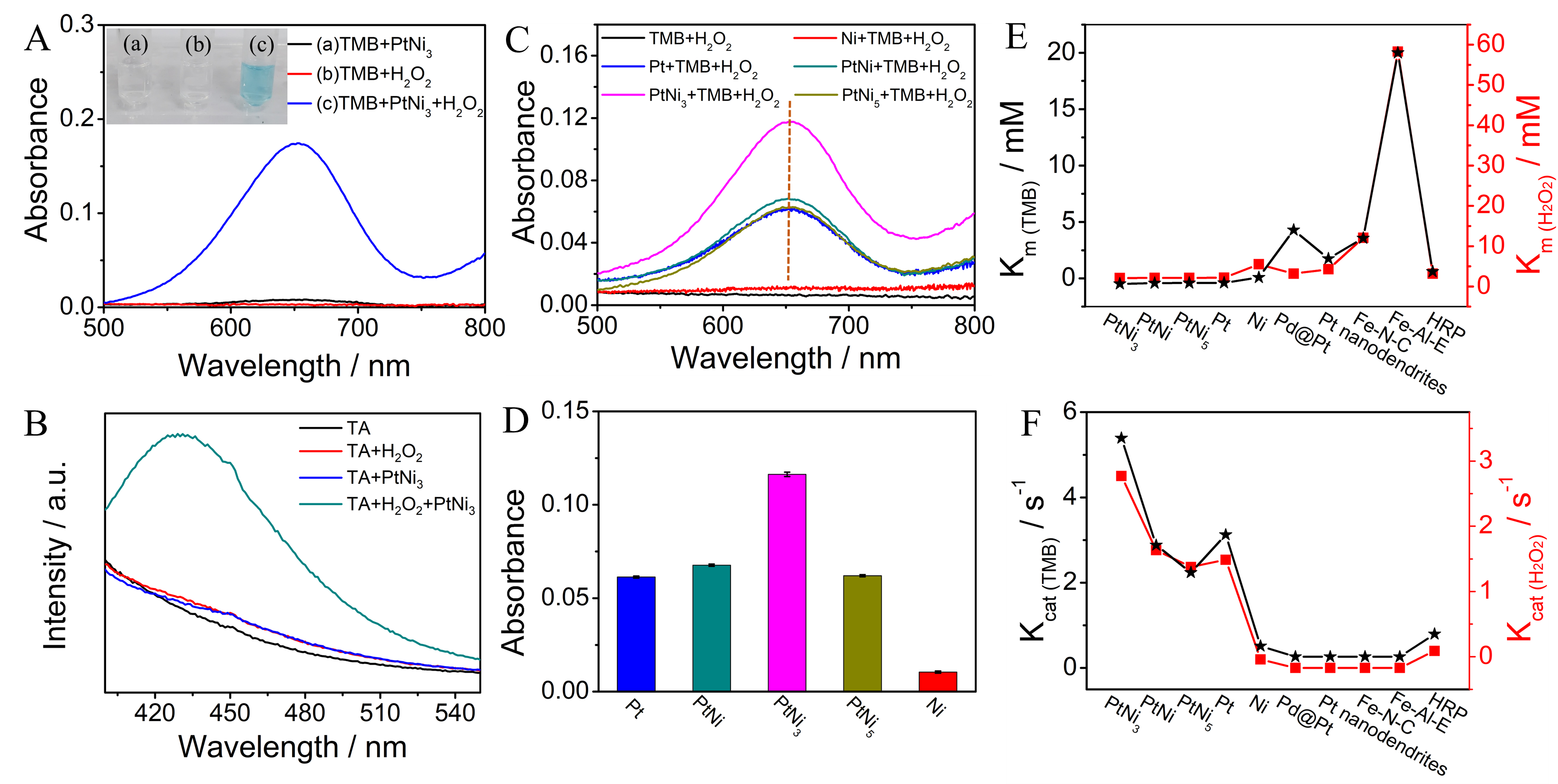
The microstructures of the Pt-Ni hydrogels have been deeply investigated in our previous work (Small, 19, 2206868). In this article, we mainly explored their catalytic performances toward H2O2, especially their peroxidase-like activities. As shown in Figure 2, we engineered the component of the Pt-Ni hydrogels to find out the optimal ratio of Pt and Ni elements. Finally, the PtNi3 hydrogel was select out due to its highest affinity and catalytic activity.
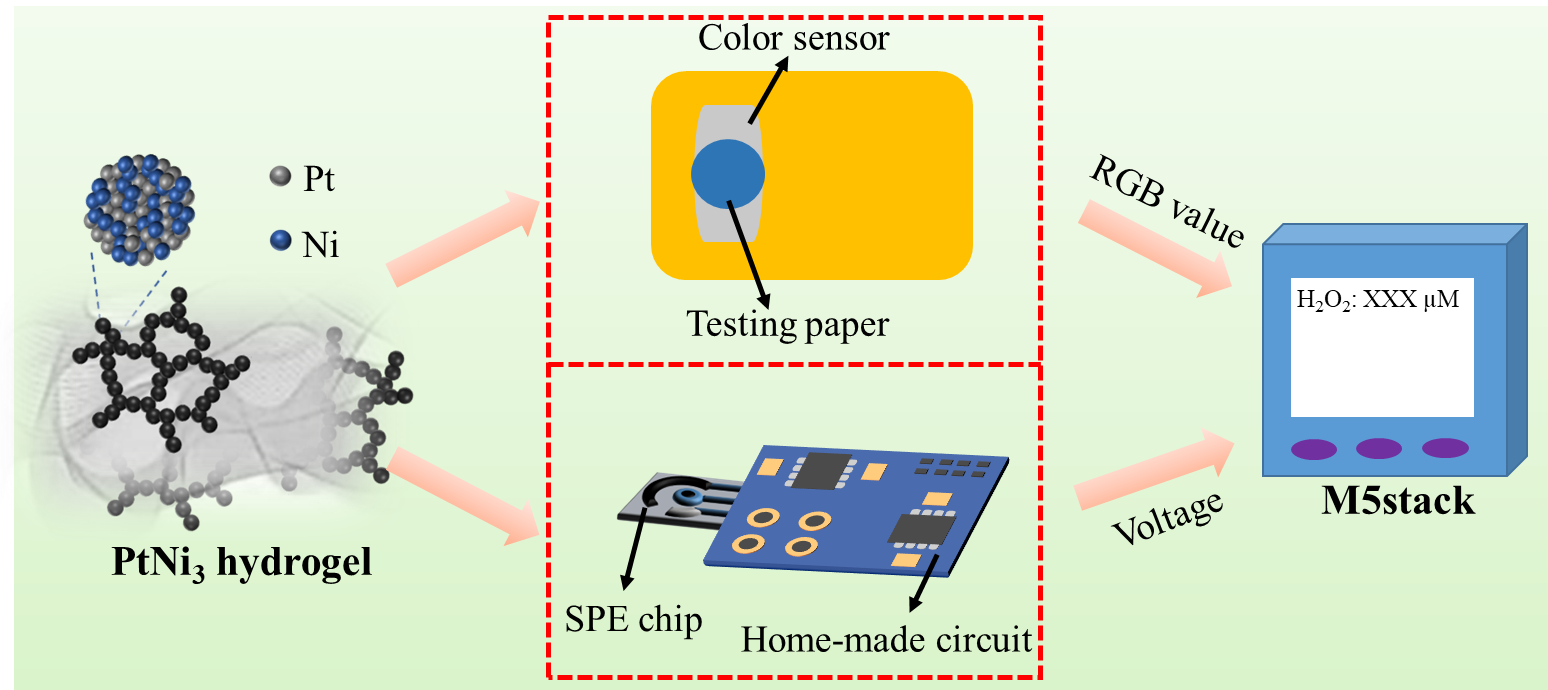
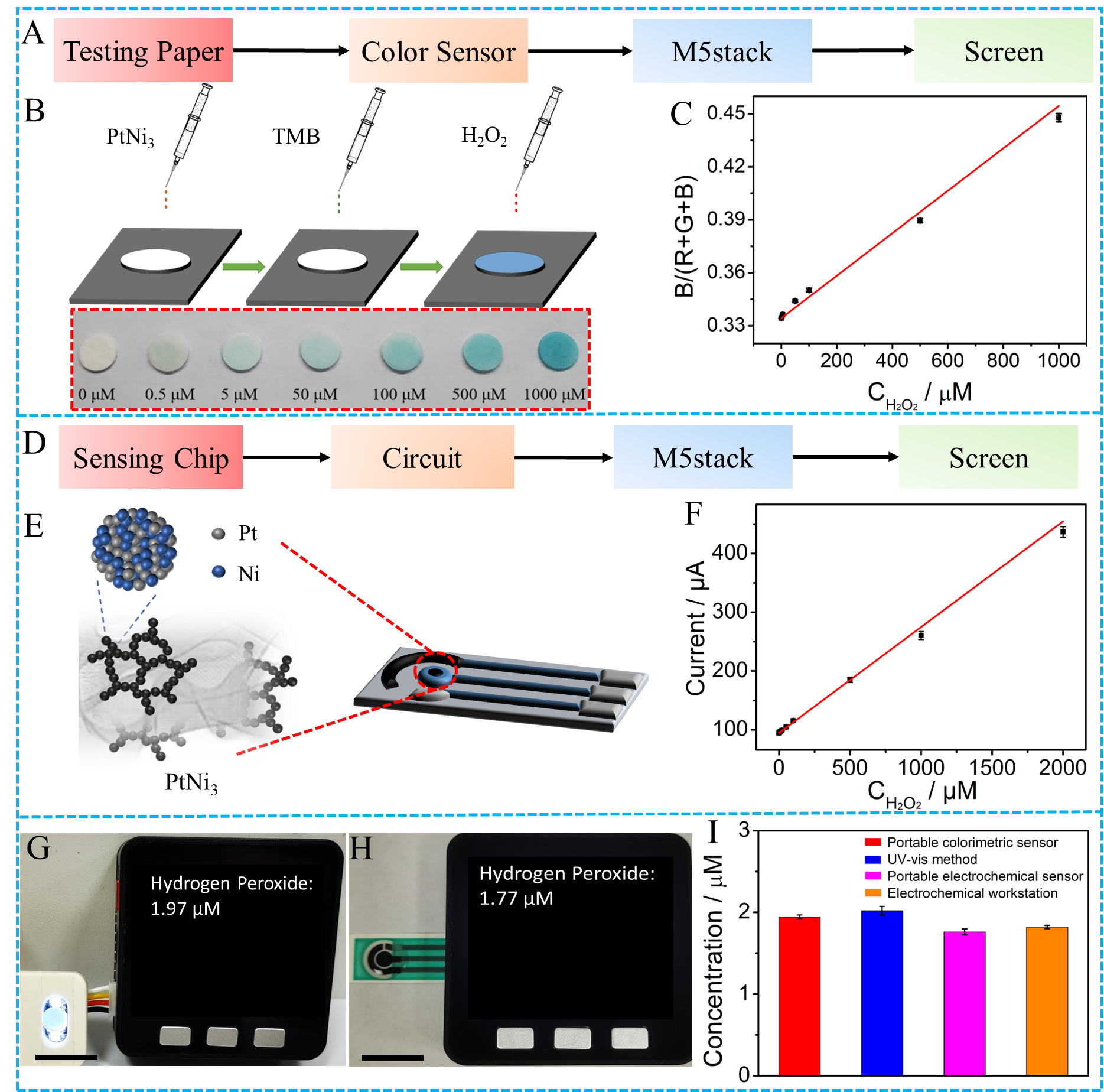
The outstanding electrocatalytic characteristics of the PtNi3 hydrogel toward H2O2 was also demonstrated in this work. Finally, we utilized the PtNi3 dual-structure hydrogel to construct a portable biosensor which can support both the visual and electrochemical testing modes (Figure 3). Considering the importance of H2O2 analysis in the living cells, we used these two modes of portable sensor to measure the H2O2 content in the CHAPS-stimulated HeLa cells. The testing results were verified by the standard instruments, demonstrating the reliability of the developed sensing platform.
You can read more about our work in the article in Microsystem & Nanoengineering following the link: https://www.nature.com/articles/s41378-023-00623-y
Follow the Topic
-
Microsystems & Nanoengineering
This journal, with a target for a high-end journal for years to come, seeks to promote research on all aspects of microsystems and nanoengineering from fundamental to applied research.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in